Advanced Synthetic Route for Epinastine Intermediate Manufacturing and Scale-up
Introduction to Patent CN114835712A
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance economic viability with stringent safety standards, particularly for antihistamine agents like Epinastine. Patent CN114835712A introduces a transformative synthetic methodology that fundamentally restructures the production landscape for this critical active pharmaceutical ingredient. By leveraging cheap and readily available 2-aminobenzophenone as the foundational starting material, this innovation bypasses the historical reliance on prohibitively expensive silane reagents and hazardous azide chemistry. The technical breakthrough lies in the strategic application of borohydride reduction coupled with Lewis acid catalysis, creating a streamlined sequence that achieves exceptional molar yields while adhering to modern industrial safety protocols. This report analyzes the mechanistic superiority and commercial implications of this route for global supply chain stakeholders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
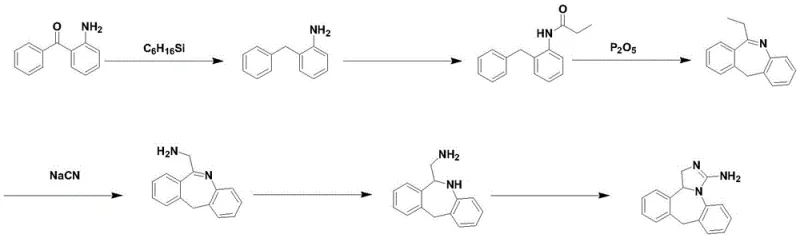
Historically, the industrial synthesis of Epinastine has been plagued by significant economic and safety bottlenecks that hinder scalable production. Prior art, such as the methods disclosed in CN103012408A, relied heavily on triethylsilane for the reduction of the ketone moiety, a reagent characterized by its exorbitant market price and volatility which severely erodes profit margins. Furthermore, these legacy processes necessitated the use of phosphorus pentachloride or phosphorus pentoxide for dehydration steps, chemicals known for their violent reactivity with water and substantial heat generation, posing acute thermal runaway risks in large-scale reactors. Perhaps most critically, traditional routes often employed sodium azide for nitrogen introduction, a compound infamous for its extreme toxicity and potential for catastrophic explosion, rendering it increasingly unacceptable under modern occupational health and safety regulations.
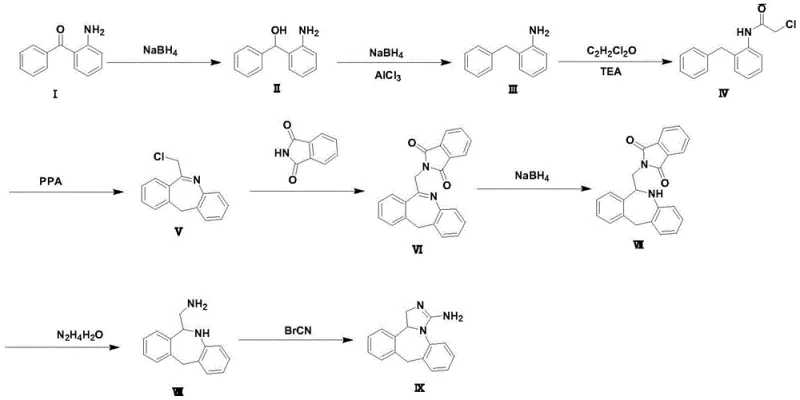
The Novel Approach
The methodology outlined in CN114835712A presents a paradigm shift by substituting these problematic reagents with benign and cost-effective alternatives. The core innovation involves a two-stage reduction strategy where sodium borohydride first reduces the ketone to an alcohol, followed by a Lewis acid-mediated deoxygenation using aluminum trichloride to yield 2-benzylaniline. This sequence completely eliminates the need for triethylsilane, resulting in drastic raw material cost optimization. Additionally, the cyclization step utilizes polyphosphoric acid (PPA) instead of phosphorus pentoxide, offering a controlled and safer dehydration environment. The final ring closure is achieved through a mild reaction with cyanogen bromide, avoiding the high-pressure hydrogenation equipment and palladium catalysts required in other competing patents like CN101130544, thereby simplifying the engineering requirements for commercial manufacturing facilities.
Mechanistic Insights into Lewis Acid-Catalyzed Reduction and Cyclization
The chemical elegance of this synthesis is rooted in the precise manipulation of reduction potentials and electrophilic activation. In the conversion of Compound II to Compound III, the system employs a synergistic interaction between sodium borohydride and aluminum trichloride. The Lewis acid coordinates with the hydroxyl group of the intermediate alcohol, enhancing its leaving group ability and facilitating a hydride attack that effectively deoxygenates the benzylic position. This mechanism operates efficiently at moderate temperatures of 60-70°C, avoiding the harsh conditions typical of catalytic hydrogenation. Subsequently, the formation of the seven-membered dibenzazepine ring (Compound V) is driven by the strong dehydrating capability of polyphosphoric acid, which promotes intramolecular nucleophilic attack of the amide nitrogen onto the activated benzyl carbon. This cyclization proceeds with high regioselectivity, minimizing the formation of polymeric byproducts and ensuring a clean impurity profile that simplifies downstream purification.
Impurity control is further enhanced by the choice of reagents in the later stages. The reduction of the imide double bond in Compound VI to Compound VII utilizes sodium borohydride in a methanol-dichloromethane system, a mild condition that prevents over-reduction or ring opening of the sensitive azepine core. The final hydrolysis of the phthalimide protecting group using hydrazine hydrate is a classic yet highly effective strategy that releases the primary amine (Compound VIII) without affecting the newly formed heterocyclic ring. This careful orchestration of reaction conditions ensures that the final Epinastine product (Compound IX) achieves an HPLC purity of 99.848% as demonstrated in Example 1, meeting the rigorous quality standards required for pharmaceutical registration.
How to Synthesize Epinastine Efficiently
The synthesis of Epinastine via this patented route involves a sequential eight-step process that transforms 2-aminobenzophenone into the final active ingredient through a series of reduction, acylation, cyclization, and condensation reactions. Each step has been optimized for maximum yield and minimal waste, utilizing standard laboratory equipment that translates easily to pilot and production scales. The detailed standardized synthesis steps are provided in the guide below.
- Reduce 2-aminobenzophenone to (2-aminophenyl)(phenyl)methanol using sodium borohydride in ethanol at 75-80°C.
- Perform Lewis acid catalyzed reduction with AlCl3 and NaBH4 to obtain 2-benzylaniline, followed by acylation with chloroacetyl chloride.
- Cyclize using polyphosphoric acid (PPA) to form the dibenzazepine core, followed by phthalimide substitution and final ring closure with cyanogen bromide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling strategic advantages centered around cost stability and operational reliability. By decoupling the production process from volatile specialty reagents like triethylsilane and palladium on carbon, manufacturers can secure a more predictable cost structure that is less susceptible to market fluctuations in rare metal or organosilicon pricing. The elimination of high-pressure hydrogenation steps removes the need for specialized autoclaves and the associated maintenance costs, allowing production to occur in standard glass-lined or stainless steel reactors found in most multipurpose chemical facilities. This flexibility significantly enhances supply chain resilience, enabling faster response times to market demand surges without the bottleneck of equipment availability.
- Cost Reduction in Manufacturing: The substitution of expensive silane reagents with commodity chemicals like sodium borohydride and aluminum trichloride results in substantial raw material savings. Furthermore, the avoidance of noble metal catalysts eliminates the complex and costly recovery processes typically associated with palladium removal, streamlining the overall production economics and reducing the total cost of goods sold significantly.
- Enhanced Supply Chain Reliability: The starting material, 2-aminobenzophenone, is a widely available bulk chemical with a stable global supply network, ensuring consistent feedstock availability. The simplified reaction conditions, which do not require cryogenic temperatures or ultra-high pressures, reduce the risk of unplanned shutdowns due to equipment failure, thereby guaranteeing more reliable delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The process avoids the generation of heavy metal waste and toxic azide byproducts, drastically simplifying wastewater treatment and hazardous waste disposal protocols. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental compliance burden, making the technology highly scalable from kilogram to multi-ton production volumes without prohibitive infrastructure investments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Epinastine synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on yield expectations, safety profiles, and scalability factors for potential licensees and manufacturing partners.
Q: How does this new synthesis route improve safety compared to conventional methods?
A: The novel process eliminates the use of highly toxic and explosive sodium azide, dangerous phosphorus pentoxide, and expensive triethylsilane. It replaces high-pressure hydrogenation with mild borohydride reduction, significantly lowering operational risks and EHS compliance burdens.
Q: What are the purity specifications achievable with this manufacturing method?
A: Experimental data demonstrates consistent HPLC purity exceeding 99.8% for the final Epinastine product (Compound IX), with intermediate purities maintained above 99.4% throughout the critical cyclization and reduction steps.
Q: Why is the replacement of triethylsilane significant for cost reduction?
A: Triethylsilane is a high-cost reagent that drastically impacts the bill of materials. By substituting it with inexpensive sodium borohydride and aluminum trichloride, the overall production cost is substantially reduced without compromising yield or reaction efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epinastine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest international standards.
We invite you to collaborate with us to leverage this innovative Epinastine synthesis route for your supply chain needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and efficiency for your organization.
