Advanced Visible-Light Photocatalysis for High-Purity Carbazole Derivatives Manufacturing
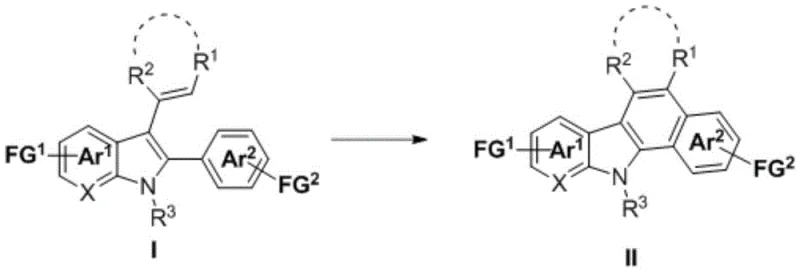
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient methods for constructing complex heterocyclic scaffolds, which serve as the backbone for numerous bioactive molecules. Patent CN116178243A introduces a groundbreaking preparation method for (hetero)aryl fused carbazole compounds that leverages visible light photocatalysis to achieve cyclization and dehydroaromatization under exceptionally mild conditions. This technology represents a significant paradigm shift from traditional thermal or high-energy UV methods, offering a pathway to access structurally diverse carbazole derivatives with high precision and minimal environmental impact. By utilizing alkenyl-substituted indoles as substrates, this process facilitates an energy transfer mechanism that drives the formation of the rigid carbazole core without the need for harsh reagents or extreme temperatures. For R&D directors and procurement specialists, this innovation promises not only enhanced synthetic flexibility but also a robust solution for scaling the production of high-purity pharmaceutical intermediates and optoelectronic materials.
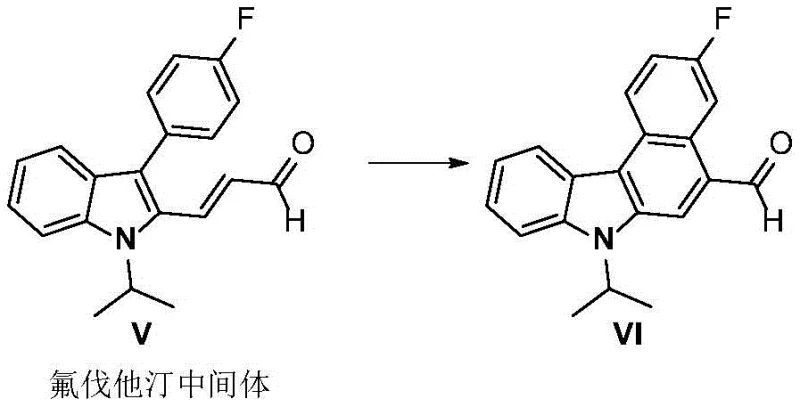
The practical applicability of this method is further underscored by its successful deployment in the synthesis of critical drug intermediates, such as those found in lipid-lowering agents like Fluvastatin. The ability to perform these transformations with such high fidelity ensures that supply chains for complex active pharmaceutical ingredients (APIs) can be stabilized and optimized. As a reliable pharmaceutical intermediates supplier, understanding and adopting such advanced synthetic methodologies is crucial for maintaining competitiveness in a market that demands both speed and quality. The transition to visible-light-driven chemistry aligns perfectly with global trends towards greener manufacturing processes, reducing the carbon footprint associated with the production of high-value heterocyclic compounds while simultaneously improving operator safety by eliminating hazardous UV radiation sources.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl-fused carbazoles from 2-aryl-3-enylindole derivatives has relied heavily on photochemical cyclization-dehydroaromatization reactions driven by high-pressure mercury lamps emitting ultraviolet (UV) light. These conventional protocols suffer from severe drawbacks that hinder their widespread adoption in modern industrial settings, primarily due to their excessive energy consumption, often requiring power inputs as high as 500W to sustain the reaction. Furthermore, the high energy of UV photons frequently leads to poor functional group compatibility, causing degradation of sensitive moieties such as esters, aldehydes, or amines, which necessitates additional protection and deprotection steps that inflate costs and extend timelines. From a safety perspective, the operation of high-intensity UV sources mandates rigorous protective measures to prevent skin and eye damage to laboratory personnel, adding layers of operational complexity and regulatory compliance burdens that slow down the commercial scale-up of complex heterocyclic compounds.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a visible light-induced energy transfer process mediated by specific photocatalysts, operating at a mere 30W power input with blue LEDs emitting at wavelengths between 390-484nm. This drastic reduction in energy demand, coupled with the use of ambient temperature conditions (25°C), creates a significantly milder reaction environment that preserves the integrity of sensitive functional groups without the need for extensive substrate modification. The method employs iridium-based photocatalysts, such as Ir(dFCF3ppy)2(dtbbpy)PF6, which efficiently absorb visible light and transfer triplet energy to the substrate, triggering the desired cyclization with remarkable efficiency. This technological leap not only enhances the safety profile of the manufacturing process by removing hazardous UV exposure but also streamlines the workflow, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing where yield and purity are paramount.
Mechanistic Insights into Visible-Light Photocatalytic Cyclization
The core of this innovative synthesis lies in the sophisticated photophysical mechanism wherein the photocatalyst acts as a mediator for triplet-triplet energy transfer. Upon irradiation with blue light, the iridium complex absorbs photons and transitions to an excited triplet state, which then transfers its energy to the ground-state alkenyl-indole substrate. This energy transfer promotes the substrate to a reactive triplet state, facilitating an intramolecular cyclization that forms the new carbon-carbon bond essential for the carbazole framework. Following the cyclization event, a spontaneous or catalyst-assisted dehydroaromatization occurs, restoring aromaticity to the newly formed ring system and yielding the stable fused carbazole product. This mechanism bypasses the high-energy barriers associated with thermal cyclization and avoids the non-selective bond cleavage often observed with direct UV excitation, thereby ensuring a cleaner reaction profile with fewer by-products.
From an impurity control perspective, the mildness of the visible light conditions plays a pivotal role in maintaining high product purity. Traditional UV methods often generate radical species that can attack various parts of the molecule, leading to a complex mixture of side products that are difficult to separate. In this visible-light protocol, the selective energy transfer ensures that only the specific alkene-indole moiety is activated, leaving other functional groups like carboxyl, aldehyde, and alkylamine groups untouched. This high degree of chemoselectivity simplifies the downstream purification process, typically requiring only standard column chromatography to achieve analytical purity. For quality control teams, this means more consistent batch-to-batch reproducibility and a reduced risk of genotoxic impurities that might arise from harsher reaction conditions, ultimately supporting the stringent purity specifications required for clinical-grade materials.
How to Synthesize (Hetero)aryl Fused Carbazole Efficiently
The synthesis protocol outlined in the patent provides a straightforward and reproducible procedure for accessing these valuable scaffolds, beginning with the dissolution of the 2-aryl-3-enylindole substrate and the photocatalyst in a binary solvent system. The reaction is conducted under an inert argon atmosphere to prevent oxygen quenching of the excited states, ensuring maximum catalytic turnover. The mixture is then irradiated with 30W blue LEDs at a controlled distance, maintaining the temperature at 25°C for approximately 18 hours to allow for complete conversion. Detailed standardized synthesis steps are provided in the guide below to assist process chemists in replicating these results effectively.
- Prepare the reaction mixture by dissolving the 2-aryl-3-enylindole substrate and the iridium-based photocatalyst in a mixed solvent system of dichloromethane and dimethyl sulfoxide.
- Place the reaction vessel under an inert argon atmosphere and irradiate with 30W blue LEDs (wavelength 440nm) at room temperature (25°C) for approximately 18 hours.
- Upon completion, perform aqueous workup with ethyl acetate extraction, concentrate the organic phase, and purify the crude product via column chromatography to obtain the target carbazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible-light photocatalytic method offers substantial strategic advantages that directly impact the bottom line and operational resilience. The shift from 500W mercury lamps to 30W LED arrays represents a dramatic decrease in energy consumption, which translates to significantly reduced utility costs over the lifecycle of large-scale production runs. Moreover, the elimination of high-energy UV radiation removes the need for specialized shielding and safety protocols, lowering the capital expenditure required for reactor setup and reducing the administrative burden associated with workplace safety compliance. These factors combined create a more agile manufacturing environment where resources can be allocated more efficiently towards innovation and capacity expansion rather than maintenance and hazard mitigation.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive high-pressure mercury lamps and the associated cooling systems required to manage their heat output. By operating at room temperature with low-power LEDs, the process drastically simplifies the reactor design and reduces the overall energy load, leading to substantial cost savings in utilities. Additionally, the superior functional group compatibility means that fewer synthetic steps are needed to protect sensitive groups, reducing the consumption of reagents and solvents, which further lowers the cost of goods sold (COGS) for the final active ingredient.
- Enhanced Supply Chain Reliability: The mild reaction conditions and the use of commercially available iridium catalysts ensure a robust supply chain that is less susceptible to disruptions caused by specialized equipment failures or hazardous material handling restrictions. The ability to synthesize complex intermediates like the Fluvastatin precursor with high reliability means that downstream API production schedules can be met with greater certainty. This stability is crucial for long-term contracts with pharmaceutical partners who require guaranteed continuity of supply for their critical drug pipelines, thereby strengthening the manufacturer's position as a trusted partner.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging due to light penetration issues, but the use of low-energy visible light and flow-compatible LED setups makes this process highly amenable to scale-up from gram to ton quantities. The reduced energy footprint and the absence of toxic UV waste streams align perfectly with increasingly strict environmental regulations, facilitating smoother permitting processes for new manufacturing facilities. This environmental compliance not only mitigates regulatory risk but also enhances the brand reputation of the supplier as a leader in green chemistry and sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light photocatalytic technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows and for assessing the quality standards achievable with this new synthetic route.
Q: What are the advantages of using visible light over UV light for carbazole synthesis?
A: Visible light photocatalysis eliminates the need for high-energy, high-consumption mercury lamps (500W), reducing energy costs and safety risks associated with UV exposure. It also offers superior functional group compatibility, allowing sensitive groups like aldehydes and carboxylic acids to remain intact during the reaction.
Q: Which photocatalysts are effective for this transformation?
A: The patent specifies iridium-based complexes such as Ir(dFCF3ppy)2(dtbbpy)PF6 and fac-Ir(ppy)3 as highly effective photocatalysts capable of absorbing visible light and transferring triplet energy to the substrate to drive the cyclization.
Q: Can this method be applied to drug intermediates like Fluvastatin?
A: Yes, the methodology is explicitly demonstrated for the cyclodehydroaromatization of indole-based drug intermediates, including the synthesis of Fluvastatin intermediates and Smad3 inhibitors, proving its utility in late-stage pharmaceutical modification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible-light photocatalysis in the synthesis of high-value heterocyclic compounds, and we are fully equipped to leverage this technology for our clients' most challenging projects. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the identity and quality of every batch of carbazole derivatives produced, guaranteeing that our materials meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to explore how this advanced synthesis method can optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your development goals and accelerate your time to market.
