Advanced Catalytic Oxidation for High-Purity Fluorine-Containing Glycine Ester Derivatives
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways to introduce fluorine atoms into organic scaffolds, given the profound impact of fluorination on metabolic stability and bioavailability. Patent CN109081785B introduces a groundbreaking synthetic method for fluorine-containing glycine ester derivatives that addresses critical bottlenecks in traditional organic synthesis. This technology leverages a copper-catalyzed oxidative coupling strategy, utilizing molecular oxygen as a terminal oxidant to drive the reaction between glycine ester derivatives and difluoroenol silyl ethers. By shifting away from hazardous halogenated reagents and stoichiometric oxidants, this method represents a significant leap forward in green chemistry, offering a reliable fluorine-containing glycine ester derivative supplier pathway that aligns with modern sustainability goals. The process operates under mild conditions, specifically at room temperature, which not only preserves the integrity of sensitive functional groups but also drastically reduces energy consumption during large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of geminal difluoromethyl compounds has been plagued by significant operational and safety challenges that hinder scalable production. Traditional routes often rely on Reformatsky addition reactions using bromodifluoroacetic acid ethyl ester, a reagent known for its high toxicity and instability, posing severe risks to personnel and requiring specialized containment infrastructure. Furthermore, alternative methods involving alpha-halogenated glycine esters necessitate multi-step preparations that generate substantial amounts of toxic waste, creating a heavy burden on environmental compliance and waste treatment budgets. Another common approach requires the condensation of beta-amino acids with difluoroalkylating reagents followed by sigma-rearrangement, a process that demands harsh conditions such as strong alkalis and cryogenic temperatures down to -78°C. These extreme conditions not only inflate utility costs but also limit the substrate scope, making it difficult to apply these methods to complex, functionalized molecules required in advanced drug discovery pipelines.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN109081785B offers a streamlined, direct oxidative coupling route that eliminates the need for pre-functionalized imine substrates. By employing copper trifluoromethanesulfonate as a catalyst and oxygen from the air as the oxidant, this method achieves high atom economy and operational simplicity. The reaction proceeds efficiently at room temperature in dichloromethane, avoiding the energy-intensive cooling and heating cycles associated with prior art. This novel approach allows for the direct modification of glycine ester hydrocarbons, bypassing the tedious and yield-limiting steps of imine preparation. As illustrated in the reaction scheme below, the coupling of the glycine derivative with the difluoroenol silyl ether occurs smoothly, delivering the target fluorinated scaffold with impressive efficiency.
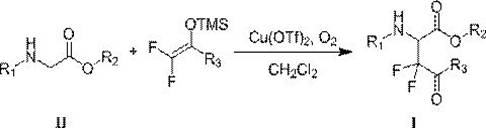
The versatility of this new methodology is evidenced by its broad substrate tolerance, accommodating various N-aryl glycine esters with electron-donating or electron-withdrawing groups. This flexibility is crucial for medicinal chemists who need to rapidly iterate on lead compounds without being constrained by synthetic feasibility. Moreover, the use of oxygen as a green oxidant ensures that the only byproduct is water, significantly simplifying the downstream purification process and reducing the environmental footprint of the manufacturing operation. This positions the technology as a superior choice for cost reduction in pharmaceutical intermediates manufacturing, where waste disposal and raw material costs are primary drivers of the final price.
Mechanistic Insights into Cu(OTf)2-Catalyzed Oxidative Coupling
The core of this innovation lies in the unique role of the copper catalyst in activating both the oxygen oxidant and the glycine substrate for C-H functionalization. The mechanism likely involves the formation of a copper-oxygen species that facilitates the single-electron oxidation of the glycine ester derivative, generating a reactive radical or cationic intermediate at the alpha-position. This activated species then undergoes nucleophilic attack by the difluoroenol silyl ether, forging the critical carbon-carbon bond that installs the geminal difluoromethyl ketone moiety. The use of Cu(OTf)2 is particularly advantageous due to its Lewis acidity and redox potential, which are finely tuned to promote this transformation without degrading the sensitive fluorine-containing components. Experimental data from the patent highlights that maintaining an oxygen atmosphere is critical; reactions conducted under nitrogen yielded only 31%, whereas oxygen-saturated conditions pushed yields significantly higher, confirming the essential role of O2 in the catalytic cycle.
From an impurity control perspective, the mild reaction conditions play a pivotal role in ensuring high product purity. Traditional methods utilizing strong bases or high temperatures often lead to elimination side reactions, racemization of chiral centers, or decomposition of the fluorinated motifs. By operating at room temperature, this catalytic system minimizes thermal degradation pathways, resulting in a cleaner crude reaction profile. The patent data demonstrates that optimizing the catalyst loading from 0.05 equivalents to 0.2 equivalents can boost yields from 57% to 93%, indicating a robust and controllable process window. This level of control is essential for producing high-purity pharmaceutical intermediates that meet stringent regulatory standards, as it reduces the burden on downstream purification steps like column chromatography or recrystallization, thereby enhancing overall process throughput.
How to Synthesize Fluorine-Containing Glycine Ester Derivatives Efficiently
To implement this synthesis effectively, operators must adhere to precise protocols regarding catalyst activation and gas flow management to ensure reproducibility. The process begins with the dissolution of the copper catalyst and substrate in dichloromethane, followed by a crucial stirring period under oxygen to establish the active catalytic species before the introduction of the coupling partner. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these high-yielding results.
- Prepare the reaction system by adding copper trifluoromethanesulfonate (0.1-0.2 equiv) and glycine ester derivatives into a reactor with dichloromethane as the solvent.
- Stir the mixture under an oxygen atmosphere for at least 30 minutes to ensure catalyst activation and oxygen saturation before adding the difluoroenol silyl ether.
- Add difluoroenol silyl ether (1.0-1.2 equiv) and react at room temperature for over 12 hours, followed by standard workup including brine wash, extraction, and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond simple chemical transformation. The elimination of expensive and hazardous stoichiometric oxidants like DDQ or bromodifluoroacetates directly translates to substantial cost savings in raw material procurement. Furthermore, the removal of the imine pre-preparation step consolidates what was previously a multi-stage process into a single pot operation, drastically reducing labor hours, solvent consumption, and reactor occupancy time. This streamlining of the workflow enhances the overall equipment effectiveness (OEE) of manufacturing facilities, allowing for greater production capacity without the need for capital expenditure on new reactors. The reliance on oxygen, an abundant and inexpensive resource, further insulates the supply chain from volatility in the pricing of specialized chemical oxidants.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the replacement of high-cost reagents with catalytic systems and atmospheric oxygen. By avoiding the purchase of toxic halogenated difluoroalkylation reagents and eliminating the separate synthesis of imine intermediates, the total cost of goods sold (COGS) is significantly lowered. Additionally, the mild conditions reduce energy costs associated with heating and cooling, while the simplified workup procedure minimizes solvent usage and waste disposal fees. These cumulative efficiencies create a leaner manufacturing process that improves margin potential for high-volume commercial production.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key reagents—glycine esters and silyl ethers—are commodity chemicals with robust global supply chains, unlike niche fluorinating agents that may face availability bottlenecks. The simplicity of the reaction setup, requiring only standard glassware and oxygen lines rather than specialized cryogenic or high-pressure equipment, means that production can be easily distributed across multiple manufacturing sites to mitigate risk. This decentralization capability ensures business continuity and reduces lead time for high-purity fluorine-containing glycine ester derivatives, allowing partners to respond more agilely to market demand fluctuations.
- Scalability and Environmental Compliance: The green nature of this chemistry aligns perfectly with increasingly strict environmental regulations, reducing the regulatory burden on manufacturing sites. The absence of heavy metal waste (beyond trace catalyst levels) and toxic halogenated byproducts simplifies effluent treatment and lowers compliance costs. The process has been demonstrated to be scalable to gram-level production in the patent examples with maintained efficiency, suggesting a clear path to kilogram and ton-scale commercialization. This scalability ensures that the supply chain can grow seamlessly alongside the clinical development of the drug candidate, preventing supply disruptions during critical scale-up phases.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential partners.
Q: What are the advantages of using oxygen over traditional oxidants in this synthesis?
A: According to patent CN109081785B, using oxygen as a green oxidant avoids the use of equivalent chemical oxidants like DDQ, making the process simpler, greener, and safer while significantly reducing raw material costs compared to stoichiometric oxidants.
Q: Is pre-preparation of imine substrates required for this reaction?
A: No, a key advantage of this novel approach is that it avoids the pre-preparation of imines. It directly utilizes glycine ester hydrocarbons for modification under catalytic transition metal conditions, meeting atom economy requirements and simplifying the workflow.
Q: What is the optimal catalyst loading for maximum yield?
A: Experimental data indicates that a molar ratio of copper trifluoromethanesulfonate to glycine ester derivative of 0.2 yields up to 93%, whereas reducing the loading to 0.05 drops the yield significantly to 57%, suggesting 0.1-0.2 equivalents is the optimal range.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorine-Containing Glycine Ester Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed oxidative coupling technology for the next generation of fluorinated therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of fluorine-containing glycine ester derivatives meets the highest quality standards required by global regulatory bodies. We are committed to leveraging our technical expertise to optimize this green synthesis route for your specific application needs.
We invite you to engage with our technical procurement team to discuss how this innovative method can enhance your project's viability. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as your trusted partner in advancing complex fluorinated chemistry.
