Advanced Iridium-Catalyzed N-Alkylation for Scalable 2-(N-Alkyl)aminopyrimidine Production
The pharmaceutical and agrochemical industries are constantly seeking more efficient and environmentally benign pathways to access privileged heterocyclic scaffolds. Patent CN102491949A introduces a groundbreaking methodology for the synthesis of 2-(N-alkyl)aminopyrimidine derivatives, a class of compounds renowned for their broad biological activities ranging from kinase inhibition to receptor antagonism. This innovation shifts the paradigm from traditional, waste-intensive halogenation routes to a direct, catalytic N-alkylation strategy using simple alcohols. By leveraging transition metal catalysis, specifically utilizing complexes of Iridium, Rhodium, or Ruthenium, this process achieves high atom economy and exceptional selectivity. For R&D directors and procurement specialists, this technology represents a significant leap forward in sustainable manufacturing, offering a reliable route to high-purity pharmaceutical intermediates while drastically reducing the environmental footprint associated with legacy synthetic methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(N-alkyl)aminopyrimidine derivatives has relied heavily on nucleophilic aromatic substitution (SNAr) reactions. This conventional approach necessitates the preparation of 2-halogenated aminopyrimidine precursors, a multi-step process that inherently introduces significant inefficiencies and safety hazards. The halogenation step typically requires the use of stoichiometric amounts of highly toxic and corrosive reagents such as phosphorus oxychloride (POCl3) or thionyl chloride (SOCl2). These reagents not only pose severe handling risks for operators but also generate substantial quantities of acidic waste streams that require costly neutralization and disposal procedures. Furthermore, the SNAr reaction often struggles with selectivity issues, potentially leading to mixtures of mono- and di-alkylated products or unwanted alkylation on the ring nitrogens, thereby complicating downstream purification and reducing overall yield. The reliance on halogenated starting materials also ties the supply chain to volatile commodity markets for halogenating agents, introducing potential bottlenecks in production continuity.
The Novel Approach
In stark contrast, the method disclosed in CN102491949A utilizes a direct N-alkylation strategy where commercially available or easily synthesized 2-aminopyrimidine derivatives react directly with alcohols. This approach eliminates the need for pre-functionalization with halogens, thereby bypassing the use of toxic chlorinating agents entirely. The reaction is catalyzed by transition metal complexes and proceeds under relatively mild thermal conditions, typically between 120°C and 150°C. A defining feature of this novel pathway is its green chemistry profile; the only by-product generated is water, which simplifies work-up procedures and eliminates the burden of hazardous waste treatment. The process demonstrates remarkable robustness across a wide range of substrates, including various alkyl, aryl, and substituted alcohols, making it a versatile platform for generating diverse chemical libraries. This shift from halide-based chemistry to alcohol-based alkylation fundamentally alters the cost structure and safety profile of manufacturing these valuable intermediates.
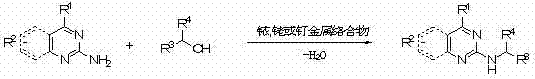
Mechanistic Insights into Transition Metal-Catalyzed N-Alkylation
The core of this technological advancement lies in the utilization of transition metal catalysts, such as pentamethylcyclopentadienyl iridium dichloride dimer ([Cp*IrCl2]2), to facilitate the coupling of amines and alcohols. While the exact mechanistic cycle can vary depending on the specific metal center, the process generally operates through a borrowing hydrogen or hydrogen autotransfer mechanism. In this cycle, the metal catalyst first dehydrogenates the alcohol to generate a reactive aldehyde intermediate and a metal-hydride species. The aldehyde then undergoes condensation with the amine substrate to form an imine intermediate. Subsequently, the metal-hydride species reduces the imine back to the saturated amine product, regenerating the active catalyst. This elegant cascade allows for the formation of the C-N bond without the need for external reducing agents or activating groups. The choice of base, such as sodium hydroxide or potassium carbonate, plays a critical role in facilitating the dehydrogenation step and maintaining the catalytic cycle's efficiency. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters for specific substrate classes.
From an impurity control perspective, this catalytic system offers distinct advantages over uncatalyzed thermal alkylation. The patent data highlights complete regioselectivity and chemoselectivity, meaning the reaction exclusively targets the exocyclic amino group at the 2-position of the pyrimidine ring. There is no observed alkylation of the endocyclic nitrogen atoms within the aromatic heterocycle, nor is there significant formation of over-alkylated 2-(N,N-dialkyl) by-products. This high level of specificity is crucial for pharmaceutical applications where impurity profiles are strictly regulated. The ability to suppress side reactions reduces the complexity of the crude reaction mixture, allowing for simpler purification protocols such as standard column chromatography or crystallization. This selectivity is attributed to the specific coordination environment of the transition metal catalyst, which directs the reactivity towards the desired mono-alkylation pathway, ensuring consistent product quality batch after batch.
How to Synthesize 2-(N-Alkyl)aminopyrimidine Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction conditions and reagent ratios to maximize yield and purity. The general procedure involves charging a reaction vessel with the 2-aminopyrimidine substrate, a selected transition metal catalyst, a base, and the desired alcohol alkylating agent. The reaction can be conducted under either nitrogen protection or in air, offering operational flexibility. Typical catalyst loadings range from 0.05 to 0.2 mol%, demonstrating the high turnover efficiency of the metal complexes. The mixture is then heated to temperatures between 120°C and 150°C for a duration of 1 to 12 hours, depending on the reactivity of the specific alcohol used. Upon completion, the solvent is removed via rotary evaporation, and the crude product is purified. Detailed standardized synthesis steps follow below.
- Combine 2-aminopyrimidine derivatives, transition metal catalyst (e.g., [Cp*IrCl2]2), base, and alcohol in a reaction vessel under nitrogen or air.
- Heat the reaction mixture to 120-150°C and maintain for 1-12 hours to facilitate the N-alkylation process.
- Cool to room temperature, remove solvent via rotary evaporation, and purify the target compound using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic N-alkylation technology translates into tangible strategic benefits beyond mere technical novelty. The primary advantage lies in the drastic simplification of the raw material supply chain. By replacing specialized, hazardous halogenated intermediates with commodity alcohols, manufacturers can leverage existing, stable supply networks for bulk chemicals. Alcohols such as n-butanol, n-hexanol, and benzyl alcohol are produced on a massive industrial scale, ensuring consistent availability and shielding the production process from the supply volatility often associated with fine chemical precursors. Furthermore, the elimination of toxic reagents like phosphorus oxychloride reduces the regulatory burden and insurance costs associated with storing and handling hazardous materials, contributing to a safer and more compliant operational environment.
- Cost Reduction in Manufacturing: The economic implications of this process are profound. By removing the multi-step halogenation sequence, the overall number of unit operations is reduced, leading to lower labor and utility costs. The high atom economy of the reaction ensures that a greater proportion of the starting mass ends up in the final product, minimizing raw material waste. Additionally, the use of low-loading transition metal catalysts means that expensive metal costs are amortized over a large amount of product. The generation of water as the sole by-product eliminates the need for expensive waste treatment facilities required for acidic or halogenated waste streams, resulting in substantial cost savings in environmental compliance and disposal. These factors combine to create a significantly more cost-effective manufacturing route compared to traditional SNAr methodologies.
- Enhanced Supply Chain Reliability: Reliability is paramount in the pharmaceutical supply chain, and this method enhances it by decoupling production from the availability of niche halogenating agents. Since the alkylating agents are common alcohols, lead times for raw material procurement are significantly shortened. The robustness of the reaction conditions, which tolerate air and moisture better than many sensitive organometallic processes, further reduces the risk of batch failures due to environmental fluctuations. This stability ensures a consistent output of high-purity intermediates, allowing downstream API manufacturers to plan their production schedules with greater confidence. The ability to source materials globally without specialized hazardous transport requirements adds another layer of resilience to the supply chain.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this technology is inherently designed for scalability. The absence of exothermic hazards associated with chlorinating agents makes the scale-up from gram to ton scale much safer and more predictable. The green chemistry metrics of the process align perfectly with modern corporate sustainability goals and increasingly stringent environmental regulations. By minimizing waste generation and avoiding persistent organic pollutants, manufacturers can achieve a lower carbon footprint and easier permitting for new production lines. This environmental compatibility not only future-proofs the manufacturing asset but also enhances the brand value of the final pharmaceutical products by associating them with sustainable production practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this N-alkylation technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this method into their existing portfolios. The versatility of the catalyst system and the breadth of the substrate scope are key takeaways that demonstrate the maturity of this synthetic approach.
Q: What are the advantages of this N-alkylation method over traditional SNAr reactions?
A: Unlike traditional SNAr reactions that require toxic halogenating agents like phosphorus oxychloride and produce hazardous waste, this method uses non-toxic alcohols as alkylating agents and generates only water as a by-product, offering superior atom economy and environmental safety.
Q: Does this process suffer from over-alkylation or regioselectivity issues?
A: No, the reaction exhibits complete regioselectivity and chemoselectivity. It specifically targets the exocyclic amino group without alkylating the nitrogen atoms within the aromatic ring or forming unwanted 2-(N,N-dialkyl)aminopyrimidine by-products.
Q: Which catalysts are suitable for this transformation?
A: The patent specifies several effective transition metal complexes, including Iridium ([Cp*IrCl2]2, [IrCl(cod)]2), Rhodium ([Cp*RhCl2]2), and Ruthenium ([Ru(p-cymene)Cl2]2) catalysts, allowing flexibility based on availability and cost considerations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(N-Alkyl)aminopyrimidine Supplier
The transition to greener, more efficient synthetic routes requires a partner with deep technical expertise and proven scale-up capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, offering comprehensive CDMO services tailored to the production of complex heterocyclic intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from benchtop discovery to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest industry standards. Our commitment to quality and consistency makes us the preferred choice for global pharmaceutical companies seeking reliable supply partners.
We invite you to explore how this advanced N-alkylation technology can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis specific to your target molecules. We are ready to provide specific COA data and detailed route feasibility assessments to support your R&D and sourcing strategies. Let us collaborate to bring your next-generation therapeutics to market faster and more sustainably.
