Advanced Synthesis of Chiral BIDIME Selenium Sulfur Ligands for Commercial Scale-Up
The landscape of asymmetric catalysis is continuously evolving, driven by the demand for more robust and versatile ligand systems capable of handling complex molecular architectures. A significant breakthrough in this domain is detailed in patent CN115490730A, which discloses a novel class of methods for synthesizing selenium and sulfur-based ligands derived from chiral BIDIME (benzoxaphosphine bisaryl monophosphine) scaffolds. This technology addresses critical limitations in traditional ligand design by introducing a rigid backbone that restricts phosphino group rotation, thereby establishing a defined atomic orientation for metal coordination. Unlike earlier generations of ligands such as SPhos or XPhos, which rely on flexible biaryl bonds, the BIDIME framework offers superior stereocontrol. The patent highlights a streamlined oxidative protocol where chiral BIDIME derivatives react directly with elemental selenium or sulfur powders. This approach not only simplifies the synthetic pathway but also ensures high catalytic efficiency and broad substrate tolerance, making it a highly attractive candidate for the production of high-purity pharmaceutical intermediates.
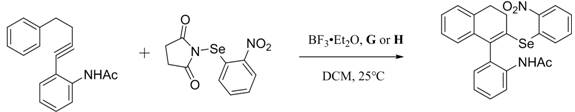
Furthermore, the utility of these ligands extends beyond simple coupling reactions. As illustrated in the provided reaction schemes, these chiral catalysts facilitate the construction of axially chiral selenium-containing bisaryl derivatives through reactions involving benzyne precursors. The ability to access such structurally complex motifs with high enantioselectivity is paramount for modern drug discovery, particularly in the development of bioactive molecules where axial chirality plays a decisive role in biological activity. For R&D directors seeking reliable solutions for challenging transformations, this technology represents a substantial leap forward, offering a versatile toolkit that combines the stability of phosphine oxides with the unique electronic properties of selenium and sulfur donors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of diaryl monophosphine ligands with rigid backbones has been a challenging endeavor in organometallic chemistry. Early reports, such as those by Buchwald and colleagues, focused heavily on ligands with flexible biaryl bonds, which, while effective for many Suzuki-Miyaura couplings, often suffer from conformational freedom that can dilute stereochemical induction in asymmetric catalysis. Attempts to introduce rigidity were often limited to specific substituents like dicyclohexyl or di-tert-butyl groups, restricting the tunability of the electronic and steric environment around the phosphorus center. Moreover, traditional purification methods for sensitive phosphine ligands often involve laborious chromatographic separations that can lead to product degradation or loss of optical purity. The reliance on transition metal catalysts that require extensive removal steps further complicates the manufacturing process, increasing both the cost and the environmental footprint of producing these valuable fine chemicals.
The Novel Approach
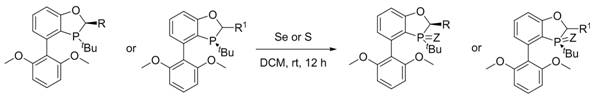
The methodology presented in patent CN115490730A fundamentally shifts the paradigm by utilizing a direct oxidation strategy on pre-formed chiral BIDIME skeletons. Instead of constructing the chiral environment from scratch for each variation, this approach leverages the inherent rigidity of the benzoxaphosphine backbone and simply modifies the phosphorus center through oxidation with elemental chalcogens. This results in a library of ligands (designated as Formula A through H) that maintain the crucial rigid geometry while offering diverse electronic properties via the P=Se or P=S bonds. The process is remarkably efficient, operating under mild conditions—typically room temperature in dichloromethane—and avoids the need for harsh reagents or extreme temperatures. By integrating a novel purification step using chemically modified diatomite, the method ensures that the final ligands possess exceptional purity levels, which is critical for their performance in sensitive catalytic cycles. This simplicity and robustness make the novel approach ideal for cost reduction in fine chemical manufacturing.
Mechanistic Insights into Chalcogen Oxidation and Purification
The core chemical transformation involves the nucleophilic attack of the trivalent phosphorus atom in the BIDIME derivative on the elemental selenium or sulfur lattice. Under anhydrous and oxygen-free conditions, the lone pair on the phosphorus attacks the chalcogen, forming a stable pentavalent phosphorus-chalcogen double bond (P=Z, where Z is Se or S). This oxidation step is thermodynamically favorable and proceeds cleanly without generating significant byproducts, which is a key factor in achieving the reported high yields of ≥90%. The rigidity of the BIDIME scaffold prevents the ligand from adopting non-productive conformations during this process, ensuring that the stereochemical information encoded in the backbone is preserved throughout the reaction. This mechanistic clarity allows for precise control over the final product quality, minimizing the formation of diastereomers or oxidized impurities that could poison downstream catalytic reactions.
A distinguishing feature of this technology is the innovative purification protocol employing modified diatomite. Standard diatomite often contains surface hydroxyl groups that can interact unpredictably with polar organophosphorus compounds, leading to tailing or incomplete recovery. In this process, the diatomite is chemically modified using dimethylcarbamoyl chloride and p-methoxycinnamoyl chloride. This modification reduces the surface hydroxyl density and introduces aromatic moieties that improve the wettability and dispersion of the filter aid in organic solvents like dichloromethane. The result is a filtration medium that effectively adsorbs polar impurities and unreacted starting materials while allowing the target ligand to pass through with minimal retention. This tailored purification step is instrumental in achieving the high purity specifications required for commercial-grade catalysts, effectively replacing more expensive and time-consuming recrystallization or preparative HPLC processes.
How to Synthesize Chiral BIDIME Ligands Efficiently
The synthesis of these advanced ligands is designed for operational simplicity, making it accessible for both laboratory-scale optimization and industrial production. The process begins with the dissolution of the chiral BIDIME precursor in ultra-dry dichloromethane, ensuring that no moisture interferes with the sensitive phosphorus center. Following this, a stoichiometric excess of elemental selenium or sulfur powder is introduced to drive the oxidation to completion. The reaction mixture is then stirred at ambient temperature for a period ranging from 10 to 15 hours, allowing sufficient time for the solid chalcogen to react fully with the dissolved ligand. Monitoring via TLC confirms the consumption of the starting material, after which the mixture is subjected to the specialized filtration process. The detailed standardized synthesis steps for specific analogues, such as Compound A, are outlined in the guide below.
- Dissolve the chiral BIDIME derivative in ultra-dry dichloromethane under anhydrous and oxygen-free conditions.
- Add elemental selenium powder or sulfur powder at room temperature and stir the reaction mixture for 10 to 15 hours.
- Filter the reaction mixture using modified diatomite, concentrate the filtrate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method offers tangible strategic benefits that extend beyond mere technical performance. The primary advantage lies in the drastic simplification of the manufacturing workflow. By eliminating the need for complex multi-step syntheses or cryogenic conditions, the process significantly reduces energy consumption and equipment wear. The use of inexpensive, commodity-grade reagents like elemental sulfur and selenium, combined with a solvent system that is easily recovered, contributes to substantial cost savings in raw material procurement. Furthermore, the high reaction yields minimize waste generation, aligning with increasingly stringent environmental regulations and reducing the costs associated with waste disposal. This efficiency translates directly into a more competitive pricing structure for the final ligand products.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in the ligand synthesis itself, coupled with the use of ambient temperature conditions, drastically lowers the operational expenditure. The modified diatomite purification replaces costly chromatographic resins or multiple recrystallization steps, streamlining the downstream processing. This lean manufacturing approach ensures that the cost of goods sold (COGS) is minimized, allowing for better margin management in the supply of high-value pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable starting materials such as chiral BIDIME derivatives and elemental powders mitigates the risk of supply disruptions often associated with sensitive organometallic reagents. The robustness of the reaction conditions means that production can be scaled up without requiring specialized infrastructure, ensuring consistent availability of these critical catalysts. This reliability is crucial for maintaining continuous production schedules in the pharmaceutical sector, where delays in intermediate supply can have cascading effects on drug development timelines.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-kilogram commercial batches. The use of dichloromethane, a common industrial solvent, facilitates easy solvent recovery and recycling. Additionally, the solid waste generated (spent diatomite and unreacted chalcogens) is manageable and can be treated using standard hazardous waste protocols. This scalability ensures that the technology can meet the growing demand for chiral ligands without compromising on environmental safety or regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What is the primary advantage of using modified diatomite in this synthesis?
A: The modified diatomite, treated with dimethylcarbamoyl chloride and p-methoxycinnamoyl chloride, significantly enhances purification efficiency by reducing surface hydroxyl groups and improving dispersion in organic solvents, leading to higher product purity compared to standard filtration methods.
Q: What yields can be expected from this oxidation process?
A: The patent data indicates that the synthesis of chiral BIDIME selenium/sulfur ligands consistently achieves high reaction yields, typically exceeding 90%, with optimized examples reaching up to 96%.
Q: Can these ligands be used for axial chiral compound synthesis?
A: Yes, these ligands serve as effective catalysts for the enantioselective cross-coupling of benzyne derivatives and selenium aryl reagents, demonstrating broad catalytic universality and high efficiency in forming axial chiral bisaryl derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral BIDIME Ligands Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that high-performance ligands play in accelerating drug discovery and process development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop innovation to industrial reality is seamless. We are committed to delivering these chiral BIDIME selenium and sulfur ligands with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our capability to handle complex organophosphorus chemistry positions us as a strategic partner for organizations seeking to secure their supply of advanced catalytic materials.
We invite you to engage with our technical procurement team to discuss how this technology can be integrated into your specific workflow. Whether you require a Customized Cost-Saving Analysis for your current ligand sourcing or need specific COA data and route feasibility assessments for new projects, we are ready to provide the support you need. Contact us today to request samples or initiate a dialogue about optimizing your catalytic processes with our premium range of chiral intermediates.
