Advanced Synthesis of 1,2,4-Oxadiazole Bipyridine Benzamides for High-Efficiency Fungicides
The global agrochemical sector is constantly driven by the demand for novel modes of action to combat resistant fungal pathogens, necessitating the development of sophisticated heterocyclic scaffolds. Patent CN111592533A introduces a significant advancement in this domain by detailing the preparation and application of 1,2,4-oxadiazole bipyridine substituted benzamide compounds. These molecules represent a complex fusion of nitrogen-rich heterocycles designed to exhibit potent fungicidal activity against critical agricultural threats such as Sclerotinia sclerotiorum and Botrytis cinerea. The structural versatility of this scaffold allows for extensive derivatization, making it a highly valuable template for next-generation crop protection agents. As a leading entity in fine chemical manufacturing, understanding the nuances of this synthetic pathway is crucial for securing a reliable agrochemical intermediate supplier partnership that can deliver high-purity materials at scale.
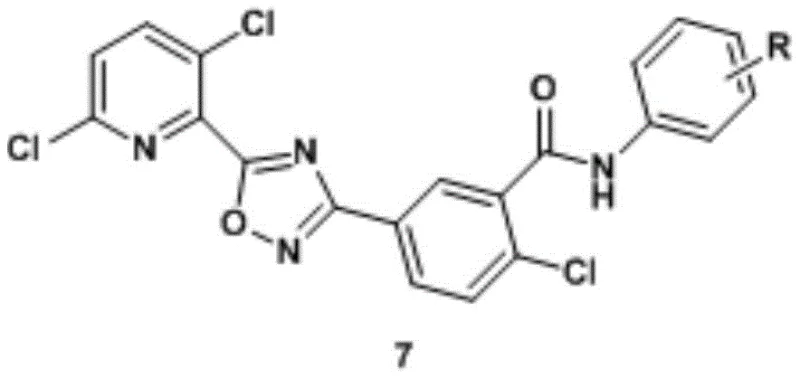
The core innovation lies in the strategic assembly of the 1,2,4-oxadiazole ring linked to a dichloropyridine moiety and a substituted benzamide. This specific architecture, as depicted in the general formula, combines the biological efficacy of oxadiazoles with the metabolic stability of pyridine rings. For R&D directors evaluating new leads, the ability to systematically vary the 'R' group on the terminal phenyl ring offers a powerful structure-activity relationship (SAR) exploration tool. The patent confirms that substituents ranging from simple halogens to bulky tert-butyl groups can be accommodated, providing a broad chemical space for optimization without compromising the integrity of the central pharmacophore.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional routes to similar benzamide-oxadiazole hybrids often suffer from inefficient ring-closing steps that require harsh acidic conditions or prolonged heating, leading to significant degradation of sensitive functional groups. Conventional cyanation strategies frequently rely on stoichiometric amounts of toxic heavy metals or expensive palladium catalysts, which not only inflate the raw material costs but also create substantial downstream purification burdens to meet stringent residual metal specifications. Furthermore, older methodologies for constructing the oxadiazole core often involve the dehydration of amidoximes under forcing conditions that result in poor atom economy and difficult-to-remove polymeric byproducts. These inefficiencies translate directly into higher manufacturing costs and extended lead times, creating bottlenecks for procurement managers seeking cost reduction in agrochemical intermediate manufacturing.
The Novel Approach
The methodology outlined in CN111592533A overcomes these hurdles through a meticulously optimized five-step sequence that prioritizes yield and operational simplicity. A standout feature is the use of L-proline as a ligand in the copper-catalyzed cyanation step, which dramatically improves reaction efficiency compared to ligand-free systems. This approach allows the transformation to proceed in DMF at controlled temperatures, minimizing side reactions. Additionally, the final coupling step utilizes EDC·HCl, a carbodiimide reagent known for its mildness and effectiveness in forming amide bonds under ambient or slightly cooled conditions. This shift towards milder reagents ensures that the complex heterocyclic core remains intact, resulting in superior product quality and significantly simplified workup procedures that enhance overall throughput.
Mechanistic Insights into Copper-Catalyzed Cyanation and Cyclization
The heart of this synthetic strategy is the conversion of the aryl iodide to the corresponding nitrile, a transformation that sets the stage for the entire molecular architecture. In this specific protocol, cuprous cyanide acts as the cyanide source, but its reactivity is unlocked by the coordination of L-proline. This amino acid ligand likely forms a soluble copper complex that facilitates the oxidative addition into the carbon-iodine bond, followed by transmetallation and reductive elimination to install the cyano group. This mechanistic pathway is far more robust than traditional nucleophilic aromatic substitutions, which would require extremely electron-deficient substrates. The ability to functionalize the 5-position of the benzoate ring with high fidelity is critical, as any failure here would halt the entire synthesis.
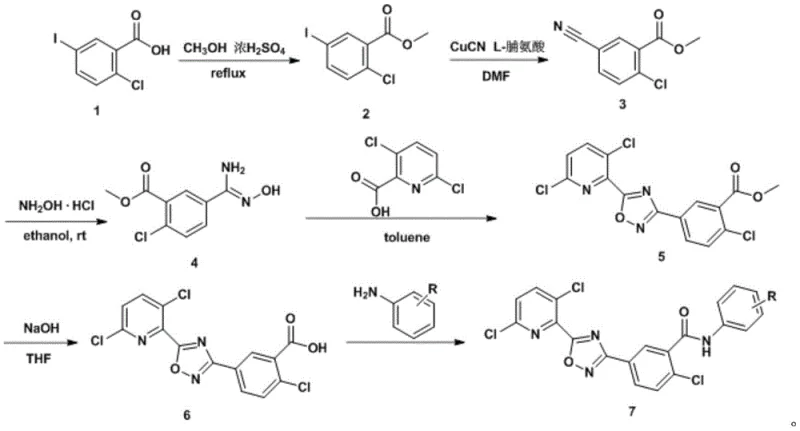
Following the installation of the nitrile, the formation of the 1,2,4-oxadiazole ring proceeds via a condensation with hydroxylamine to form an amidoxime intermediate, which is then acylated by 3,6-dichloropicolinic acid (activated as the acid chloride). The subsequent thermal cyclization involves the nucleophilic attack of the amidoxime oxygen onto the activated carbonyl, followed by dehydration. This cascade is elegantly managed in toluene, a solvent that allows for the removal of water via azeotropic distillation, driving the equilibrium towards the desired heterocycle. Understanding these mechanistic details is vital for process chemists aiming to replicate this commercial scale-up of complex agrochemical intermediates, as slight deviations in temperature or stoichiometry during the cyclization phase can lead to open-chain impurities that are difficult to purge.
How to Synthesize 1,2,4-Oxadiazole Benzamides Efficiently
The synthesis described in the patent provides a robust framework for producing these high-value intermediates, balancing reaction kinetics with practical handling requirements. The process begins with a straightforward Fischer esterification, followed by the critical cyanation and ring-closing steps discussed previously. The final stages involve hydrolysis of the methyl ester to the free acid and subsequent coupling with various anilines. For technical teams looking to implement this route, attention to detail in the purification of the intermediate nitrile and the control of pH during the final workup are paramount to achieving the reported high purities. The detailed standardized synthesis steps see the guide below for a breakdown of the specific operational parameters.
- Esterification of 5-iodo-2-chlorobenzoic acid with methanol using concentrated sulfuric acid catalyst under reflux to form methyl 5-iodo-2-chlorobenzoate.
- Copper-catalyzed cyanation using cuprous cyanide and L-proline in DMF at elevated temperatures (70-100°C) to generate the nitrile intermediate.
- Reaction with hydroxylamine hydrochloride followed by cyclization with 3,6-dichloropicolinic acid to construct the 1,2,4-oxadiazole core.
- Final hydrolysis and EDC-mediated amide coupling with substituted anilines to yield the target benzamide compounds.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers distinct advantages over legacy methods, primarily driven by the high efficiency of the individual transformations. The reported yields for key steps, such as the initial esterification (92.1%) and the final hydrolysis (93.6%), indicate a process that minimizes material loss and maximizes output per batch. For procurement managers, this efficiency translates into a more predictable supply of raw materials and reduced waste disposal costs, contributing to substantial cost savings without the need for speculative financial projections. The use of commodity chemicals like methanol, DMF, and toluene further insulates the production cost from the volatility associated with exotic reagents.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts in favor of copper and organic ligands drastically reduces the bill of materials. Moreover, the high selectivity of the L-proline mediated cyanation means that less starting material is wasted on side products, effectively lowering the cost per kilogram of the active intermediate. The avoidance of chromatographic purifications in favor of crystallization and filtration, as suggested by the isolation of solid products in the examples, is a critical factor in reducing processing time and solvent consumption, leading to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as 5-iodo-2-chlorobenzoic acid and substituted anilines ensures that the supply chain is not vulnerable to single-source bottlenecks. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require ultra-low temperature cryogenics, makes the process adaptable to various manufacturing sites globally. This flexibility is essential for reducing lead time for high-purity agrochemical intermediates, ensuring that production schedules can be met even during periods of high market demand or logistical disruption.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by maximizing atom economy in the cyclization steps and utilizing recyclable solvents like toluene and ethyl acetate. The absence of heavy metal contaminants in the final product simplifies the regulatory approval process for new pesticide formulations, as residual metal testing is less likely to be a failure point. Scalability is further supported by the exothermic profiles of the reactions, which are manageable in standard stainless steel reactors, allowing for a seamless transition from pilot plant to multi-ton commercial production without the need for specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these 1,2,4-oxadiazole derivatives. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity for partners evaluating this technology for their own pipelines. Understanding these specifics helps in aligning expectations regarding quality standards and production capabilities.
Q: What is the key catalytic advantage in the cyanation step of this process?
A: The process utilizes L-proline as a ligand for the cuprous cyanide catalyst. This organic amino acid ligand enhances the solubility and reactivity of the copper species in DMF, allowing for efficient substitution of the iodide group at moderate temperatures (70-100°C) without requiring expensive palladium catalysts.
Q: How does this synthesis route improve impurity control compared to traditional methods?
A: By employing a stepwise approach where the oxadiazole ring is formed prior to the final amide coupling, side reactions associated with direct amidation of unstable intermediates are minimized. The use of EDC·HCl as a mild coupling agent further ensures high purity by avoiding harsh activation conditions that often lead to racemization or decomposition.
Q: What represents the primary scalability bottleneck in this manufacturing process?
A: The management of exothermic reactions during the large-scale addition of thionyl chloride for acid activation and the subsequent handling of copper residues from the cyanation step require careful engineering controls. However, the high yields reported (over 90% in esterification and hydrolysis steps) suggest a robust process suitable for metric-ton scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Oxadiazole Benzamide Supplier
The technological potential of the 1,2,4-oxadiazole bipyridine scaffold is immense, offering a pathway to next-generation fungicides with improved resistance profiles. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready product is seamless. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of intermediate meets the exacting standards required for modern agrochemical formulation. We understand that consistency is key in this industry, and our process engineering team is dedicated to maintaining the highest levels of quality control throughout the manufacturing lifecycle.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to determine how our manufacturing capabilities can best support your R&D and commercial goals. Let us be your strategic partner in bringing high-performance crop protection solutions to the global market.
