Revolutionizing Arylacrylonitrile Production: A Green, Solvent-Free Catalytic Approach for Industrial Scale-Up
The global demand for high-purity arylacrylonitrile compounds, which serve as critical building blocks in the synthesis of pharmaceuticals, agrochemicals, and functional materials, continues to escalate. However, traditional manufacturing routes often struggle with significant environmental burdens and inefficient atom economy. Patent CN108164435B introduces a transformative methodology that addresses these challenges through a green, efficient synthesis protocol. This innovation utilizes a heterogeneous Ruthenium on Carbon (Ru/C) catalyst to facilitate the oxidative coupling of arylacetonitriles with methyl cyanoacetate under solvent-free conditions. By leveraging air as the terminal oxidant and operating at elevated temperatures between 150°C and 160°C, this process eliminates the need for toxic organic solvents and stoichiometric bases. For R&D directors and procurement managers seeking a reliable arylacrylonitrile supplier, this technology represents a paradigm shift towards sustainable manufacturing, offering a robust pathway to reduce production costs while maintaining stringent quality standards required for high-purity pharmaceutical intermediates.
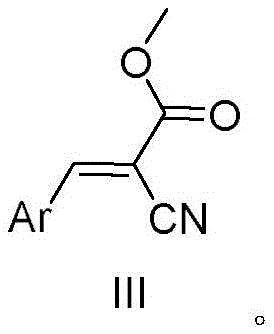
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of arylacrylonitrile derivatives has relied heavily on the Knoevenagel condensation reaction between aromatic aldehydes and cyanoacetates. While chemically straightforward, this classical approach suffers from severe drawbacks that hinder its viability in modern green manufacturing environments. The reaction necessitates the use of stoichiometric or excess amounts of basic catalysts, such as piperidine or ammonium acetate, to drive the dehydration step. Consequently, the post-reaction workup generates substantial volumes of alkaline wastewater, posing significant disposal challenges and increasing the environmental footprint of the facility. Furthermore, the separation of the product from the basic medium often requires extensive neutralization and washing steps, which not only consumes additional resources but also risks hydrolyzing sensitive functional groups on the molecule. These inefficiencies translate directly into higher operational expenditures and complicate the regulatory compliance landscape for manufacturers aiming to meet increasingly strict environmental discharge standards.
The Novel Approach
In stark contrast, the methodology disclosed in the patent data offers a streamlined, base-free alternative that fundamentally redesigns the synthetic logic. Instead of condensing an aldehyde, this novel route employs a direct oxidative coupling between an arylacetonitrile and methyl cyanoacetate. The key enabler is the Ru/C catalyst, which activates the C-H bonds of the substrates in the presence of molecular oxygen from the air. This solvent-free protocol not only simplifies the reaction setup by removing the need for large volumes of organic media but also drastically reduces the E-factor of the process. The absence of base means zero alkaline wastewater generation, solving a major pain point for environmental health and safety teams. Moreover, the use of a heterogeneous catalyst allows for easy separation via filtration, enabling the catalyst to be recycled and reused multiple times without significant loss of activity, thereby enhancing the overall economic feasibility of cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Ru/C-Catalyzed C-H Activation and Oxidative Coupling
From a mechanistic perspective, this transformation is driven by a sophisticated dual-cycle catalytic system involving C-H activation and oxidation. As illustrated in the patent's mechanistic diagrams, the Ru/C catalyst facilitates the activation of the benzylic C-H bond of the arylacetonitrile substrate. Simultaneously, it promotes the dehydrogenation of the methyl cyanoacetate. The reaction proceeds through an oxidative pathway where molecular oxygen serves as the hydrogen acceptor, generating water as the only byproduct of the oxidation step. This mechanism bypasses the formation of carbanion intermediates typical of base-catalyzed condensations, thereby avoiding side reactions associated with strong bases. The synergy between the oxidation cycle (Cycle A) and the coupling cycle (Cycle B) ensures high selectivity for the desired olefinic product. For technical teams evaluating process robustness, understanding this mechanism is crucial as it highlights the tolerance of the system towards various electronic environments on the aromatic ring, allowing for the synthesis of diverse derivatives without modifying the core reaction conditions.
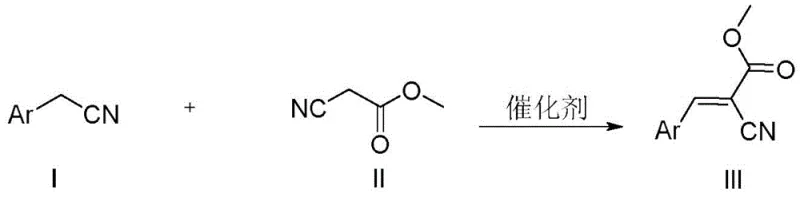
Impurity control is another critical aspect where this catalytic method excels. In traditional base-mediated reactions, self-condensation of the aldehyde or polymerization of the acrylonitrile product can lead to complex impurity profiles that are difficult to purge. The Ru/C catalyzed oxidative coupling, however, operates under neutral conditions, significantly suppressing these base-sensitive degradation pathways. The high selectivity of the ruthenium catalyst ensures that the reaction stops predominantly at the mono-coupled alkene stage, minimizing the formation of bis-alkylated or over-oxidized byproducts. This inherent selectivity simplifies the downstream purification process, often allowing for high-purity isolation through simple recrystallization rather than energy-intensive chromatography. For quality assurance professionals, this translates to a more consistent impurity profile and easier validation of the cleaning procedures, which is essential for maintaining the supply continuity of high-purity arylacrylonitriles.
How to Synthesize Arylacrylonitrile Efficiently
The practical implementation of this synthesis route is designed for scalability and ease of operation, making it highly attractive for commercial scale-up of complex pharmaceutical intermediates. The process begins with the charging of the arylacetonitrile substrate and a slight excess of methyl cyanoacetate into a reaction vessel equipped with heating and stirring capabilities. A commercially available Ru/C catalyst is added in a precise molar ratio relative to the substrate, typically ranging from 2‰ to 10‰. The reaction mixture is then heated to 150-160°C under an ambient air atmosphere, eliminating the need for inert gas purging or high-pressure oxygen equipment. Reaction progress is monitored via TLC until the starting material is fully consumed, usually within 3 to 6 hours. Upon completion, the mixture is cooled, and the solid catalyst is removed by filtration. The detailed standardized synthesis steps for optimizing yield and purity are provided in the guide below.
- Charge arylacetonitrile, Ru/C catalyst (2‰-10‰ molar ratio), and excess methyl cyanoacetate into a reaction vessel.
- Stir the mixture at 150-160°C under an air atmosphere until TLC indicates complete consumption of the arylacetonitrile starting material.
- Cool to room temperature, filter to recover the Ru/C catalyst, distill the filtrate to recover methyl cyanoacetate, and recrystallize the residue from ethanol to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Ru/C catalyzed technology offers compelling strategic advantages beyond mere technical feasibility. The shift from a homogeneous, base-heavy process to a heterogeneous, solvent-free system fundamentally alters the cost structure and risk profile of the supply chain. By eliminating the consumption of stoichiometric bases and the associated waste treatment costs, the overall variable cost of goods sold is significantly reduced. Furthermore, the ability to recover and recycle the expensive ruthenium catalyst and the excess methyl cyanoacetate reagent creates a closed-loop system that minimizes raw material wastage. This efficiency is critical for maintaining competitive pricing in a volatile raw material market, ensuring that the reliable arylacrylonitrile supplier can offer stable long-term contracts without frequent price adjustments due to waste disposal fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of costly waste treatment and the maximization of raw material utility. Since the reaction does not require organic solvents, the capital expenditure on solvent recovery units is minimized, and the energy load for distillation is drastically lowered. The heterogeneous nature of the Ru/C catalyst allows for simple filtration recovery, avoiding the complex extraction processes needed for homogeneous catalysts. Additionally, the patent data indicates that the excess methyl cyanoacetate can be distilled and reused in subsequent batches, effectively turning a potential waste stream into a valuable asset. These factors combine to create a lean manufacturing process that delivers substantial cost savings without compromising on the quality of the final active pharmaceutical ingredient intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and robustness of the reaction conditions. Operating under air atmosphere removes the dependency on specialized inert gases or high-pressure oxygen infrastructure, reducing the risk of supply interruptions due to utility failures. The broad substrate scope demonstrated in the patent examples means that the same production line can be easily adapted to manufacture various substituted arylacrylonitriles, providing flexibility to respond to changing market demands. Moreover, the use of commercially available Ru/C catalysts ensures that there are no bottlenecks related to proprietary reagent sourcing. This operational flexibility allows manufacturers to maintain high inventory turnover rates and reduce lead time for high-purity arylacrylonitriles, ensuring that downstream customers receive their materials on schedule.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial tonnage is facilitated by the absence of exothermic hazards often associated with base-catalyzed condensations. The solvent-free nature of the reaction reduces the reactor volume required per unit of product, effectively increasing the throughput of existing facilities without the need for major capital expansion. From an environmental compliance standpoint, the process aligns perfectly with green chemistry metrics by preventing pollution at the source rather than treating it post-generation. The lack of alkaline wastewater and organic solvent emissions simplifies the permitting process for new production lines and reduces the regulatory burden on existing plants. This sustainability profile is increasingly becoming a prerequisite for partnerships with top-tier multinational corporations that prioritize ESG (Environmental, Social, and Governance) criteria in their supplier selection.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing production portfolios.
Q: What are the primary environmental advantages of this Ru/C catalyzed method compared to traditional Knoevenagel condensation?
A: Unlike traditional methods that require stoichiometric amounts of base and generate significant alkaline wastewater, this novel process operates under solvent-free conditions using air as the oxidant. It eliminates alkaline waste entirely and allows for the recycling of both the heterogeneous catalyst and excess reagents, aligning with strict green chemistry principles.
Q: Can the Ru/C catalyst be reused effectively in industrial production?
A: Yes, the patent data demonstrates that the Ru/C catalyst is heterogeneous and can be easily recovered by simple filtration after the reaction. Experimental results confirm that the recovered catalyst maintains high activity over multiple cycles without significant loss in yield or purity, ensuring long-term cost efficiency.
Q: What is the substrate scope for this synthesis method?
A: The method exhibits broad substrate tolerance, successfully converting various arylacetonitriles including those with electron-donating groups (methyl, methoxy), electron-withdrawing groups (nitro, chloro), and heterocyclic systems (naphthyl, thienyl) into the corresponding arylacrylonitrile derivatives with high yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arylacrylonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is not just an environmental imperative but a strategic business advantage. Our team of expert chemists has extensively evaluated the Ru/C catalyzed synthesis of arylacrylonitriles and confirmed its potential for delivering high-quality intermediates at a competitive cost. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch meets the exacting standards required for pharmaceutical applications, providing our partners with the confidence they need to accelerate their drug development pipelines.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Whether you require custom synthesis of novel arylacrylonitrile derivatives or scale-up of existing routes, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and sustainability can drive value for your organization.
