Advanced Synthesis of 4-Bromo-1-oxypentafluorosulfanylbenzene for High-Performance Material Applications
The chemical industry is constantly seeking more efficient pathways to synthesize complex fluorinated intermediates, particularly those containing the pentafluorosulfanyl (SF5) group, which imparts unique lipophilicity and metabolic stability to bioactive molecules. Patent CN101104600A introduces a groundbreaking methodology for the preparation of 4-bromo-1-oxypentafluorosulfanylbenzene, a critical building block for advanced agrochemicals and liquid crystal materials. This innovation addresses the longstanding inefficiencies associated with introducing the SF5 moiety onto aromatic rings, shifting the synthetic strategy from harsh, non-selective direct fluorosulfanylation to a milder, highly selective electrophilic bromination of a pre-functionalized precursor. By leveraging specific catalytic systems, this process achieves exceptional yields and purity profiles that were previously unattainable with conventional techniques.
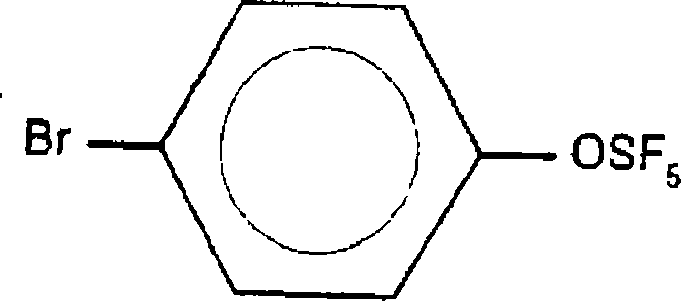
For R&D directors focusing on the development of next-generation active pharmaceutical ingredients (APIs) and electronic materials, the structural integrity and purity of intermediates like 4-bromo-1-oxypentafluorosulfanylbenzene are paramount. The SF5 group is increasingly recognized as a superior pharmacophore compared to the traditional trifluoromethyl group, offering greater steric bulk and electron-withdrawing capability. However, the synthesis of aryl-OSF5 compounds has historically been plagued by difficult reaction conditions and poor regiocontrol. The disclosed technology provides a robust solution, enabling the reliable production of high-purity pharmaceutical intermediates with a defined substitution pattern, thereby streamlining the downstream synthesis of complex drug candidates and optoelectronic monomers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the standard approach for synthesizing brominated pentafluorosulfanyl benzenes involved the direct reaction of bromobenzene with bis(pentafluorosulfanyl) peroxide (SF5OOSF5) or similar SF5 sources. As detailed in background references such as DE 100 58 472 A1, this legacy method necessitates extreme reaction conditions, typically requiring heating in a nickel-lined autoclave at temperatures around 150°C for extended periods exceeding 18 hours. These harsh thermal conditions not only pose significant safety risks due to the potential instability of peroxide species at high temperatures but also lead to extensive decomposition and side reactions. The resulting reaction mixtures are notoriously complex, containing a myriad of isomers, poly-brominated byproducts, and defluorinated impurities, making the isolation of the desired para-isomer a laborious and yield-limiting step involving difficult fractional distillations.
The Novel Approach
In stark contrast, the novel approach described in the patent reverses the synthetic logic by starting with pentafluorosulfanyloxybenzene and performing a controlled bromination. This strategic shift allows the reaction to proceed under remarkably mild conditions, often at temperatures as low as 0°C, utilizing standard laboratory glassware rather than specialized high-pressure equipment. By employing specific brominating agents like 1,3-dibromo-5,5-dimethylhydantoin (DBDMH) or N-bromosuccinimide (NBS) in the presence of a strong acid catalyst, the process achieves a dramatic improvement in efficiency. The mildness of the reaction preserves the sensitive SF5-O bond, preventing the degradation observed in thermal methods, and ensures that the bromine atom is introduced selectively at the para-position relative to the oxygen atom, drastically simplifying the purification workflow and boosting overall material throughput.
Mechanistic Insights into Triflic Acid-Catalyzed Electrophilic Bromination
The core of this technological advancement lies in the mechanistic pathway of the electrophilic aromatic substitution facilitated by trifluoromethanesulfonic acid (TfOH). In this system, the strong Brønsted acid acts as a potent activator for the brominating agent, generating a highly reactive electrophilic bromine species in situ. When this activated species encounters the pentafluorosulfanyloxybenzene substrate, the electron-rich aromatic ring undergoes attack. Although the -OSF5 group is electron-withdrawing due to the highly electronegative fluorine atoms, the oxygen atom possesses lone pairs that can donate electron density into the ring through resonance, rendering the ring sufficiently nucleophilic for substitution under these activated conditions. The steric bulk of the pentavalent sulfur group exerts a powerful directing effect, effectively blocking the ortho-positions and channeling the electrophilic attack almost exclusively to the para-position, which explains the high regioselectivity observed in the experimental data.
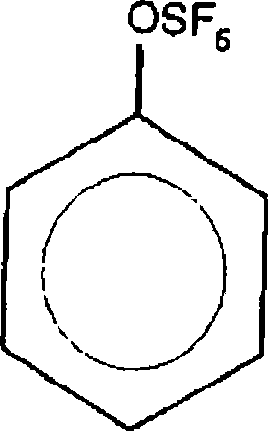
Furthermore, the choice of solvent and catalyst loading plays a critical role in controlling the impurity profile. The patent specifies the use of non-polar or moderately polar organic solvents such as dichloromethane, which solubilize both the organic substrate and the ionic intermediates effectively without participating in side reactions. The catalyst, trifluoromethanesulfonic acid, is used in stoichiometric or near-stoichiometric amounts relative to the brominating agent to ensure complete activation. This precise control over the reaction environment minimizes the formation of poly-brominated species and prevents the hydrolysis of the SF5 group, which is a common failure mode in less optimized acidic environments. For quality control teams, understanding this mechanism is vital for setting appropriate in-process controls, ensuring that the final product meets the stringent purity specifications required for high-value applications in the electronics and pharmaceutical sectors.
How to Synthesize 4-Bromo-1-oxypentafluorosulfanylbenzene Efficiently
The synthesis protocol outlined in the patent offers a streamlined pathway that is amenable to both laboratory scale optimization and industrial production. The process begins with the careful preparation of the reaction mixture under an inert atmosphere to exclude moisture, which could degrade the reagents. The specific order of addition—introducing the brominating agent first, followed by the substrate solution and then the catalyst—is crucial for maintaining control over the exotherm and reaction rate. Detailed standardized operating procedures for this synthesis, including exact molar ratios and workup parameters, are essential for reproducibility.
- Prepare a reaction vessel under nitrogen atmosphere and cool to 0°C, adding the brominating agent such as 1,3-dibromo-5,5-dimethylhydantoin.
- Add a solution of pentafluorosulfanyloxybenzene in dichloromethane followed by the catalyst trifluoromethanesulfonic acid.
- Monitor reaction progress via GC-MS, quench with saturated sodium bicarbonate, and purify the crude product using silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from the legacy thermal process to this catalytic bromination method represents a significant opportunity for cost reduction in electronic chemical manufacturing and pharmaceutical intermediate sourcing. The elimination of high-temperature autoclaves reduces capital expenditure requirements and lowers energy consumption substantially, as the reaction proceeds efficiently at near-ambient or cooled temperatures. Furthermore, the drastic improvement in yield—from the low efficiencies of the old method to over 90% in the new process—means that less raw material is wasted, directly lowering the cost of goods sold (COGS). The simplified purification process, which avoids complex fractional distillations of hazardous mixtures, also reduces processing time and solvent usage, contributing to a leaner and more sustainable supply chain.
- Cost Reduction in Manufacturing: The new method eliminates the need for expensive nickel-lined high-pressure reactors and the extensive energy input required to maintain 150°C for nearly a day. By shifting to a mild, catalytic process, manufacturers can utilize standard glass-lined or stainless steel reactors, significantly reducing depreciation costs and utility bills. Additionally, the high selectivity minimizes the generation of hard-to-separate isomers, reducing the volume of solvents and adsorbents needed for purification, which translates to substantial operational savings.
- Enhanced Supply Chain Reliability: Reliance on harsh thermal conditions often leads to unpredictable batch failures and equipment downtime due to corrosion or fouling. The milder nature of this bromination process enhances equipment longevity and batch-to-batch consistency, ensuring a more reliable supply of critical intermediates. The use of commercially available and stable reagents like NBS and DBDMH, rather than unstable peroxide precursors, further de-risks the supply chain by reducing hazards associated with storage and transport.
- Scalability and Environmental Compliance: Scaling up high-pressure peroxide reactions poses significant safety and regulatory hurdles. This new atmospheric pressure process is inherently safer and easier to scale from kilogram to multi-ton quantities without requiring specialized safety infrastructure. The reduction in hazardous waste generation, specifically the avoidance of complex fluorinated byproduct streams, simplifies waste treatment compliance and aligns with increasingly strict environmental regulations governing fluorine chemistry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on performance metrics and operational feasibility.
Q: What are the advantages of this bromination method over prior art?
A: Unlike prior art methods requiring high temperatures (150°C) and long reaction times (18+ hours) which result in complex mixtures, this patented process operates at mild conditions (0°C) with significantly higher regioselectivity for the para-isomer and yields exceeding 90%.
Q: Which brominating agents are most effective for this transformation?
A: The patent identifies 1,3-dibromo-5,5-dimethylhydantoin (DBDMH) and N-bromosuccinimide (NBS) as the most preferred reagents, particularly when used in conjunction with trifluoromethanesulfonic acid as a catalyst.
Q: Is this process suitable for large-scale manufacturing of electronic chemicals?
A: Yes, the elimination of harsh autoclave conditions and the use of standard organic solvents like dichloromethane make this route highly scalable for the production of liquid crystal media and other electronic chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Bromo-1-oxypentafluorosulfanylbenzene Supplier
As the global demand for fluorinated functional materials continues to surge, securing a supply partner with deep technical expertise in complex fluorine chemistry is essential. NINGBO INNO PHARMCHEM stands at the forefront of this sector, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 4-bromo-1-oxypentafluorosulfanylbenzene meets the exacting standards required for liquid crystal displays and advanced pharmaceutical research. We understand the critical nature of these intermediates in your value chain and are committed to delivering consistent quality.
We invite you to collaborate with us to optimize your sourcing strategy for this high-value intermediate. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized manufacturing processes can lower your total landed cost. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your next breakthrough in material science.
