Advanced One-Pot Protection Strategy for High-Purity Clarithromycin Manufacturing
The global demand for second-generation macrolide antibiotics continues to surge, driven by the need for acid-stable and bioavailable treatments for respiratory tract infections. At the forefront of this chemical landscape is Clarithromycin, a semi-synthetic derivative of Erythromycin A known for its superior pharmacokinetic profile. However, the manufacturing of this critical Active Pharmaceutical Ingredient (API) has historically been plagued by complex purification challenges and low selectivity during the key methylation step. Patent CN1098272C introduces a transformative synthetic methodology that addresses these bottlenecks through a novel protective group strategy. By employing a specific sequence of etherification and silylation, this process creates a highly reactive yet selective intermediate that streamlines the conversion of Erythromycin A 9-oxime into the final therapeutic agent. This technical breakthrough not only enhances the chemical purity of the output but also offers a robust framework for scalable manufacturing. The target molecule, Clarithromycin, is structurally defined by a 14-membered macrolide ring with specific methylation at the 6-position, a modification that is notoriously difficult to achieve without affecting other hydroxyl groups on the sugar moieties. 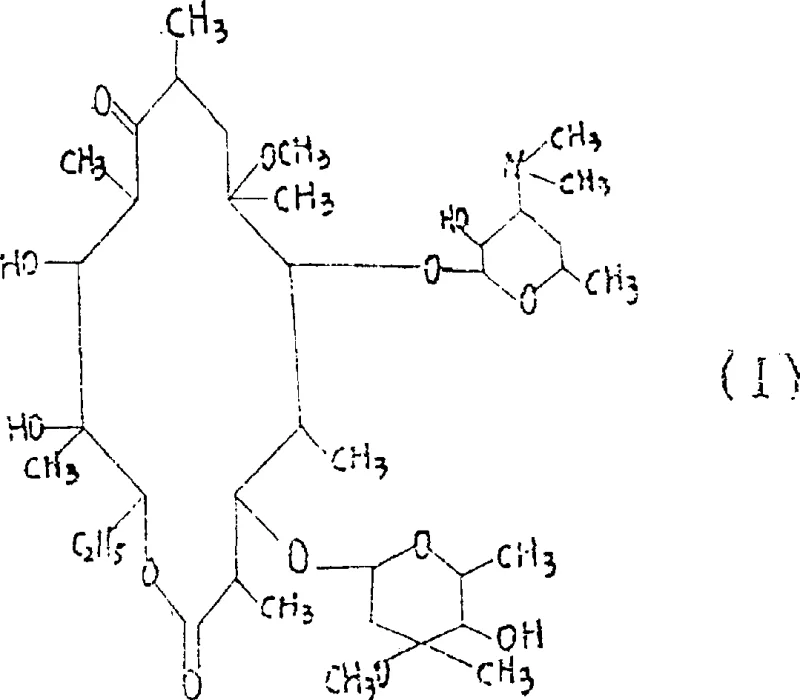
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Clarithromycin has relied on methodologies disclosed in earlier patents such as USP 4,311,803 and European Patent 158,467, which utilize dimethyl ketal protection at the 2' and 3' positions of the cladinose sugar. While effective to a degree, these traditional routes suffer from significant drawbacks that impact both yield and operational efficiency. The formation of dimethyl ketals often requires harsh acidic conditions and generates multiple by-products due to the presence of several hydroxyl groups on the erythromycin backbone. Consequently, the subsequent 6-O-methylation step frequently results in a mixture of regioisomers, necessitating complex and costly chromatographic separations or repeated recrystallizations to achieve pharmaceutical-grade purity. Furthermore, the removal of ketal protecting groups in the final stages often involves catalytic reduction or hydrolysis steps that can degrade the sensitive macrolide ring, leading to substantial material loss. These inefficiencies translate directly into higher production costs and extended lead times, creating a fragile supply chain for this essential antibiotic.
The Novel Approach
In stark contrast to the legacy ketal-based routes, the process outlined in CN1098272C employs a sophisticated dual-protection strategy involving etherification of the 9-oxime hydroxyl followed immediately by silylation of remaining hydroxyl groups. This approach generates a unique intermediate, represented generally by Formula (II), which acts as a precision template for the subsequent methylation. By utilizing reagents such as 2-methoxypropene for etherification and trimethylchlorosilane or hexamethyldisilazane for silylation, the method effectively masks all reactive sites except for the target 6-hydroxyl group. This steric shielding dramatically increases the regioselectivity of the methylation reaction, ensuring that the methyl group is installed exclusively at the desired position. The ability to perform these protection steps potentially in a telescoped manner without isolating the intermediate significantly reduces solvent usage and processing time. Moreover, the silyl protecting groups are designed to be removed under mild conditions that preserve the integrity of the macrolide structure, thereby maximizing overall yield and minimizing the formation of degradation impurities. 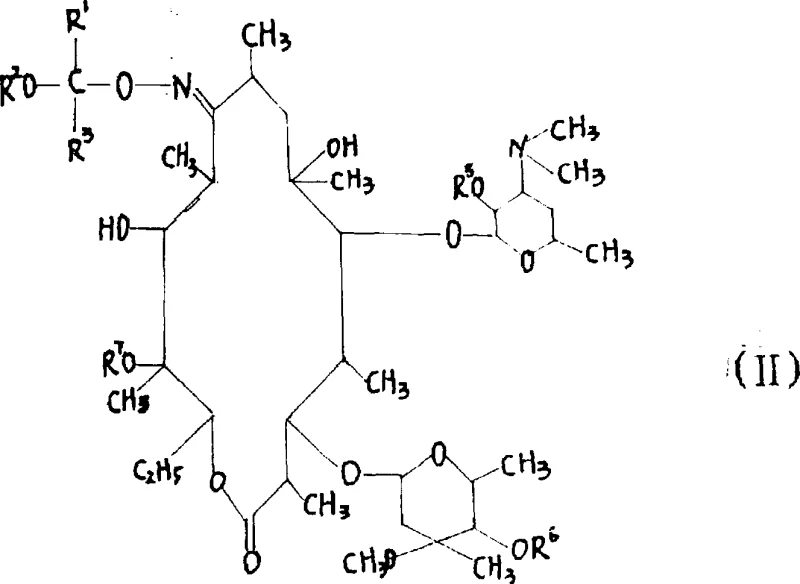
Mechanistic Insights into Selective 6-O-Methylation
The core innovation of this synthesis lies in the electronic and steric modulation of the Erythromycin A 9-oxime substrate through the formation of the silylated intermediate. In the conventional unprotected or ketal-protected states, the nucleophilicity of the various hydroxyl groups (2', 3', 4'', 6, 11, 12) is often similar, leading to competitive alkylation. However, the introduction of bulky trimethylsilyl (TMS) or related silyl groups at the 2', 4'', and potentially other positions creates a formidable steric barrier. This barrier physically blocks the approach of the methylating agent, such as methyl iodide or methyl bromide, to the protected hydroxyls. Simultaneously, the etherification of the 9-oxime prevents side reactions at the C-9 ketone, which is prone to enolization or rearrangement under basic conditions. The result is a substrate where the 6-hydroxyl group remains the most accessible and reactive nucleophile. When treated with a base like potassium hydroxide or sodium hydroxide in a polar aprotic solvent such as DMSO or DMF, the 6-oxygen anion is generated and rapidly attacks the methyl halide in an SN2 fashion. This precise control over reactivity eliminates the formation of 2'-O-methyl or 3'-O-methyl isomers, which are difficult to separate and constitute critical quality attributes (CQAs) in regulatory filings.
Furthermore, the stability of the silyl-ether intermediate contributes significantly to impurity control throughout the synthesis. Unlike acetals or ketals which can be sensitive to moisture and pH fluctuations, silyl ethers offer robust stability during the methylation step, preventing premature deprotection that could lead to scrambling of the product profile. The deprotection phase, typically achieved using aqueous acid or specific fluoride sources depending on the silyl group chosen, is clean and high-yielding. The patent data indicates that the crude product obtained after deprotection requires minimal purification, often just a simple recrystallization from ethanol or isopropanol with activated carbon treatment to remove colored impurities. This streamlined downstream processing is a direct consequence of the high fidelity of the upstream protection chemistry. By minimizing the generation of structural analogs and degradation products, the process ensures that the final API meets stringent pharmacopoeial standards for identity and purity without the need for resource-intensive preparative HPLC.
How to Synthesize Clarithromycin Efficiently
The synthesis protocol derived from this patent offers a practical roadmap for producing high-quality Clarithromycin intermediates. The process begins with the dissolution of Erythromycin A 9-oxime in a mixed solvent system, followed by the sequential addition of the etherifying agent and the silylating reagent in the presence of a catalyst like pyridine hydrochloride. This generates the fully protected intermediate in situ. Subsequent methylation is performed under controlled temperatures to ensure complete conversion while avoiding thermal degradation. The detailed standardized synthesis steps, including specific molar ratios, reaction times, and workup procedures, are provided in the guide below.
- React Erythromycin A 9-oxime with an etherifying agent (e.g., 2-methoxypropene) and a silylating reagent to form the protected intermediate.
- Perform selective 6-O-methylation on the protected intermediate using a methylating agent like methyl iodide in the presence of a base.
- Execute deprotection and purification steps, including acid hydrolysis and recrystallization, to isolate high-purity Clarithromycin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ether-silyl protection technology represents a significant opportunity to optimize the cost structure and reliability of Clarithromycin sourcing. Traditional manufacturing routes are often bottlenecked by low yields and the high cost of solvents and energy required for extensive purification. By implementing this novel pathway, manufacturers can achieve a drastic simplification of the production workflow. The elimination of complex separation steps for isomeric by-products translates directly into reduced operational expenditures (OPEX). Furthermore, the reagents utilized in this process, such as 2-methoxypropene and chlorotrimethylsilane, are commodity chemicals available from a wide range of global suppliers, mitigating the risk of raw material shortages. This diversification of the supply base ensures greater continuity of supply, a critical factor for maintaining inventory levels of essential antibiotics.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the substantial increase in overall process yield resulting from improved selectivity. In traditional methods, a significant portion of the starting material is lost to isomeric by-products that must be discarded or recycled at high cost. By directing the reaction almost exclusively to the 6-position, this new method maximizes the atom economy of the methylation step. Additionally, the potential to telescope the protection steps without isolating the intermediate reduces the volume of solvents required for washing and drying, leading to lower waste disposal costs and reduced energy consumption for solvent recovery. These cumulative efficiencies allow for a more competitive pricing structure for the final API without compromising on quality margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the chemical transformations involved. The use of stable silyl protecting groups reduces the sensitivity of the intermediate to environmental factors such as humidity, allowing for more flexible storage and transport conditions if intermediate stocking is required. Moreover, the simplified purification protocol, which relies on standard crystallization rather than specialized chromatography, means that production can be scaled up rapidly in response to market demand spikes. This scalability ensures that buyers can secure large volumes of material with consistent lead times, reducing the need for safety stock and freeing up working capital.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process aligns well with modern green chemistry principles. The reduction in solvent usage and the avoidance of heavy metal catalysts (often used in hydrogenation steps for ketal removal in older routes) simplify the effluent treatment process. The waste streams generated are less complex and easier to treat, facilitating compliance with increasingly stringent environmental regulations in major manufacturing hubs. This environmental compatibility not only reduces the risk of regulatory shutdowns but also enhances the sustainability profile of the supply chain, a growing priority for multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific advantages and operational parameters detailed in the patent literature, providing clarity on how this technology differentiates itself from incumbent methods. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or capacity expansion.
Q: How does the ether-silyl protection method improve selectivity compared to traditional ketal protection?
A: The ether-silyl method described in CN1098272C utilizes bulky silyl groups to sterically hinder unwanted reaction sites, significantly enhancing the selectivity of the 6-hydroxyl methylation compared to older dimethyl ketal methods which often result in complex isomer mixtures.
Q: What are the primary cost drivers reduced by this synthetic route?
A: This route reduces costs by minimizing the number of isolation steps through telescoped reactions and simplifying the purification process, thereby lowering solvent consumption and labor hours associated with separating difficult isomers.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is suitable for large-scale industrial production due to its high yield, operational simplicity, and the use of readily available reagents like 2-methoxypropene and trimethylchlorosilane.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clarithromycin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires more than just a patent; it demands deep process engineering expertise and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the ether-silyl protection strategy are fully realized in a GMP environment. We are committed to delivering Clarithromycin and its key intermediates with stringent purity specifications, leveraging our rigorous QC labs to monitor every critical process parameter and ensure the absence of genotoxic impurities or residual solvents.
We invite global partners to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and security in your antibiotic portfolio.
