Advanced Synthesis of Benzopyran-3-ol Derivatives via Palladium-Catalyzed Cross-Dehydrogenative Coupling for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds, particularly those found in bioactive molecules. Patent CN111423405A introduces a groundbreaking methodology for the synthesis of benzopyran-3-ol derivative compounds, a structural motif prevalent in potent therapeutic agents such as the antioxidant Epicatechin and the potassium channel opener Cromakalim. This innovation addresses critical bottlenecks in traditional organic synthesis by leveraging a palladium-catalyzed cross-dehydrogenative coupling (CDC) strategy. Unlike conventional routes that often suffer from poor regioselectivity or require harsh reaction conditions, this patented approach facilitates direct intramolecular C(sp3)-H arylation. The significance of this technology lies in its ability to streamline the production of high-purity pharmaceutical intermediates, offering a robust solution for manufacturers aiming to optimize their supply chains and reduce the environmental footprint of their operations. By enabling the direct formation of carbon-carbon bonds without pre-functionalization, this method represents a paradigm shift in how we approach the construction of chroman and benzopyran cores.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the C(sp3)-C(sp2) bond essential for benzopyran frameworks has relied heavily on classical methodologies such as the Friedel-Crafts reaction or transition metal-catalyzed cross-couplings like Suzuki-Miyaura and Negishi reactions. While these methods have served the industry well, they possess inherent limitations that hinder efficiency and scalability. The Friedel-Crafts reaction, for instance, is often plagued by poor regioselectivity, leading to complex mixtures of isomers that are difficult and costly to separate, thereby reducing the overall yield of the desired product. Furthermore, traditional cross-coupling reactions typically necessitate the use of pre-functionalized starting materials, such as aryl halides and organometallic reagents. The preparation of these precursors adds extra synthetic steps, increases the consumption of raw materials, and generates significant amounts of hazardous waste. Additionally, many of these conventional processes require stringent reaction conditions, including extremely low temperatures or the use of sensitive catalysts that are prone to deactivation, which complicates the operational protocol and increases the risk of batch failure in a commercial setting.
The Novel Approach
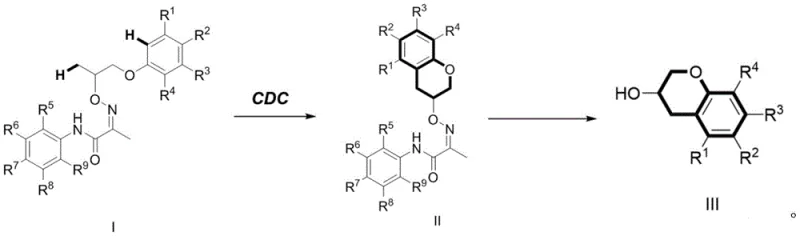
In stark contrast to these legacy methods, the technology disclosed in CN111423405A utilizes a direct cross-dehydrogenative coupling mechanism that bypasses the need for pre-functionalized substrates. This novel approach employs a palladium catalyst in conjunction with a specific oxidant to activate inert C-H bonds directly, facilitating an intramolecular cyclization that constructs the benzopyran ring system with high precision. The reaction conditions are remarkably mild, typically operating within a temperature range of 70°C to 130°C, which significantly reduces energy consumption compared to high-temperature alternatives. Moreover, the use of a removable directing group ensures exceptional regioselectivity, guiding the palladium catalyst to the specific C-H bond intended for functionalization. This results in a cleaner reaction profile with fewer by-products, simplifying the downstream purification process. The versatility of this method is further demonstrated by its tolerance to a wide variety of substituents on the aromatic rings, allowing for the synthesis of diverse derivatives from a common intermediate platform.
Mechanistic Insights into Pd-Catalyzed Intramolecular C(sp3)-H Arylation
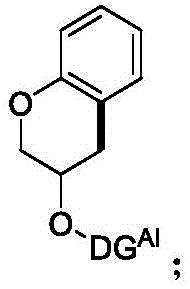
The core of this synthetic breakthrough lies in the sophisticated catalytic cycle driven by palladium. The process initiates with the coordination of the palladium species to the nitrogen-containing directing group present in Compound I. This coordination brings the metal center into close proximity with the target aliphatic C(sp3)-H bond, lowering the activation energy required for bond cleavage. In the presence of an oxidant, such as N-fluorobisbenzenesulfonamide (NFSI) or Selectfluor, the palladium center undergoes oxidation, facilitating the concerted metalation-deprotonation (CMD) or a similar C-H activation pathway. This step generates a key organopalladium intermediate where the metal is covalently bound to the carbon framework. Subsequent reductive elimination releases the cyclized product, Intermediate II, and regenerates the active palladium catalyst, completing the cycle. The choice of oxidant is critical, as it not only drives the catalytic turnover but also influences the stability of the high-valent palladium species involved in the C-H activation step.
Following the cyclization, the second critical phase involves the removal of the directing group to reveal the final benzopyran-3-ol structure. This is achieved through a distinct catalytic system utilizing molybdenum hexacarbonyl in a mixture of acetonitrile and water. The mechanism here likely involves the coordination of the carbonyl ligands to the directing group or the aromatic system, facilitating a hydrolytic or reductive cleavage that replaces the bulky directing moiety with a hydrogen atom. This step is crucial for obtaining the biologically active scaffold without residual synthetic handles. The structural integrity of Intermediate II, as confirmed by spectroscopic data, shows the successful formation of the chroman ring with the directing group still intact, serving as a testament to the high selectivity of the initial CDC step. The ability to install and subsequently remove this group with high fidelity is what allows this route to achieve superior purity profiles compared to non-directed methods.
How to Synthesize Benzopyran-3-ol Derivatives Efficiently
The practical implementation of this synthesis route is designed for reproducibility and ease of handling in a laboratory or pilot plant environment. The process begins with the precise mixing of the substrate Compound I with the palladium catalyst and oxidant in a suitable solvent like 1,2-dichloroethane. The reaction vessel is sealed and heated to the specified temperature, allowing the cyclization to proceed to completion over a period of 1 to 24 hours. Upon cooling, the reaction mixture undergoes a straightforward workup involving dilution with ethyl acetate, filtration to remove insoluble catalyst residues, and concentration under reduced pressure. The resulting crude Intermediate II is then purified via column chromatography. The second stage involves dissolving the purified intermediate in acetonitrile with water and adding molybdenum hexacarbonyl under an inert nitrogen atmosphere. Heating this mixture promotes the deprotection reaction, after which standard extraction and purification techniques yield the final high-purity benzopyran-3-ol derivative. For detailed standardized synthesis steps, please refer to the guide below.
- Mix Compound I with a palladium catalyst (e.g., Pd(OAc)2), an oxidant (e.g., N-fluorobisbenzenesulfonamide), and a solvent (e.g., 1,2-dichloroethane). React at 70-130°C for 1-24 hours to form Intermediate II via intramolecular cyclization.
- Perform post-treatment on the reaction mixture by dilution, filtration, and solvent removal. Purify the residue via column chromatography to isolate Intermediate II.
- React Intermediate II with molybdenum hexacarbonyl in acetonitrile and water at 85-95°C for 1-24 hours under nitrogen. Purify the final mixture to obtain the target Benzopyran-3-ol derivative (Product III).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the substantial reduction of raw material complexity. By eliminating the requirement for pre-halogenated starting materials, which are often more expensive and subject to greater supply chain volatility than their non-halogenated counterparts, manufacturers can secure a more stable and cost-effective supply of inputs. This simplification of the bill of materials directly translates to improved margin protection and reduced exposure to price fluctuations in the global chemical market. Furthermore, the streamlined nature of the process, which combines bond formation and ring closure in a single catalytic step, reduces the total number of unit operations required. Fewer processing steps mean lower labor costs, reduced equipment occupancy time, and diminished utility consumption, all of which contribute to a leaner and more competitive manufacturing cost structure.
- Cost Reduction in Manufacturing: The elimination of pre-functionalization steps removes the need for purchasing expensive halogenated precursors and the reagents required to synthesize them. This consolidation of synthetic steps drastically reduces the overall material throughput and waste generation, leading to significant operational expenditure savings. Additionally, the use of earth-abundant solvents and the potential for catalyst recycling further enhance the economic viability of the process, making it an attractive option for cost-sensitive large-scale production campaigns.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals for the starting materials ensures a robust supply chain that is less susceptible to disruptions. Unlike specialized intermediates that may have limited suppliers, the substrates for this CDC reaction are often accessible from multiple global sources. This diversification of supply options mitigates the risk of shortages and allows for more flexible sourcing strategies. Moreover, the mild reaction conditions reduce the demand for specialized high-pressure or cryogenic equipment, enabling production across a wider range of manufacturing facilities and increasing overall supply resilience.
- Scalability and Environmental Compliance: The process is inherently scalable due to its simple operational parameters and the absence of hazardous reagents typically associated with organometallic couplings. The reduced generation of halogenated waste streams aligns with increasingly stringent environmental regulations, lowering the costs associated with waste disposal and treatment. The high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, ensuring that the final product meets rigorous quality standards with minimal reprocessing. This efficiency supports the rapid scale-up from kilogram to multi-ton quantities, ensuring consistent supply continuity for downstream pharmaceutical applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. Understanding these aspects is crucial for R&D teams evaluating the feasibility of integrating this route into their existing pipelines and for procurement specialists assessing the long-term viability of the supply source.
Q: What are the advantages of this CDC method over traditional Suzuki coupling?
A: This method eliminates the need for pre-functionalized halogenated substrates, reducing raw material costs and waste generation while simplifying the synthetic route.
Q: What are the typical reaction conditions for the cyclization step?
A: The cyclization typically proceeds at moderate temperatures between 70°C and 130°C using a palladium catalyst and an oxidant in solvents like 1,2-dichloroethane.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes readily available reagents and standard workup procedures like filtration and column chromatography, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzopyran-3-ol Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the Pd-catalyzed CDC method in driving the next generation of pharmaceutical innovation. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of benzopyran-3-ol derivatives we deliver adheres to the highest international quality standards. We are committed to leveraging our technical expertise to optimize this specific route for your unique application needs, ensuring maximum yield and minimal impurity profiles.
We invite you to collaborate with us to unlock the full commercial potential of this synthesis method. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to specific COA data and comprehensive route feasibility assessments that will empower your decision-making process. Contact us today to discuss how we can support your supply chain with reliable, high-quality benzopyran-3-ol derivatives manufactured through this cutting-edge technology.
