Advanced Metal-Free Synthesis of (Iso)quinoline Substituted Flavones for Commercial API Production
Advanced Metal-Free Synthesis of (Iso)quinoline Substituted Flavones for Commercial API Production
The pharmaceutical and agrochemical industries are constantly seeking more efficient, sustainable, and cost-effective synthetic routes for complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN110790752A, which discloses a novel method for synthesizing (iso)quinoline and quinoxaline substituted flavone and quinolone derivatives. This technology represents a paradigm shift from traditional transition metal-catalyzed cross-coupling reactions to a streamlined, metal-free acid-catalyzed cyclization process. By utilizing readily available alkynone compounds and nitrogen oxide heterocycles as starting materials, this invention achieves high reaction efficiency under mild conditions without the need for expensive palladium or toxic tin reagents. For R&D directors and procurement managers alike, this development offers a compelling value proposition: a cleaner, safer, and potentially much more economical pathway to high-value bioactive intermediates that are crucial for drug discovery and development programs targeting bacterial growth and cancer.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of (iso)quinoline-substituted flavonoids or quinolones has relied heavily on classical cross-coupling methodologies, which present substantial drawbacks for large-scale manufacturing. As illustrated in the prior art, such as the work by Minna Bui et al. (2015), the Stille coupling reaction was employed to synthesize these targets using tin reagents and 3-iodo-substituted quinolinones.
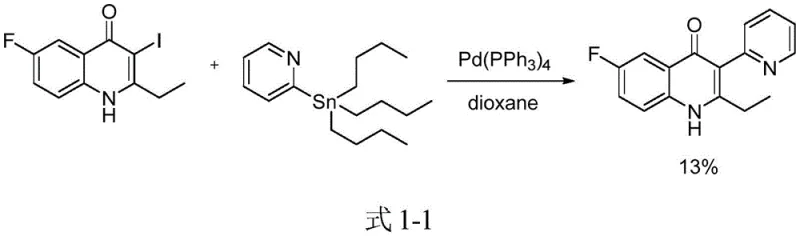
This conventional approach suffers from critically low yields and necessitates the use of highly toxic organotin compounds, posing severe environmental and safety hazards that complicate waste disposal and regulatory compliance. Furthermore, the reliance on palladium catalysts introduces significant cost volatility and the risk of heavy metal residues in the final API, requiring rigorous and expensive purification steps to meet stringent pharmacopeial limits. Other methods, such as those reported by Mohamed et al. involving Suzuki-Miyaura coupling, still depend on palladium participation, while alternative oxidative cyclizations using cerium ammonium nitrate generate substantial chemical waste, rendering them less attractive for green chemistry initiatives and cost-sensitive commercial production.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN110790752A introduces a direct, one-pot synthesis strategy that completely eliminates the need for transition metals. The core innovation involves reacting an alkynone compound with an (iso)quinoline or quinoxaline N-oxide in the presence of a simple acid catalyst, such as concentrated hydrochloric acid. This reaction proceeds smoothly in common organic solvents like DMF, DMSO, or acetonitrile at moderate temperatures ranging from 100°C to 140°C. The operational simplicity is remarkable; the process avoids the preparation of sensitive organometallic intermediates and bypasses the need for inert atmosphere conditions often required for palladium catalysis. By shifting the mechanistic paradigm to acid-promoted cyclization, this method not only simplifies the operational workflow but also drastically reduces the impurity profile associated with metal catalysts, thereby enhancing the overall quality and safety of the resulting pharmaceutical intermediates.
Mechanistic Insights into Acid-Catalyzed Cyclization
The mechanistic elegance of this transformation lies in the activation of the nitrogen oxide moiety by the acid catalyst, which facilitates a nucleophilic attack or cycloaddition with the electron-deficient alkynone system. Unlike transition metal cycles that involve oxidative addition and reductive elimination steps prone to catalyst deactivation, this acid-catalyzed pathway relies on protonation to enhance the electrophilicity of the reactants, driving the formation of the new carbon-carbon and carbon-heteroatom bonds required to close the flavone or quinolone ring system. The general reaction scheme demonstrates the versatility of this approach, accommodating a wide array of substituents on both the alkynone and the nitrogen heterocycle components.
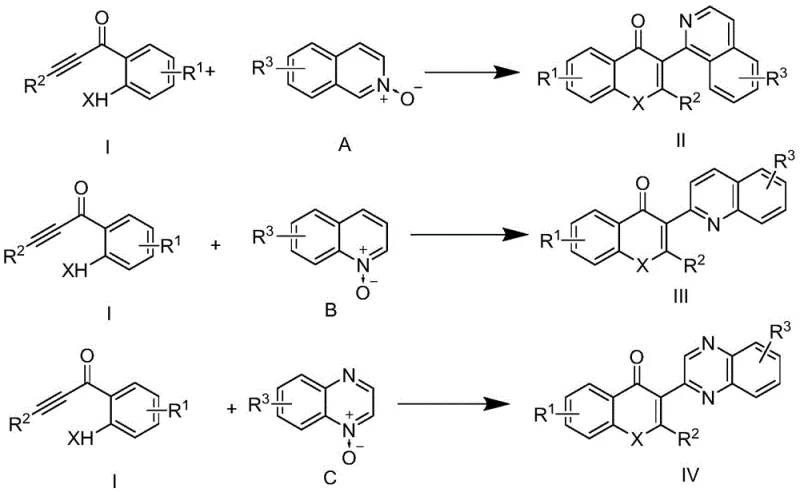
From an impurity control perspective, the absence of metal catalysts inherently removes a major class of difficult-to-remove contaminants, simplifying the downstream purification process significantly. The reaction tolerates diverse functional groups, including halogens, alkyl chains, alkoxy groups, and even heteroaryl moieties like thiophene, as evidenced by the broad substrate scope explored in the patent examples. This robustness suggests that the mechanism is not overly sensitive to steric hindrance or electronic variations, allowing for the synthesis of a diverse library of analogs without needing to re-optimize catalytic systems for each new substrate. For process chemists, this predictability is invaluable, as it reduces the risk of batch-to-batch variability and ensures consistent product quality essential for regulatory filings.
How to Synthesize (Iso)quinoline Substituted Flavones Efficiently
The synthesis protocol outlined in the patent provides a straightforward roadmap for producing these high-value derivatives. The process begins with the precise stoichiometric mixing of the alkynone precursor and the nitrogen oxide heterocycle in a polar aprotic solvent, followed by the careful addition of the acid catalyst. The reaction mixture is then heated to the specified temperature range, where monitoring via TLC ensures complete conversion before workup. While the specific experimental details regarding stoichiometry and purification are critical for reproducibility, the general procedure highlights the accessibility of this method for standard laboratory and pilot plant equipment.
- Mix alkynone compound, isoquinoline/quinoline N-oxide, and concentrated HCl in an organic solvent like DMF.
- Heat the reaction mixture to 100-140°C and stir for 2-6 hours until completion.
- Recover solvent via distillation and purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route translates into tangible strategic advantages that extend beyond mere technical feasibility. The elimination of precious metal catalysts like palladium and toxic reagents like tin directly addresses two of the most persistent pain points in fine chemical sourcing: cost volatility and environmental compliance. By removing these expensive and regulated inputs, manufacturers can achieve a more stable cost structure that is less susceptible to fluctuations in the global precious metals market. Furthermore, the simplified waste profile reduces the burden on environmental health and safety teams, lowering the overhead costs associated with hazardous waste disposal and regulatory reporting.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete removal of palladium catalysts and organotin reagents from the bill of materials. Palladium prices are notoriously volatile and can constitute a significant portion of the COGS for metal-catalyzed processes; eliminating this requirement results in substantial raw material savings. Additionally, the absence of heavy metals removes the need for specialized scavenging resins or complex purification protocols designed to lower metal residues to ppm levels, further streamlining the production cost. The use of inexpensive hydrochloric acid as the promoter instead of proprietary ligand-catalyst systems also contributes to a leaner, more cost-effective manufacturing process that enhances margin potential for high-volume commercial orders.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by relying on commodity chemicals rather than specialized catalytic systems. Alkynones and nitrogen oxides are generally more accessible and have more stable supply chains compared to sensitive transition metal complexes which may have limited suppliers or long lead times. The robustness of the reaction conditions—tolerating standard solvents and moderate heating—means that production is less likely to be disrupted by minor variations in utility quality or equipment performance. This reliability ensures consistent delivery schedules for downstream API manufacturers, reducing the risk of stockouts and enabling better inventory planning for long-term projects.
- Scalability and Environmental Compliance: The one-pot nature of this synthesis is inherently scalable, moving seamlessly from gram-scale laboratory optimization to multi-kilogram pilot runs and eventually to ton-scale commercial production. The lack of air- or moisture-sensitive reagents simplifies the engineering requirements for large reactors, avoiding the need for expensive inert gas blanketing or specialized handling equipment. From an environmental standpoint, the reduction in toxic heavy metal waste aligns perfectly with modern green chemistry principles and increasingly strict global environmental regulations. This compliance advantage facilitates faster regulatory approvals and minimizes the risk of production shutdowns due to environmental violations, securing the long-term viability of the supply source.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology, derived from the specific embodiments and comparative data provided in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this metal-free synthesis over traditional methods?
A: Unlike traditional Stille or Suzuki couplings that require expensive palladium catalysts and toxic tin reagents, this method uses simple acid catalysis (HCl), eliminating heavy metal contamination risks and significantly reducing raw material costs.
Q: What is the typical reaction temperature and time for this process?
A: The reaction typically proceeds efficiently at temperatures between 100°C and 140°C, with completion times ranging from 2 to 6 hours depending on the specific substrate substituents.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the one-pot nature, use of common solvents like DMF, and absence of sensitive transition metal catalysts make this process highly scalable and robust for industrial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (Iso)quinoline Substituted Flavones Supplier
The technological advancements described in CN110790752A underscore the immense potential of metal-free synthesis in modernizing the production of complex pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative academic and patent literature into robust, commercial-grade manufacturing processes. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this acid-catalyzed route are fully realized in practical, high-volume operations. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of verifying the absence of heavy metal contaminants, guaranteeing that every batch meets the exacting standards required by global regulatory bodies.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this cost-effective and environmentally friendly synthesis platform. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific project needs, comparing this metal-free route against your current supply chain metrics. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can accelerate your development timelines and optimize your overall manufacturing economics.
