Advanced Rhodium-Catalyzed Synthesis of Bioactive 1,3-Benzodiazepine Intermediates for Oncology Drug Development
Advanced Rhodium-Catalyzed Synthesis of Bioactive 1,3-Benzodiazepine Intermediates for Oncology Drug Development
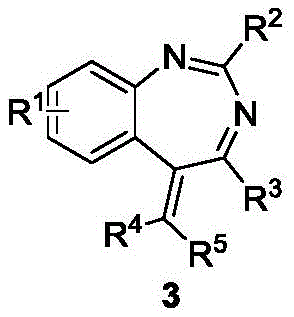
The pharmaceutical industry is constantly seeking efficient pathways to access privileged scaffolds that exhibit potent biological activities, particularly in the realm of oncology. Patent CN112174901A discloses a groundbreaking synthetic methodology for constructing 1,3-benzodiazepine compounds, a class of heterocycles known for their significant anticancer, analgesic, and antidepressant properties. This innovation addresses the critical need for streamlined processes in the production of high-purity pharmaceutical intermediates, utilizing a robust rhodium-catalyzed annulation strategy. The disclosed method transforms simple N-arylamidines and propargyl alcohol esters into complex benzodiazepine cores under remarkably mild conditions, offering a viable solution for scaling up the production of potential drug candidates. By leveraging this technology, manufacturers can overcome the historical bottlenecks associated with benzodiazepine synthesis, such as multi-step sequences and low overall yields. The resulting compounds have demonstrated remarkable efficacy against cancer cell lines like HeLa and Ramos, positioning this synthetic route as a cornerstone for future drug discovery programs targeting solid tumors and hematological malignancies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,3-benzodiazepine skeleton has been plagued by significant synthetic challenges that hinder both research and commercial production. Traditional methods often rely on the condensation of o-phenylenediamines with carboxylic acid derivatives or similar precursors, which frequently require harsh reaction conditions, strong acids or bases, and elevated temperatures that can degrade sensitive functional groups. Furthermore, existing protocols typically suffer from poor atom economy and generate substantial amounts of chemical waste, conflicting with modern green chemistry principles essential for sustainable API manufacturing. The reliance on difficult-to-obtain starting materials further exacerbates supply chain vulnerabilities, leading to increased costs and extended lead times for process development teams. Additionally, many conventional routes lack the flexibility to introduce diverse substituents at key positions on the benzodiazepine ring, limiting the ability of medicinal chemists to perform extensive structure-activity relationship (SAR) studies efficiently. These cumulative drawbacks result in low overall yields and high production costs, making the commercial scale-up of complex benzodiazepine derivatives a financially risky endeavor for many pharmaceutical enterprises.
The Novel Approach
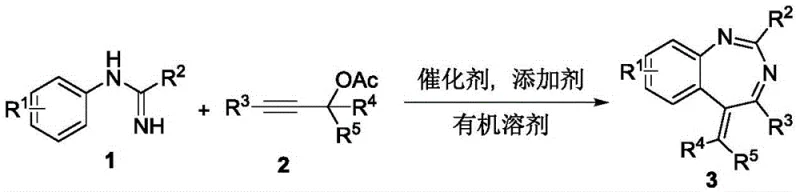
In stark contrast to these legacy methods, the invention described in CN112174901A introduces a highly efficient, one-pot annulation reaction that fundamentally reshapes the synthetic landscape for this scaffold. This novel approach utilizes a rhodium(III) or ruthenium(II) catalyst to mediate the coupling of readily available N-arylamidines with propargyl alcohol esters, proceeding through a cascade sequence that constructs the seven-membered diazepine ring with high precision. The reaction operates under mild thermal conditions, typically around 90°C, and tolerates a wide range of solvents including methanol and ethanol, which are inexpensive and environmentally benign compared to chlorinated alternatives. Crucially, this method exhibits exceptional substrate scope, accommodating various electronic and steric environments on both the amidine and alkyne components without compromising yield or selectivity. The operational simplicity of mixing reagents in a single vessel significantly reduces labor intensity and equipment requirements, facilitating a smoother transition from laboratory discovery to pilot plant operations. This streamlined process not only enhances the speed of compound library generation but also ensures a more reliable supply of high-quality intermediates for downstream drug development.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Annulation
The core of this transformative synthesis lies in the sophisticated organometallic mechanism driven by the pentamethylcyclopentadienyl rhodium(III) catalyst, which facilitates selective C-H bond activation and subsequent cyclization. The catalytic cycle likely initiates with the coordination of the N-arylamidine nitrogen to the cationic rhodium species generated in situ by the silver or copper additive, directing the metal center to the ortho-position of the aromatic ring. This proximity allows for the cleavage of the inert C-H bond, forming a stable five-membered rhodacycle intermediate that serves as the pivotal junction for carbon-carbon bond formation. The propargyl alcohol ester then coordinates to the metal center, undergoing migratory insertion into the Rh-C bond, which extends the carbon framework and sets the stage for ring closure. Subsequent nucleophilic attack by the amidine nitrogen onto the activated alkyne moiety, followed by protonolysis and catalyst regeneration, completes the formation of the 1,3-benzodiazepine core. This mechanistic pathway is highly advantageous as it avoids the use of pre-functionalized halides, thereby reducing the number of synthetic steps and minimizing the generation of stoichiometric salt byproducts. Understanding this cycle is crucial for process chemists aiming to optimize reaction parameters such as catalyst loading and additive ratios to maximize turnover numbers and minimize residual metal content in the final API.
From an impurity control perspective, the mild nature of this catalytic system plays a vital role in ensuring the purity profile of the final product meets stringent regulatory standards. Unlike harsh acidic cyclizations that can promote polymerization or decomposition of sensitive intermediates, this rhodium-catalyzed protocol proceeds with high chemoselectivity, preserving delicate functional groups such as halogens, ethers, and esters present on the substrate. The use of copper acetate monohydrate as a co-catalyst or oxidant further aids in maintaining the active oxidation state of the rhodium species, preventing the accumulation of inactive catalyst aggregates that could lead to incomplete conversions and difficult-to-remove impurities. Moreover, the reaction's tolerance to air atmosphere simplifies the operational setup, reducing the risk of contamination from inert gas lines and lowering the barrier for implementation in standard manufacturing facilities. The ability to achieve high yields with minimal side reactions means that downstream purification processes, such as crystallization or chromatography, are less burdensome, directly contributing to cost reduction in API manufacturing. This level of control over the reaction trajectory is essential for producing clinical-grade materials where impurity profiles must be rigorously characterized and controlled.
How to Synthesize 2-(tert-Butyl)-4-phenyl-5-(propan-2-ylidene)-5H-benzo[d][1,3]diazepine Efficiently
To successfully implement this cutting-edge synthesis in a laboratory or pilot setting, precise adherence to the optimized reaction conditions detailed in the patent examples is paramount for achieving reproducible results. The standard procedure involves combining the N-arylamidine substrate, the propargyl alcohol ester coupling partner, the rhodium dimer catalyst, and the copper additive in a suitable alcohol solvent such as methanol. The mixture is then heated to approximately 90°C for a duration of 5 hours, allowing the catalytic cycle to proceed to completion while monitoring conversion via TLC or HPLC. Upon completion, the reaction mixture is cooled to ambient temperature, filtered to remove insoluble salts, and concentrated under reduced pressure to afford the crude product. Final purification is typically achieved through silica gel column chromatography using a petroleum ether and ethyl acetate gradient, yielding the target benzodiazepine as a high-purity solid or oil suitable for biological evaluation. For detailed standardized synthesis steps and specific safety precautions, please refer to the guide below.
- Charge a reaction vessel with N-arylamidine (1a), methanol solvent, rhodium catalyst [RhCp*Cl2]2, and copper acetate additive.
- Add the propargyl alcohol ester compound (2a) to the mixture and seal the vessel.
- Heat the reaction mixture to 90°C in an oil bath for 5 hours under air atmosphere with stirring.
- Cool to room temperature, filter, concentrate, and purify via silica gel column chromatography to isolate the yellow solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this rhodium-catalyzed methodology represents a strategic opportunity to enhance the resilience and cost-efficiency of the pharmaceutical supply chain. The reliance on commercially abundant starting materials, such as substituted anilines and simple alkynes, mitigates the risk of raw material shortages that often plague projects dependent on exotic or custom-synthesized building blocks. Furthermore, the robustness of the reaction conditions allows for execution in standard glass-lined reactors without the need for specialized high-pressure or cryogenic equipment, significantly lowering capital expenditure requirements for scale-up. The high atom economy and reduced waste generation align perfectly with increasingly strict environmental regulations, potentially lowering waste disposal costs and simplifying permitting processes for new manufacturing sites. By streamlining the synthesis from multiple steps to a single pot, companies can drastically reduce the overall production timeline, enabling faster time-to-market for new oncology therapies and improving responsiveness to market demands. This operational efficiency translates directly into a more competitive cost structure, allowing organizations to allocate resources towards further innovation and clinical development rather than struggling with inefficient manufacturing processes.
- Cost Reduction in Manufacturing: The elimination of multiple isolation and purification steps inherent in traditional multi-step syntheses leads to substantial savings in labor, solvents, and energy consumption. By utilizing a catalytic amount of rhodium rather than stoichiometric reagents, the direct material cost per kilogram of product is significantly optimized, despite the precious metal nature of the catalyst. The ability to use inexpensive solvents like methanol instead of hazardous chlorinated solvents further reduces procurement costs and simplifies solvent recovery systems. Additionally, the high yields reported across a broad substrate scope mean that less starting material is wasted, maximizing the return on investment for every batch produced. These cumulative factors create a leaner manufacturing process that is economically superior to legacy methods, providing a clear financial advantage for large-scale production.
- Enhanced Supply Chain Reliability: The use of stable, shelf-stable reagents ensures that inventory management is straightforward, reducing the risk of degradation and spoilage during storage. The reaction's tolerance to air and moisture simplifies logistics, as there is no need for complex inert atmosphere handling during the charging of reagents, which can be a bottleneck in busy manufacturing suites. The wide applicability of this method to various substituted substrates means that a single platform technology can be used to produce a diverse portfolio of intermediates, reducing the need for multiple dedicated production lines. This flexibility allows supply chain planners to respond quickly to changes in demand for different analogs without retooling entire facilities. Consequently, the reliability of supply for critical drug candidates is greatly enhanced, ensuring continuity of clinical trials and commercial launches.
- Scalability and Environmental Compliance: The mild thermal conditions and absence of hazardous reagents make this process inherently safer and easier to scale from gram to ton quantities without encountering exothermic runaway risks. The reduced generation of toxic byproducts aligns with green chemistry initiatives, helping companies meet their sustainability goals and regulatory compliance targets more easily. The simplicity of the workup procedure, involving basic filtration and chromatography, minimizes the complexity of downstream processing equipment needed for large-scale operations. This ease of scale-up reduces the technical risk associated with technology transfer from R&D to manufacturing, accelerating the path to commercial availability. Ultimately, this method supports a sustainable and scalable supply chain capable of meeting the global demand for advanced therapeutic intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route for 1,3-benzodiazepines. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation to provide clarity for potential partners. Understanding these aspects is crucial for evaluating the feasibility of integrating this technology into existing drug development pipelines. We encourage stakeholders to review these points carefully to appreciate the full value proposition of this manufacturing innovation.
Q: What are the key advantages of this Rhodium-catalyzed method over traditional synthesis?
A: This method utilizes readily available N-arylamidines and propargyl alcohol esters in a one-pot reaction, avoiding the long synthetic routes and harsh conditions typical of conventional benzodiazepine synthesis. It offers mild reaction conditions (90°C), wide substrate tolerance, and high atom economy.
Q: Does this synthesis method support structural diversity for drug screening?
A: Yes, the protocol demonstrates excellent functional group tolerance, allowing for variations at the R1, R2, R3, R4, and R5 positions. This enables the rapid generation of a diverse library of 1,3-benzodiazepine derivatives for structure-activity relationship (SAR) studies in oncology.
Q: What is the biological activity profile of the synthesized compounds?
A: The synthesized compounds exhibit significant anticancer activity against various cell lines, particularly HeLa and Ramos cells. Specific derivatives like 3c and 3d showed potent inhibition against HeLa cells, while others like 3w and 3bb were highly effective against Ramos cells, indicating strong potential as lead candidates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Benzodiazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this rhodium-catalyzed synthesis in accelerating the development of next-generation anticancer agents. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless and efficient. Our state-of-the-art facilities are equipped to handle precious metal catalysis with rigorous QC labs dedicated to monitoring residual metal levels, guaranteeing that our outputs meet the most stringent purity specifications required for clinical applications. We are committed to leveraging this advanced chemistry to deliver high-quality intermediates that empower your research and commercial success in the competitive oncology landscape.
We invite you to engage with our technical procurement team to discuss how this specific synthetic route can be tailored to your project's unique requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this efficient methodology for your specific target molecules. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to be your trusted partner in bringing life-saving medicines to patients worldwide.
