Scalable Chemical Resolution for PF-2341066: A Strategic Upgrade for Pharmaceutical Manufacturing
Scalable Chemical Resolution for PF-2341066: A Strategic Upgrade for Pharmaceutical Manufacturing
The development of potent c-Met inhibitors represents a critical frontier in oncology therapeutics, with PF-2341066 standing out as a highly competitive micromolecular inhibitor capable of effectively suppressing c-Met phosphorylation and tumor cell proliferation. As detailed in patent CN101735198A, a novel synthetic methodology has been established that fundamentally shifts the production paradigm from fragile biocatalytic processes to robust chemical resolution techniques. This transition is not merely a laboratory optimization but a strategic manufacturing upgrade that addresses the core bottlenecks of yield, cost, and scalability inherent in previous art. The target molecule, characterized by its complex heterocyclic architecture featuring a dichlorofluorophenyl moiety linked to a pyridine-pyrazole scaffold, demands precise stereochemical control which this new route delivers with exceptional fidelity. By adopting this kinetic resolution strategy, manufacturers can secure a reliable supply of high-purity pharmaceutical intermediates essential for advancing clinical candidates.
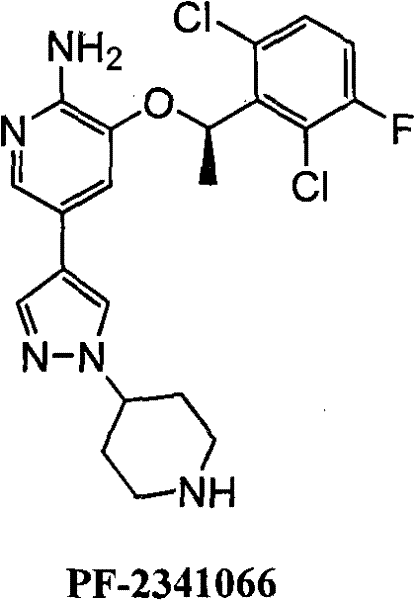
The structural complexity of PF-2341066 necessitates a synthesis route that can maintain rigorous stereochemical integrity while remaining economically viable for large-scale production. The patent disclosure outlines a comprehensive pathway starting from readily available substituted acetophenones, bypassing the limitations of earlier enzymatic approaches. This innovation is particularly relevant for procurement and supply chain leaders who require consistent quality and uninterrupted material flow. The ability to synthesize the key chiral intermediate (III) through a direct chemical resolution step, rather than a multi-step enzymatic hydrolysis, drastically simplifies the operational workflow. Furthermore, the total yield of approximately 20 percent over multiple steps, achieved through optimized conditions, underscores the efficiency of this approach in converting raw materials into valuable active pharmaceutical ingredients without excessive waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in WO2006021881A2 and related documents, relied heavily on esterase-catalyzed hydrolysis to establish the necessary chirality in the phenylethanol intermediate. While enzymatic methods can offer high selectivity, they are plagued by significant industrial drawbacks that hinder commercial viability. The enzymatic catalytic reaction requires extended reaction times and often operates under severe or highly specific conditions that are difficult to maintain in large reactors. Moreover, the cost of enzyme catalysts is prohibitively high, and the reproducibility of biological systems can vary batch-to-batch, leading to inconsistent yields and purity profiles. The process typically involves multiple steps including reduction, acetylation, enzymatic resolution, and subsequent hydrolysis, creating a long and convoluted production cycle that increases the risk of impurity accumulation and operational failure.
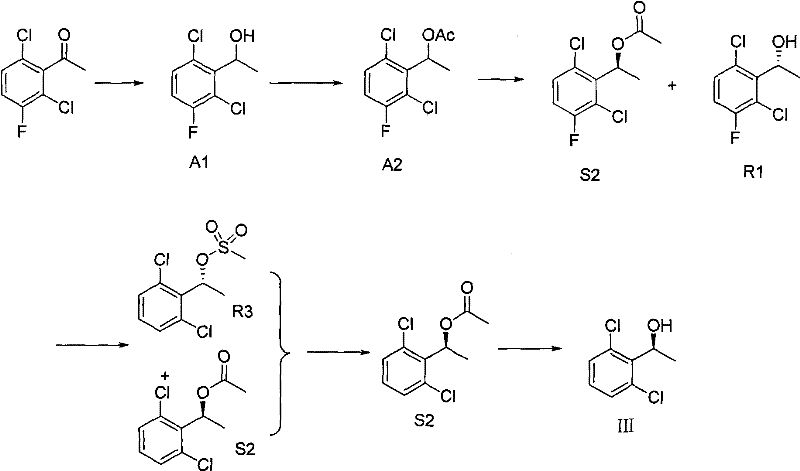
The Novel Approach
In stark contrast, the novel approach presented in CN101735198A utilizes a kinetic resolution of chiral organic small molecules to synthesize the key intermediate (III) in a single, streamlined step. This method replaces the biological catalyst with chemically defined resolving agents, such as N-protected proline or protected mandelic acid derivatives, which are far more stable and cost-effective. The reaction proceeds under mild conditions, typically at room temperature or slightly below, using standard condensing agents like EDCI and catalysts like DMAP. This shift eliminates the dependency on sensitive biological materials and allows for a much simpler workup procedure involving column chromatography to isolate the desired enantiomer. The result is a process that is not only easier to handle but also significantly more amenable to scale-up, providing a robust foundation for the commercial manufacturing of c-Met inhibitors and their derivatives.
Mechanistic Insights into Kinetic Resolution and Chiral Separation
The core of this technological breakthrough lies in the asymmetric esterification mechanism driven by chiral catalysts. In this kinetic resolution process, the racemic phenylethanol (II) reacts with a chiral resolving agent (D-Cat or L-Cat) in the presence of a condensing agent. The chiral environment created by the catalyst differentiates between the two enantiomers of the alcohol, causing one enantiomer to react significantly faster to form the ester (XIa or XIb), while the other remains largely unreacted as the free alcohol (IIIa or IIIb). This differential reaction rate allows for the physical separation of the enantiomers, typically yielding the desired chiral alcohol with an optical purity (ee value) exceeding 99 percent. The choice of protecting groups on the catalyst, such as Boc or Cbz, plays a crucial role in modulating the steric and electronic properties to maximize this selectivity.
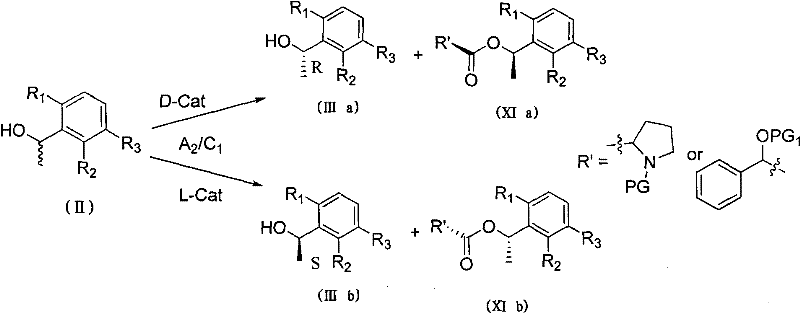
Beyond the primary resolution step, the subsequent transformation of the chiral intermediate into the final API involves a series of well-controlled reactions that preserve the established stereochemistry. The Mitsunobu reaction couples the chiral alcohol with a nitro-pyridone, followed by a reduction step using iron powder to generate the aromatic amine. Crucially, these steps are designed to prevent racemization at the chiral center, ensuring that the high optical purity achieved in the resolution step is carried through to the final product. The use of specific reagents like NBS for bromination and palladium catalysts for Suzuki coupling further demonstrates a commitment to high-yielding, selective transformations. This meticulous control over the reaction pathway minimizes the formation of diastereomeric impurities, thereby simplifying the purification process and enhancing the overall quality of the pharmaceutical intermediate.
How to Synthesize PF-2341066 Efficiently
The synthesis of PF-2341066 via this novel route offers a clear blueprint for manufacturers seeking to optimize their production capabilities. The process begins with the reduction of substituted acetophenone to the racemic alcohol, followed by the critical kinetic resolution step that sets the stereochemistry. Subsequent functionalization involves etherification, reduction, bromination, and cross-coupling reactions to assemble the final molecular architecture. The detailed standardized synthesis steps provided in the patent documentation serve as a vital guide for process chemists aiming to replicate these results in a pilot or production setting. By adhering to the specified reaction conditions and reagent ratios, facilities can achieve consistent outcomes that meet the stringent requirements of regulatory bodies.
- Reduce substituted acetophenone to racemic phenylethanol using borohydride reagents at controlled low temperatures.
- Perform kinetic resolution using chiral catalysts (D-Cat or L-Cat) and condensing agents to separate enantiomers via esterification.
- Execute downstream coupling reactions including Mitsunobu etherification, nitro reduction, bromination, and Suzuki coupling to finalize the API structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this chemical resolution methodology presents a compelling value proposition centered on cost efficiency and supply security. The elimination of expensive enzymatic catalysts and the reduction in processing time directly translate to lower manufacturing costs, making the final API more economically attractive. Furthermore, the reliance on commodity chemicals rather than specialized biological reagents mitigates the risk of supply disruptions, ensuring a more stable and predictable sourcing strategy. This robustness is essential for maintaining continuity in the production of life-saving oncology medications, where any interruption can have significant clinical consequences.
- Cost Reduction in Manufacturing: The transition from enzymatic to chemical catalysis removes the burden of high-cost biocatalysts and the complex infrastructure required to support them. By utilizing standard organic reagents and simplified reaction conditions, the overall cost of goods sold is significantly reduced. The streamlined process also reduces energy consumption and waste disposal costs associated with long reaction times and multiple purification steps, leading to substantial economic savings throughout the production lifecycle.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this synthetic route is straightforward, as substituted acetophenones and common coupling agents are widely available from multiple global suppliers. This diversity in the supply base reduces dependency on single-source vendors and protects against market volatility. Additionally, the chemical stability of the reagents allows for longer shelf lives and easier storage, further enhancing the resilience of the supply chain against logistical challenges and demand fluctuations.
- Scalability and Environmental Compliance: The simplicity of the reaction conditions facilitates easy scale-up from laboratory to commercial production volumes without the need for specialized equipment. The process generates fewer by-products and avoids the use of hazardous biological waste, aligning with modern environmental standards and green chemistry principles. This compliance not only reduces regulatory hurdles but also enhances the corporate sustainability profile of the manufacturing entity, appealing to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and commercial implications of this synthesis method. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios. The answers provided are derived directly from the technical disclosures and experimental data within the patent, ensuring accuracy and relevance for decision-makers.
Q: Why is the chemical resolution method superior to enzymatic hydrolysis for PF-2341066?
A: The chemical resolution method eliminates the need for expensive biological enzymes, significantly shortens reaction cycles, and offers better reproducibility and scalability for industrial production compared to the harsh conditions and low yields associated with enzymatic catalysis.
Q: What represents the key chiral intermediate in this synthesis route?
A: The key chiral intermediate is the substituted phenylethanol (Compound III), which is obtained with high optical purity (ee > 99%) through a one-step kinetic resolution process using protected proline or mandelic acid derivatives.
Q: How does this process impact supply chain stability for c-Met inhibitors?
A: By utilizing widely available raw materials like substituted acetophenones and standard chemical reagents instead of specialized biocatalysts, the process ensures a more robust and continuous supply chain, reducing dependency on niche biological suppliers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable PF-2341066 Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the kinetic resolution route for PF-2341066. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle complex chiral separations and multi-step syntheses with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards. We are committed to delivering excellence in every aspect of pharmaceutical intermediate production.
We invite you to collaborate with us to leverage this innovative synthesis route for your c-Met inhibitor programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your drug development timeline with reliable, high-quality supply solutions.
