Advanced Synthesis of Brexpiprazole Impurity F: Enhancing Quality Control and Supply Reliability
Advanced Synthesis of Brexpiprazole Impurity F: Enhancing Quality Control and Supply Reliability
The pharmaceutical industry's relentless pursuit of safety and efficacy places immense pressure on the availability of high-quality reference standards, particularly for complex antipsychotic agents like Brexpiprazole. Patent CN115108981A introduces a groundbreaking synthesis method for Piperazine Butoxy Double-Grafting Impurity (Compound F), a critical related substance that must be strictly monitored during the manufacturing of the active pharmaceutical ingredient (API). This innovation addresses a significant gap in the supply chain for quality control materials, offering a robust, scalable, and high-yield route that bypasses the limitations of traditional isolation methods. By leveraging a targeted nucleophilic substitution strategy, this technology ensures that manufacturers can secure reliable sources of impurity standards, thereby safeguarding the integrity of their quality assurance protocols and regulatory filings.
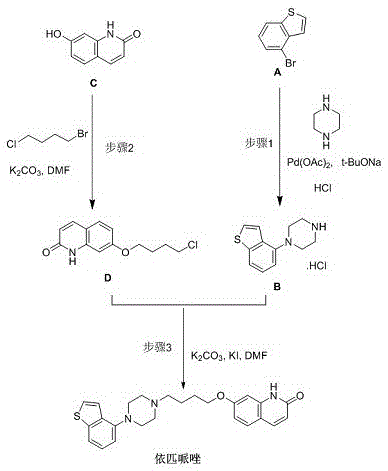
In the conventional production of Brexpiprazole, the formation of Impurity F is often an unintended side reaction occurring when residual piperazine reacts with the quinolinone intermediate. Historically, obtaining this impurity for analytical validation required laborious isolation from crude reaction masses, a process plagued by low recovery rates and inconsistent purity profiles. The background art highlights that while excess piperazine is used in Step 1 of the API synthesis to suppress other impurities (like Impurity E), any unreacted piperazine carried forward can react with Compound C or its derivatives to form Impurity F. This accidental generation makes it nearly impossible to rely on production batches as a consistent source for reference standards, creating a bottleneck for R&D teams who require gram-to-kilogram quantities for method validation and stability studies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to sourcing Brexpiprazole Impurity F were inherently inefficient and economically unsustainable for commercial-scale reference material production. The primary defect lay in the reliance on 'chance chemistry,' where the impurity was merely a byproduct of the main API synthesis rather than a target molecule. Attempting to isolate Impurity F from the complex matrix of an API reaction mixture involves navigating a minefield of structurally similar analogs, unreacted starting materials, and other process impurities. This necessitates extensive preparative HPLC or multiple recrystallization steps, which drastically reduce overall yield and inflate the cost per gram. Furthermore, the batch-to-batch variability in the formation of this side product means that supply is unpredictable; a manufacturer cannot guarantee the availability of the impurity standard simply by running more API batches, as process optimizations aimed at minimizing impurities would inadvertently starve the supply of the very standard needed to test them.
The Novel Approach
The methodology disclosed in CN115108981A represents a paradigm shift by treating Impurity F as a primary target rather than a waste product. The novel approach utilizes a direct condensation reaction between Compound D (7-(4-chlorobutoxy)quinolin-2(1H)-one) and piperazine under carefully controlled stoichiometric conditions. By deliberately employing an excess of Compound D (ranging from 1.2 to 4.0 equivalents, optimally 2.2 equivalents) relative to piperazine, the reaction thermodynamics are driven exclusively toward the double-substitution product. This strategic manipulation of molar ratios ensures that both nitrogen atoms of the piperazine ring are alkylated, effectively shutting down the formation of mono-substituted intermediates. The result is a clean reaction profile that simplifies downstream processing, allowing for high-purity isolation through straightforward crystallization and filtration techniques rather than complex chromatographic separations.
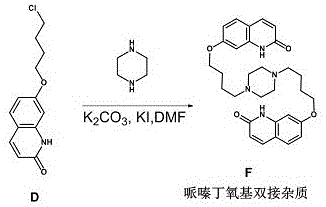
Mechanistic Insights into Base-Catalyzed Nucleophilic Substitution
The core chemical transformation driving this synthesis is a classic bimolecular nucleophilic substitution (SN2), facilitated by the presence of a strong base and polar aprotic or protic solvents. In this mechanism, the secondary amine groups of piperazine act as nucleophiles, attacking the terminal carbon of the chlorobutyl chain attached to the quinolinone ring of Compound D. The role of the base, whether it be potassium carbonate in DMF or potassium hydroxide in methanol, is critical; it serves to deprotonate the piperazine nitrogens, significantly enhancing their nucleophilicity and enabling them to displace the chloride leaving group. The addition of potassium iodide (KI) in the DMF system acts as a phase-transfer catalyst or a halogen exchange promoter, potentially converting the less reactive alkyl chloride into a more reactive alkyl iodide in situ, thereby accelerating the reaction rate and allowing it to proceed efficiently at moderate temperatures between 60°C and 140°C.
From an impurity control perspective, the mechanistic precision of this route is its greatest asset. By maintaining a strict molar excess of the electrophile (Compound D), the probability of a single piperazine molecule reacting with two different quinolinone molecules is maximized, while the probability of unreacted piperazine remaining is minimized. This stands in stark contrast to the API synthesis where piperazine is in excess to prevent double-alkylation on the quinolinone side. Understanding this inversion of stoichiometry is key for process chemists; it allows for the deterministic production of the impurity with a purity profile that can consistently exceed 99%, as evidenced by the HPLC data in the patent examples. The robustness of the mechanism across different solvent systems (DMF vs. Methanol) further indicates a wide operating window, reducing the risk of batch failure due to minor fluctuations in reaction conditions.
How to Synthesize Piperazine Butoxy Double-Grafting Impurity Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to ensure optimal yield and purity. The process is designed to be operationally simple, utilizing readily available reagents and standard glassware or stainless steel reactors. The key to success lies in the precise weighing of reagents to maintain the critical 1:2.2 molar ratio between piperazine and Compound D, as well as the selection of the appropriate base-solvent combination based on available infrastructure. While the patent outlines specific examples, the general procedure involves mixing the reagents, heating to reflux or near-reflux temperatures for an extended period (10-30 hours) to ensure complete conversion, and then employing a pH-swing crystallization technique to isolate the free base or salt form of the impurity. For detailed step-by-step instructions including exact quantities and workup procedures, please refer to the standardized guide below.
- Prepare the reaction mixture by combining piperazine, a base such as potassium carbonate or potassium hydroxide, and an organic solvent like DMF or methanol in a reactor.
- Add Compound D (7-(4-chlorobutoxy)quinolin-2(1H)-one) in a molar excess (1.2 to 4.0 equivalents relative to piperazine) along with a catalytic amount of potassium iodide if using DMF.
- Heat the mixture to 60-140°C for 10-30 hours to facilitate double nucleophilic substitution, followed by crystallization and purification to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method offers tangible strategic benefits that extend beyond mere technical feasibility. The ability to produce Brexpiprazole Impurity F on demand transforms it from a scarce, high-cost specialty chemical into a manageable commodity. This shift mitigates the risk of supply disruptions that could halt quality control testing, a scenario that can lead to costly delays in batch release and regulatory submissions. By internalizing the production of this critical reference standard or sourcing it from suppliers utilizing this efficient route, organizations can decouple their quality assurance capabilities from the vagaries of API production schedules.
- Cost Reduction in Manufacturing: The economic implications of this route are profound, primarily driven by the elimination of expensive purification steps. Traditional isolation methods often rely on preparative HPLC, which consumes vast amounts of solvent and stationary phase, driving up the cost per gram exponentially. In contrast, this direct synthesis achieves high purity through crystallization, a unit operation that is inherently cheaper and more scalable. Furthermore, the reagents involved—piperazine, potassium carbonate, and Compound D—are commodity chemicals with stable pricing, avoiding the volatility associated with precious metal catalysts or exotic ligands. The simplified workflow also reduces labor hours and energy consumption, contributing to a significantly lower overall cost of goods sold (COGS) for the reference standard.
- Enhanced Supply Chain Reliability: Supply continuity is paramount in the pharmaceutical sector, and this method enhances reliability by simplifying the raw material portfolio. Unlike routes that might depend on custom-synthesized precursors with long lead times, this process starts from Compound D, an intermediate that is structurally simple and widely accessible within the fine chemical supply chain. The robustness of the reaction conditions means that production is less susceptible to minor variations in raw material quality or environmental factors, ensuring consistent output. This predictability allows supply chain planners to maintain leaner inventories of the impurity standard without fear of stockouts, optimizing working capital and storage resources.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process is exceptionally well-suited for commercial expansion. The use of common solvents like DMF or Methanol facilitates easy solvent recovery and recycling, aligning with green chemistry principles and reducing waste disposal costs. The absence of heavy metals or toxic reagents simplifies the effluent treatment process, lowering the environmental footprint of the manufacturing site. Moreover, the reaction does not require extreme pressures or cryogenic temperatures, meaning it can be scaled from gram to multi-kilogram quantities using existing general-purpose reactors without the need for capital-intensive equipment upgrades, ensuring a smooth transition from R&D to commercial supply.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Brexpiprazole Impurity F. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, aiming to clarify the operational benefits and regulatory relevance of this synthesis method for industry stakeholders.
Q: Why is the synthesis of Piperazine Butoxy Impurity F critical for Brexpiprazole manufacturing?
A: Impurity F is a potential process-related impurity formed when excess piperazine reacts with the quinolinone intermediate during API synthesis. Having a certified reference standard is mandatory for regulatory compliance and accurate HPLC quantification to ensure patient safety.
Q: How does this patented method improve purity compared to isolation from crude API batches?
A: Traditional isolation from crude reaction mixtures yields low quantities and requires complex chromatography. This method uses controlled stoichiometry (excess Compound D) to drive the reaction specifically toward the double-substituted product, achieving purities exceeding 99% with simpler workup procedures.
Q: What are the key scalability advantages of this synthetic route?
A: The process utilizes common industrial solvents (DMF, Methanol) and inexpensive inorganic bases (K2CO3, KOH). It avoids sensitive catalysts and operates at moderate temperatures (60-140°C), making it highly suitable for multi-kilogram production without specialized equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brexpiprazole Impurity F Supplier
As the global demand for high-quality antipsychotic medications continues to rise, the need for rigorous quality control becomes ever more critical. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies like the one described in CN115108981A to deliver superior reference standards. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether for early-stage method development or full-scale QC operations. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Brexpiprazole Impurity F meets the highest international standards.
We invite you to collaborate with us to optimize your supply chain for Brexpiprazole related substances. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific consumption patterns and quality needs. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations that demonstrate how our efficient manufacturing processes can add value to your organization.
