Advanced Diazoxide Manufacturing: Overcoming Toxicity and Cost Barriers in API Intermediates
Advanced Diazoxide Manufacturing: Overcoming Toxicity and Cost Barriers in API Intermediates
The pharmaceutical landscape for cardiovascular and hypoglycemic treatments relies heavily on the consistent supply of critical active ingredients like Diazoxide, chemically known as 7-chloro-3-methyl-2H-1,2,4-benzothiadiazine 1,1-dioxide. As detailed in patent CN110903264B, a significant technological breakthrough has emerged that fundamentally alters the production economics and safety profile of this essential medicine. Traditional manufacturing pathways have long been plagued by the use of hazardous reagents such as chlorosulfonyl isocyanate and the necessity for extreme thermal conditions exceeding 240°C, which impose severe limitations on scalability and operator safety. The novel methodology introduced in this intellectual property leverages a sophisticated imidazole hydrochloride-catalyzed cyclization in N,N-dimethylacetamide, offering a streamlined alternative that bypasses these historical bottlenecks. For R&D directors and procurement specialists alike, this shift represents not merely a chemical optimization but a strategic supply chain enhancement, ensuring that high-purity diazoxide can be produced with greater reliability and reduced environmental impact. By adopting this refined synthetic architecture, manufacturers can achieve total yields surpassing 90% while drastically simplifying the purification workflow.
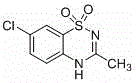
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of diazoxide has been constrained by three primary routes, labeled A, B, and C in prior art, each carrying distinct operational liabilities that hinder efficient commercial scale-up of complex heterocyclic intermediates. Routes A and B, for instance, initiate with p-chloroaniline and necessitate the utilization of chlorosulfonyl isocyanate, a reagent notorious for its extreme toxicity, high reactivity, and sensitivity to moisture, which demands rigorous anhydrous conditions and specialized handling infrastructure. Furthermore, these legacy processes often require Lewis acids like aluminum chloride, which are prone to hydrolysis and generate substantial acidic waste streams, complicating downstream processing and wastewater treatment. The final cyclization steps in these older methods frequently demand aggressive conditions, such as heating in acetic acid at 130°C or utilizing concentrated sulfuric acid, which accelerates equipment corrosion and increases maintenance overheads. Route C, while avoiding some halogenating agents, still relies on triethyl orthoacetate and requires high-temperature reflux, presenting flammability risks and higher raw material costs compared to modern alternatives. Collectively, these factors result in longer production cycles, lower overall atom economy, and a heightened risk profile that is increasingly untenable in modern regulated manufacturing environments.
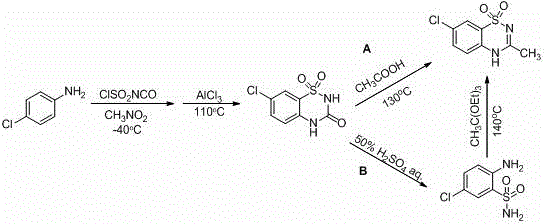
The Novel Approach
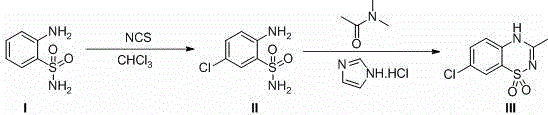
In stark contrast to these cumbersome predecessors, the innovative strategy disclosed in the patent introduces two highly efficient pathways, designated as Route D and Route E, which prioritize mild conditions and commercially accessible starting materials. Route D, for example, initiates with the electrophilic chlorination of o-aminobenzenesulfonamide using N-chlorosuccinimide (NCS) in a benign chlorinated solvent, followed by a catalytic cyclization in N,N-dimethylacetamide. This approach completely obviates the need for chlorosulfonyl isocyanate and strong mineral acids, thereby neutralizing the corrosion and toxicity issues that plague conventional methods. The use of imidazole hydrochloride as a catalyst is particularly transformative, enabling the ring-closure reaction to proceed at moderate temperatures (120-150°C) with exceptional conversion rates. Similarly, Route E offers a flexible alternative where the cyclization occurs prior to chlorination, demonstrating the robustness of the imidazole-mediated mechanism. By replacing expensive and unstable reagents like triethyl orthoacetate with stable, low-cost amides, this new chemistry significantly lowers the barrier to entry for production, facilitating cost reduction in pharmaceutical intermediates manufacturing while simultaneously improving the purity profile of the final API.
Mechanistic Insights into Imidazole-Catalyzed Cyclization
The core innovation of this patent lies in the mechanistic role of the imidazole salt, specifically imidazole hydrochloride, which serves as a potent yet manageable proton source to drive the thermodynamic equilibrium toward the formation of the benzothiadiazine ring. In the context of Route D, once the 2-amino-5-chlorobenzenesulfonamide intermediate is generated, the subsequent reaction with N,N-dimethylacetamide involves a nucleophilic attack by the sulfonamide nitrogen on the carbonyl carbon of the amide. The imidazole cation stabilizes the transition state and facilitates the elimination of dimethylamine, a volatile byproduct that drives the reaction forward according to Le Chatelier's principle. This catalytic cycle is remarkably efficient, requiring only 1 to 12 mol% of the salt, with optimal performance observed at approximately 5 mol%, ensuring that the catalyst loading remains economically viable for large-scale operations. The choice of N,N-dimethylacetamide as the solvent is critical, as it provides the necessary polarity to solubilize the polar sulfonamide substrate while remaining stable under the reflux conditions required for cyclization, unlike DMF which was found ineffective in comparative experiments. This precise tuning of reaction parameters ensures that side reactions, such as polymerization or over-chlorination, are minimized, leading to the high purity levels (>99%) reported in the experimental examples.
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed routes that often generate sulfonated byproducts or charred residues due to localized overheating. The homogeneous nature of the imidazole hydrochloride catalyst in the amide solvent ensures uniform heat distribution and reaction kinetics, preventing the formation of hot spots that could degrade the sensitive diazoxide structure. Furthermore, the mild acidity of the imidazole salt prevents the hydrolysis of the sulfonamide group, a common degradation pathway in strongly acidic media like sulfuric acid. The chlorination step using NCS is equally selective, targeting the para-position relative to the amino group with high regioselectivity, thereby avoiding the formation of isomeric impurities that would be difficult to separate later. This level of control is paramount for meeting the stringent purity specifications required for injectable formulations, where even trace impurities can trigger adverse immunological responses. Consequently, the mechanistic elegance of this route translates directly into a cleaner crude product, reducing the burden on downstream purification units and enhancing the overall process mass intensity.
How to Synthesize Diazoxide Efficiently
Implementing this novel synthetic route requires careful attention to solvent quality and temperature control to maximize the benefits of the imidazole catalysis. The process begins with the preparation of the key intermediate, 2-amino-5-chlorobenzenesulfonamide, which serves as the foundational building block for the subsequent ring closure. Operators must ensure that the chlorination step is driven to completion to prevent carryover of unreacted starting material, which could complicate the cyclization kinetics. Following isolation, the intermediate is subjected to the cyclization conditions in N,N-dimethylacetamide, where the precise molar ratio of the catalyst becomes a critical process parameter. While the patent outlines a broad range of effective catalyst loadings, adhering to the optimized 5 mol% range provides the best balance between reaction rate and cost efficiency. The detailed standardized synthesis steps below outline the specific operational parameters derived from the patent examples to guide process engineers in replicating these high-yield results.
- React o-aminobenzenesulfonamide with N-chlorosuccinimide (NCS) in a chlorinated solvent like dichloromethane or chloroform under reflux to obtain 2-amino-5-chlorobenzenesulfonamide.
- Mix the resulting 2-amino-5-chlorobenzenesulfonamide with N,N-dimethylacetamide (DMA) and a catalytic amount of imidazole hydrochloride (approx. 5 mol%).
- Heat the mixture to reflux temperature (approx. 120-150°C) for 6-8 hours to effect cyclization, followed by purification to isolate diazoxide with yields exceeding 90%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers profound strategic benefits that extend far beyond simple chemical yield improvements. By transitioning away from hazardous reagents like chlorosulfonyl isocyanate and aluminum chloride, facilities can significantly reduce their expenditure on specialized corrosion-resistant equipment and hazardous waste disposal services. The elimination of strong acids and high-temperature steps also lowers the energy consumption profile of the manufacturing process, contributing to broader sustainability goals and reducing utility costs. Furthermore, the reliance on commodity chemicals such as N,N-dimethylacetamide and N-chlorosuccinimide ensures a stable and resilient supply chain, mitigating the risks associated with sourcing niche or highly regulated precursors. This robustness allows for more predictable production scheduling and reduces the likelihood of supply disruptions caused by raw material shortages or regulatory changes affecting toxic substances.
- Cost Reduction in Manufacturing: The economic implications of this new route are substantial, primarily driven by the substitution of expensive and unstable reagents with cost-effective alternatives. Triethyl orthoacetate, used in older Route C, is significantly more expensive than N,N-dimethylacetamide, and the new process reportedly utilizes a solvent that is over 15 times cheaper while delivering superior yields. Additionally, the reduction in reaction steps and the avoidance of complex workup procedures associated with Lewis acid quenching translate into lower labor and processing costs per kilogram of output. The high selectivity of the reaction minimizes the loss of valuable starting materials to byproduct formation, thereby improving the overall atom economy and reducing the cost of goods sold. These factors combine to create a leaner manufacturing model that enhances margin potential without compromising on product quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is critically dependent on the availability and stability of raw materials, and this new synthesis excels by utilizing widely available industrial chemicals. Unlike chlorosulfonyl isocyanate, which requires strict anhydrous storage and transport conditions, the reagents in this process are stable and easy to handle, reducing logistical complexities and storage costs. The shorter reaction times and milder conditions also allow for faster batch turnover, enabling manufacturers to respond more agilely to fluctuations in market demand. This flexibility is essential for maintaining service levels to downstream pharmaceutical customers who require just-in-time delivery of critical API intermediates. By de-risking the production process, companies can offer more reliable lead times and strengthen their position as a trusted partner in the global pharmaceutical supply network.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden challenges, but the simplicity of this imidazole-catalyzed route suggests excellent scalability potential. The absence of exothermic hazards associated with chlorosulfonyl isocyanate and the use of standard reflux conditions make the process safer to operate at the multi-ton scale. From an environmental perspective, the reduction in hazardous waste generation aligns with increasingly stringent global regulations on chemical manufacturing emissions. The ability to recover and recycle the amide solvent further enhances the green credentials of the process, appealing to environmentally conscious stakeholders and regulators. This compliance advantage not only future-proofs the manufacturing asset but also simplifies the permitting process for capacity expansions, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel diazoxide synthesis. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the practical aspects of the technology. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this manufacturing route.
Q: Why is imidazole hydrochloride preferred over traditional acid catalysts for diazoxide synthesis?
A: Imidazole hydrochloride acts as a mild yet effective acid catalyst that facilitates the cyclization step in N,N-dimethylacetamide without the severe corrosion risks associated with concentrated sulfuric acid or the moisture sensitivity of aluminum chloride used in legacy processes.
Q: What are the safety advantages of this new route compared to Route A and B?
A: This method eliminates the use of chlorosulfonyl isocyanate, a highly toxic and water-sensitive reagent, and avoids reaction temperatures as high as 240-250°C, significantly reducing operational hazards and equipment stress.
Q: Can the solvent N,N-dimethylacetamide be substituted in this process?
A: Experimental data indicates that substituting N,N-dimethylacetamide with DMF fails to produce the desired compound IV or diazoxide efficiently, highlighting the specific solvation and stability properties of DMA required for this catalytic cycle.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diazoxide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic methodologies requires a partner with deep technical expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent can be fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped to verify the identity and potency of every batch, guaranteeing that our diazoxide meets the exacting standards required for cardiovascular and hypoglycemic therapies. Our infrastructure is designed to handle the specific solvent systems and catalytic conditions of this new route, allowing us to deliver high-purity diazoxide with consistent reliability.
We invite pharmaceutical partners to engage with our technical procurement team to discuss how this innovative synthesis can optimize your supply chain and reduce overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and sustainable chemistry available in the market.
