Revolutionizing Pharmaceutical Intermediate Production via One-Step Cyclization Technology
Introduction to Advanced Intermediate Synthesis
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for greener, more efficient, and cost-effective synthetic routes. A pivotal advancement in this domain is detailed in patent CN111303078A, which discloses a novel synthesis method for tert-butyl 1,2,3-oxathiazolidine-3-carboxylate 2,2-dioxide compounds. These heterocyclic structures serve as critical building blocks in medicinal chemistry, often acting as key scaffolds for bioactive molecules. The traditional reliance on multi-step sequences involving heavy metal catalysts has long been a bottleneck for both R&D scalability and commercial viability. This new technology addresses these pain points directly by introducing a streamlined one-step cyclization strategy that utilizes sulfuryl chloride as a dual-purpose reagent. For procurement managers and supply chain directors, this represents a significant opportunity to optimize the sourcing of high-purity pharmaceutical intermediates while mitigating risks associated with complex supply chains dependent on scarce catalytic materials.
The strategic importance of this innovation cannot be overstated for stakeholders focused on long-term production stability. By simplifying the molecular construction of these oxathiazolidine dioxides, the patent offers a pathway to reduce lead times and enhance the overall robustness of the manufacturing process. The ability to generate diverse derivatives, including chiral variants with methyl, ethyl, or tert-butyl substitutions, underscores the versatility of this method. As a reliable pharmaceutical intermediate supplier, understanding and adopting such cutting-edge methodologies is essential for maintaining a competitive edge in the global market. The following analysis delves deep into the technical nuances, comparing legacy methods with this breakthrough approach to highlight its transformative potential for cost reduction in pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4- or 5-substituted tert-butyl 1,2,3-oxathiazolidine-3-carboxylate 2,2-dioxide compounds has been plagued by inefficiencies inherent in two-step protocols. As illustrated in prior art such as Chinese patent CN1250549C and literature from Organic Letters (2018), the conventional route typically involves an initial cyclization using thionyl chloride followed by a separate oxidation step. 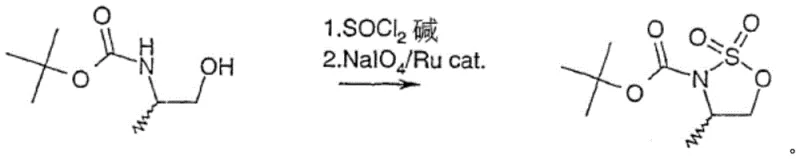
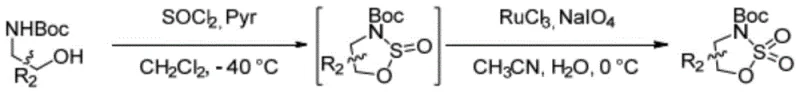 This second oxidation stage critically depends on the use of ruthenium-based catalysts, such as ruthenium trichloride or ruthenium dioxide, alongside strong oxidants like sodium metaperiodate. The reliance on these transition metals introduces severe complications, including the necessity for rigorous heavy metal removal processes to meet stringent pharmaceutical purity standards. Furthermore, the use of toxic substances and the generation of substantial chemical waste during the oxidation phase significantly escalate environmental compliance costs and operational hazards.
This second oxidation stage critically depends on the use of ruthenium-based catalysts, such as ruthenium trichloride or ruthenium dioxide, alongside strong oxidants like sodium metaperiodate. The reliance on these transition metals introduces severe complications, including the necessity for rigorous heavy metal removal processes to meet stringent pharmaceutical purity standards. Furthermore, the use of toxic substances and the generation of substantial chemical waste during the oxidation phase significantly escalate environmental compliance costs and operational hazards.
Beyond the environmental and safety concerns, the economic implications of the conventional two-step method are profound. Ruthenium is a precious metal with volatile pricing and limited availability, making the cost of goods sold (COGS) unpredictable and often prohibitively high for large-scale applications. The multi-step nature also inherently lowers the overall yield due to material losses during isolation and purification between steps. For a procurement manager, this translates to higher unit costs and increased vulnerability to supply chain disruptions caused by catalyst shortages. Additionally, the complexity of the reaction conditions, often requiring inert atmospheres and precise temperature controls for multiple distinct stages, demands specialized equipment and highly skilled labor, further eroding profit margins. These cumulative factors render the traditional approach suboptimal for modern, high-volume commercial production where efficiency and sustainability are paramount.
The Novel Approach
In stark contrast to the cumbersome legacy methods, the technology disclosed in CN111303078A presents a paradigm shift through a direct, one-step cyclization process. 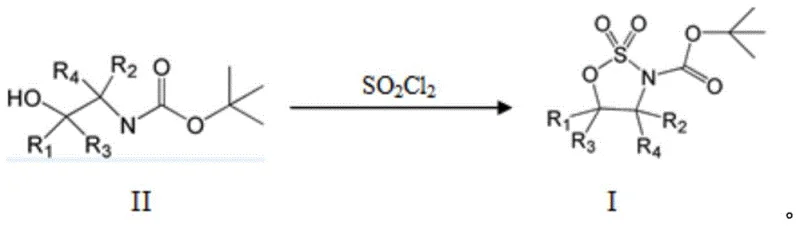 This innovative route reacts a compound of formula II, which is an N-Boc protected amino alcohol, directly with sulfuryl chloride (SO2Cl2) in the presence of an organic solvent to yield the target formula I compound. By consolidating the cyclization and oxidation into a single transformation, this method completely eliminates the need for external oxidizing agents and expensive ruthenium catalysts. The reaction proceeds efficiently under mild conditions, typically between -20°C and -5°C, utilizing a solvent system that promotes rapid conversion. This simplification not only drastically reduces the reaction time but also minimizes the formation of byproducts, leading to cleaner reaction profiles and easier downstream processing. The elimination of the heavy metal catalyst is perhaps the most significant advantage, removing the burden of metal scavenging and ensuring the final product meets rigorous impurity specifications without additional purification burdens.
This innovative route reacts a compound of formula II, which is an N-Boc protected amino alcohol, directly with sulfuryl chloride (SO2Cl2) in the presence of an organic solvent to yield the target formula I compound. By consolidating the cyclization and oxidation into a single transformation, this method completely eliminates the need for external oxidizing agents and expensive ruthenium catalysts. The reaction proceeds efficiently under mild conditions, typically between -20°C and -5°C, utilizing a solvent system that promotes rapid conversion. This simplification not only drastically reduces the reaction time but also minimizes the formation of byproducts, leading to cleaner reaction profiles and easier downstream processing. The elimination of the heavy metal catalyst is perhaps the most significant advantage, removing the burden of metal scavenging and ensuring the final product meets rigorous impurity specifications without additional purification burdens.
The versatility of this novel approach is demonstrated by its applicability to a wide range of substrates with varying steric and electronic properties. Whether the starting material contains methyl, ethyl, tert-butyl, or methoxymethyl groups at the 4 or 5 positions, the reaction maintains high efficiency and selectivity. 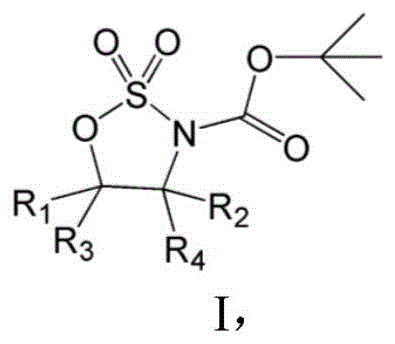 This broad substrate scope is crucial for R&D directors who require flexible synthetic platforms to access diverse libraries of analogs for drug discovery programs. The use of readily available reagents like sulfuryl chloride and common organic bases like DIPEA ensures that the supply chain remains robust and cost-effective. From a commercial perspective, this one-step protocol offers a compelling value proposition by lowering the barrier to entry for producing these valuable intermediates. It aligns perfectly with the industry's push towards green chemistry principles by reducing waste generation and energy consumption, thereby enhancing the sustainability profile of the manufacturing process while delivering substantial cost savings.
This broad substrate scope is crucial for R&D directors who require flexible synthetic platforms to access diverse libraries of analogs for drug discovery programs. The use of readily available reagents like sulfuryl chloride and common organic bases like DIPEA ensures that the supply chain remains robust and cost-effective. From a commercial perspective, this one-step protocol offers a compelling value proposition by lowering the barrier to entry for producing these valuable intermediates. It aligns perfectly with the industry's push towards green chemistry principles by reducing waste generation and energy consumption, thereby enhancing the sustainability profile of the manufacturing process while delivering substantial cost savings.
Mechanistic Insights into SO2Cl2-Mediated Cyclization
The core of this technological breakthrough lies in the unique reactivity of sulfuryl chloride (SO2Cl2) which acts as both a chlorinating agent and a source of the sulfonyl group required for ring closure. In the proposed mechanism, the hydroxyl group of the N-Boc amino alcohol precursor initially attacks the sulfur atom of the sulfuryl chloride, displacing a chloride ion and forming a reactive chlorosulfite intermediate. This species is highly electrophilic and primed for intramolecular nucleophilic attack. Subsequently, the nitrogen atom, activated by the adjacent electron-withdrawing Boc group but still possessing sufficient nucleophilicity, attacks the sulfur center, displacing the remaining chloride or facilitating the elimination of HCl to close the five-membered oxathiazolidine ring. The presence of a base, such as DIPEA or triethylamine, is critical in this sequence to scavenge the generated hydrogen chloride, driving the equilibrium forward and preventing acid-catalyzed decomposition of the sensitive Boc protecting group. This concerted mechanism allows for the simultaneous formation of the S-O and S-N bonds required to establish the cyclic sulfonamide structure in a single operational step.
Impurity control is inherently superior in this one-step design compared to the multi-step alternative. In the conventional ruthenium-catalyzed oxidation, over-oxidation or incomplete oxidation can lead to complex mixtures of sulfoxides, sulfones, and degraded byproducts that are difficult to separate. In the SO2Cl2 mediated pathway, the oxidation state of the sulfur is fixed at the +6 state (sulfone) from the outset, as sulfuryl chloride is already fully oxidized. This thermodynamic stability prevents the formation of lower oxidation state impurities like sulfinamides. Furthermore, the reaction conditions are sufficiently mild to preserve the stereochemical integrity of chiral centers adjacent to the reaction site, as evidenced by the successful synthesis of enantiomerically pure (R) and (S) derivatives with high optical purity. 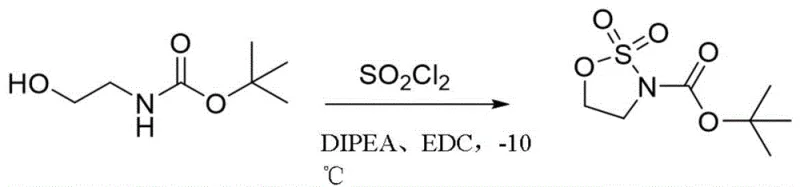 The absence of radical pathways, which are common in metal-catalyzed oxidations, further reduces the risk of racemization or skeletal rearrangement. For quality control teams, this means a simpler impurity profile that is easier to characterize and control, ensuring consistent batch-to-batch quality essential for regulatory compliance in pharmaceutical applications.
The absence of radical pathways, which are common in metal-catalyzed oxidations, further reduces the risk of racemization or skeletal rearrangement. For quality control teams, this means a simpler impurity profile that is easier to characterize and control, ensuring consistent batch-to-batch quality essential for regulatory compliance in pharmaceutical applications.
How to Synthesize Tert-Butyl 1,2,3-Oxathiazolidine-3-Carboxylate 2,2-Dioxide Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and safety, although the procedure is fundamentally straightforward. The process begins with the preparation of a reaction mixture containing the N-Boc amino alcohol substrate and an organic base dissolved in a suitable solvent system. The patent highlights that a mixed solvent system comprising N,N-diisopropylethylamine (DIPEA) and dichloroethane (DCE) in a specific volume ratio provides optimal results, promoting solubility and reaction kinetics effectively. Once the solution is cooled to the prescribed low temperature range, typically between -20°C and -5°C, the sulfuryl chloride is added dropwise. This controlled addition is vital to manage the exothermic nature of the reaction and prevent local overheating which could lead to side reactions. The reaction is then stirred for a short duration, usually 0.5 to 3 hours, under an inert atmosphere to ensure complete conversion.
- Dissolve the N-Boc protected amino alcohol precursor in a mixed solvent system of DIPEA and dichloroethane under inert atmosphere.
- Slowly add sulfuryl chloride (SO2Cl2) dropwise to the reaction mixture while maintaining a low temperature between -20°C and -5°C.
- Quench the reaction with water after completion, extract with ethyl acetate, and purify via silica gel chromatography to isolate the target cyclic sulfonamide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-step synthesis method offers transformative benefits that extend far beyond simple chemical efficiency. The most immediate impact is seen in the drastic simplification of the Bill of Materials (BOM). By eliminating the requirement for ruthenium catalysts and stoichiometric oxidants like sodium periodate, the process removes dependency on volatile precious metal markets and hazardous chemical supplies. This shift significantly de-risks the supply chain, ensuring greater continuity of supply even during periods of global raw material scarcity. Moreover, the reduction in raw material variety simplifies inventory management and reduces the administrative burden associated with sourcing and qualifying multiple specialized reagents. The use of commodity chemicals like sulfuryl chloride and common solvents ensures that the production process is grounded in a stable and widely available supply base, fostering long-term planning security for large-scale manufacturing campaigns.
Cost reduction in pharmaceutical intermediate manufacturing is achieved through multiple vectors in this new protocol. Firstly, the consolidation of two reaction steps into one inherently reduces labor costs, utility consumption, and equipment occupancy time. There is no longer a need for intermediate isolation, drying, and re-dissolution, which are resource-intensive operations. Secondly, the removal of the ruthenium catalyst eliminates the substantial cost associated with purchasing the metal and, crucially, the downstream processing required to remove trace metal residues to ppm levels. Heavy metal scavenging resins and additional purification steps represent a hidden but significant cost center in traditional routes; their elimination here leads to substantial cost savings. Finally, the improved yield and reduced byproduct formation mean that less starting material is wasted, improving the overall atom economy and further driving down the cost per kilogram of the final API intermediate. These efficiencies collectively enhance the competitiveness of the final drug product in the marketplace.
Scalability and environmental compliance are also markedly improved, addressing key concerns for modern chemical manufacturing. The one-step process generates significantly less chemical waste, particularly avoiding the heavy metal waste streams associated with ruthenium catalysts which require special disposal protocols. This aligns with increasingly stringent environmental regulations and corporate sustainability goals. The reaction conditions are mild and do not require extreme temperatures or pressures, making the process safer and easier to scale from pilot plant to multi-ton commercial production. The simplicity of the workup procedure, involving a standard aqueous quench and extraction, facilitates rapid throughput in manufacturing facilities. For supply chain directors, this translates to shorter lead times for high-purity pharmaceutical intermediates and the ability to respond more agilely to market demand fluctuations. The robustness of the method ensures that scale-up does not introduce new unforeseen complexities, providing a reliable pathway for commercializing new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology, derived from the detailed experimental data and comparative analysis provided in the patent documentation. Understanding these aspects is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing workflows. The answers reflect the specific advantages of the SO2Cl2 mediated cyclization over traditional methods, focusing on yield, purity, and operational simplicity. This information is intended to assist decision-makers in assessing the potential impact on their specific projects and supply chain strategies.
Q: What are the primary advantages of this synthesis method over conventional routes?
A: This method eliminates the need for expensive and toxic ruthenium catalysts and oxidants like sodium periodate, reducing the process from two steps to a single efficient cyclization step.
Q: What is the typical yield range for this one-step cyclization?
A: Experimental data indicates robust yields ranging from 75% to 82% across various substituted derivatives, demonstrating high efficiency and reproducibility.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the use of common solvents like dichloroethane and DIPEA, combined with mild reaction temperatures and simple workup procedures, makes it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tert-Butyl 1,2,3-Oxathiazolidine-3-Carboxylate 2,2-Dioxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and optimizing production costs. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN111303078A and is fully equipped to leverage this one-step cyclization strategy for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are designed to handle sensitive reactions under strict temperature control and inert atmospheres, guaranteeing the high quality and consistency required for pharmaceutical applications. With our rigorous QC labs and commitment to stringent purity specifications, we deliver intermediates that meet the highest global regulatory standards, minimizing your risk during the drug approval process.
We invite you to collaborate with us to unlock the full potential of this innovative synthesis route for your specific needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project volume and timeline, demonstrating exactly how this technology can improve your bottom line. We encourage you to contact us to request specific COA data for our catalog of oxathiazolidine dioxide derivatives or to discuss custom synthesis options. Let us partner with you to streamline your supply chain, reduce manufacturing costs, and bring your life-saving therapies to market faster. Reach out today for detailed route feasibility assessments and discover how NINGBO INNO PHARMCHEM can be your strategic partner in chemical innovation.
