Advanced Chiral Ferrocene-Indole Bisphosphine Ligands for High-Efficiency Asymmetric Catalysis
Advanced Chiral Ferrocene-Indole Bisphosphine Ligands for High-Efficiency Asymmetric Catalysis
The landscape of asymmetric synthesis is continuously evolving, driven by the demand for higher efficiency and selectivity in the production of chiral pharmaceutical intermediates. Patent CN112824422B introduces a significant breakthrough with the development of a novel chiral ferrocene-indole bisphosphine ligand. This technology addresses the critical need for robust catalysts capable of operating under mild conditions while delivering exceptional enantioselectivity. Unlike traditional C2-symmetric ligands, this new class leverages an asymmetric ferrocene-indole skeleton, optimizing the chiral environment around the central metal atom. For R&D directors and process chemists, this represents a pivotal shift towards more versatile catalytic systems that can handle a broader substrate scope, including challenging C=C, C=O, and C=N bond reductions. The patent details a streamlined preparation method that enhances the feasibility of large-scale adoption, positioning this ligand as a key enabler for next-generation drug manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of asymmetric hydrogenation has been dominated by C2-symmetric ligands such as BINAP and DuPhos. While these structures offer the advantage of simplified synthesis and reduced transition state complexity, they often lack the fine-tuned steric and electronic properties required for maximizing catalytic induction in specific transformations. The rigid symmetry can sometimes limit the optimizer's ability to create the ideal chiral pocket around the metal center, leading to suboptimal enantioselectivity or requiring excessive catalyst loading. Furthermore, the synthesis of highly specialized symmetric ligands can involve multi-step sequences with expensive chiral precursors, driving up the cost of goods for the final active pharmaceutical ingredient. These limitations necessitate a move towards asymmetric designs that offer greater flexibility in tuning the catalyst's performance without compromising on stability or ease of access.
The Novel Approach
The technology disclosed in CN112824422B overcomes these hurdles by introducing a non-symmetric bisphosphine ligand based on a ferrocene-indole framework. This design combines the proven planar chirality of ferrocene with the versatile reactivity of the indole moiety. The synthesis strategy is remarkably efficient, starting from readily available chiral ferrocenyl phosphine acetates and indole derivatives. As illustrated in the reaction scheme below, the process involves a direct coupling followed by a selective phosphination step, avoiding the need for complex protecting group strategies or harsh reaction conditions. This novel approach not only simplifies the supply chain for the ligand itself but also results in a catalyst precursor that exhibits superior stability and activity. The resulting ligands can be easily modified by varying the substituents on the indole nitrogen or the phosphine aryl groups, allowing for rapid optimization for specific client applications.
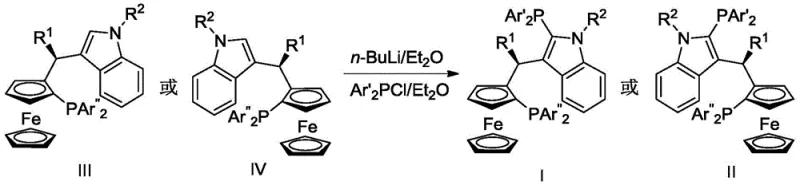
Mechanistic Insights into Ferrocene-Indole Bisphosphine Ligand Synthesis
The core innovation lies in the precise construction of the chiral backbone and the introduction of the second phosphine group. The synthesis begins with a Friedel-Crafts-type alkylation where the chiral ferrocenyl phosphine acetate reacts with the indole derivative in toluene at temperatures between 80-110°C. This step establishes the critical carbon-carbon bond between the ferrocene unit and the indole ring, preserving the stereochemical integrity of the ferrocene plane. Following the isolation of the intermediate, the key mechanistic step involves regioselective lithiation. By treating the intermediate with n-BuLi at room temperature, a lithiated species is generated specifically at the C2 position of the indole ring. This reactive intermediate is then quenched with a chlorophosphine reagent, such as ClPPh2, to install the second phosphorus donor atom. This sequence ensures that the two phosphine groups are positioned optimally to chelate the metal center, creating a rigid yet tunable coordination sphere that is essential for high enantioselectivity.
From an impurity control perspective, this mechanism offers distinct advantages. The use of mild conditions for the lithiation step minimizes side reactions such as over-lithiation or decomposition of the sensitive ferrocene moiety. The patent specifies that the reaction can be conducted in ether solvents, which facilitates easy workup and purification. The resulting ligands demonstrate remarkable stability, resisting oxidation and degradation during storage and handling. This robustness is critical for industrial applications where catalyst consistency is paramount. Furthermore, the modular nature of the synthesis allows for the introduction of diverse functional groups on the phosphine aryl rings or the indole nitrogen without altering the core reaction pathway. This flexibility enables the fine-tuning of electronic properties to match specific substrate requirements, ensuring that the catalyst maintains high turnover numbers even with sterically demanding substrates.
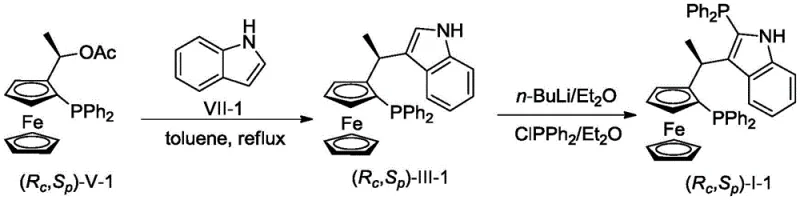
How to Synthesize Chiral Ferrocene-Indole Bisphosphine Ligand Efficiently
The preparation of these high-performance ligands follows a logical and scalable two-stage protocol that is well-suited for both laboratory optimization and commercial manufacturing. The process begins with the formation of the chiral ferrocene-indole intermediate, followed by the installation of the second phosphine group via organolithium chemistry. Detailed operational parameters, including specific molar ratios and solvent choices, are critical to achieving the reported high yields and purity levels. The following guide outlines the standardized steps derived from the patent examples, providing a clear roadmap for technical teams looking to implement this technology. For a complete breakdown of the standardized synthesis steps, please refer to the detailed guide section below.
- React chiral ferrocenyl phosphine acetate with indole derivatives in toluene at 80-110°C to form the ferrocene-indole intermediate.
- Dissolve the intermediate in ether and treat with n-BuLi at room temperature to generate the lithiated species.
- Add chlorodiphenylphosphine (ClPPh2) to the reaction mixture, stir, and purify via column chromatography to isolate the target bisphosphine ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this new ligand technology translates into tangible strategic benefits beyond mere technical performance. The synthesis route relies on commodity chemicals and established chiral building blocks, significantly reducing the risk of raw material shortages. By eliminating the need for exotic reagents or complex multi-step sequences often associated with bespoke ligand synthesis, the overall cost of goods for the catalyst system is drastically lowered. This cost efficiency is passed down the value chain, enabling more competitive pricing for the final chiral intermediates. Moreover, the robustness of the ligand reduces the frequency of catalyst replacement and minimizes waste generation, aligning with modern sustainability goals and reducing the environmental footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The streamlined synthetic pathway eliminates several purification and protection steps typically required for similar chiral ligands. By utilizing a direct coupling strategy and avoiding expensive transition metal catalysts in the ligand synthesis itself, the production costs are significantly optimized. This efficiency allows for the economic production of high-value chiral intermediates, making previously marginal processes commercially viable. The high turnover number (TON) of up to 1,000,000 means that less precious metal is required per kilogram of product, further driving down the cost of heavy metal inputs and downstream removal processes.
- Enhanced Supply Chain Reliability: The starting materials, such as chiral ferrocenyl phosphine acetates and substituted indoles, are sourced from stable supply chains with multiple global vendors. This diversification mitigates the risk of single-source dependency that often plagues specialized fine chemical manufacturing. The simplicity of the reaction conditions, which do not require cryogenic temperatures or ultra-high pressures, allows for production in standard multipurpose reactors. This flexibility ensures consistent supply continuity even during periods of high market demand or logistical disruptions, securing the production schedule for downstream API manufacturers.
- Scalability and Environmental Compliance: The process operates in common organic solvents like toluene and diethyl ether, which are easily recovered and recycled in standard distillation units. The absence of toxic heavy metals in the ligand synthesis step simplifies waste treatment and regulatory compliance. The high atom economy of the reaction minimizes the generation of hazardous byproducts, supporting greener manufacturing initiatives. Scalability is inherent in the design, as the exothermic profiles are manageable and the reaction times are short, facilitating a smooth transition from pilot plant to full-scale commercial production without the need for specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ligand technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation. They serve to clarify the operational scope and potential advantages for partners considering this technology for their process development pipelines. Understanding these details is crucial for making informed decisions about catalyst selection and process integration.
Q: What are the catalytic performance metrics of this new ligand?
A: The ligand forms complexes with Rh, Ru, and Ir that exhibit exceptional catalytic activity, with Turnover Numbers (TON) reaching up to 1,000,000 and enantioselectivity exceeding 98% ee in various asymmetric hydrogenation reactions.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the preparation method utilizes readily available raw materials and avoids extreme conditions, employing standard solvents like toluene and ether, which facilitates straightforward scale-up from laboratory to commercial manufacturing.
Q: What types of chemical bonds can this ligand reduce?
A: This versatile ligand is effective for the asymmetric hydrogenation of C=C, C=O, and C=N bonds, making it suitable for synthesizing chiral acids, amino acids, alcohols, and amines used in pharmaceutical and agrochemical industries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Ferrocene-Indole Bisphosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chiral ferrocene-indole bisphosphine ligand technology in advancing asymmetric synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to market is seamless. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of ligand or intermediate meets the highest industry standards. We understand that consistency is key in catalytic processes, and our robust quality management systems are designed to deliver the reliability your supply chain demands.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific applications. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your current manufacturing challenges. Please contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to not just a product, but a comprehensive solution that optimizes both performance and profitability in your chiral synthesis operations.
