Scalable Synthesis of High-Performance Bis-benzophenanthrene Thiophene Discotic Liquid Crystals
Scalable Synthesis of High-Performance Bis-benzophenanthrene Thiophene Discotic Liquid Crystals
The rapid advancement of organic electronics demands materials that combine high charge carrier mobility with excellent processability. Patent CN112409325A introduces a sophisticated class of bis-benzophenanthrene thiophene discotic liquid crystal compounds, specifically designed to self-assemble into stable hexagonal columnar phases. These materials, represented by general formula (I) and their precursors in general formula (II), offer a breakthrough in the development of one-dimensional charge carrier systems. The patent details a robust synthetic pathway that leverages palladium-catalyzed cross-coupling and iron-mediated oxidative cyclization to construct these complex polycyclic aromatic hydrocarbons. For R&D directors and procurement specialists in the electronic chemical sector, this technology represents a significant opportunity to access high-purity organic semiconductors suitable for next-generation displays and photovoltaics.
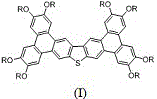
The core innovation lies in the molecular architecture, where a central thiophene-fused core is peripherally substituted with long-chain alkoxy groups ranging from C8 to C16. This structural design is not merely aesthetic; it is functional engineering at the molecular level. The extended pi-conjugation system facilitates effective orbital overlap, enhancing intermolecular interactions and carrier migration rates. Simultaneously, the flexible alkoxy chains induce liquid crystallinity, allowing the molecules to organize into ordered columns that act as molecular wires. This dual functionality addresses the critical bottleneck in organic electronics: balancing electronic performance with the ability to process the material from solution. The patent explicitly demonstrates that these compounds can form gels in various organic solvents, a property that opens new avenues for solution-processing techniques in device manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of large polycyclic aromatic hydrocarbons often relies on harsh conditions that can degrade sensitive functional groups or lead to uncontrolled polymerization. Conventional methods for creating discotic liquid crystals frequently struggle with regioselectivity, resulting in mixtures of isomers that are difficult to separate and detrimental to electronic performance. Furthermore, many existing routes require expensive catalysts or multi-step sequences with low overall yields, making commercial scale-up economically unviable. The lack of solubility in common organic solvents is another persistent issue, often necessitating high-temperature processing that limits substrate compatibility. Without a modular approach, tuning the physical properties of the final material, such as melting point or phase transition temperature, requires redesigning the entire synthetic route from scratch.
The Novel Approach
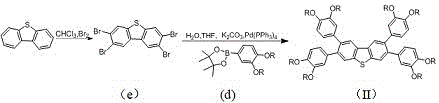
The methodology outlined in CN112409325A overcomes these hurdles through a modular, convergent synthesis strategy. By utilizing Suzuki-Miyaura coupling, the process allows for the precise assembly of the biphenyl units onto the dibenzothiophene core under relatively mild conditions. This ensures high regioselectivity and minimizes the formation of unwanted byproducts. The subsequent intramolecular oxidative cyclization using ferric chloride is a powerful tool for extending conjugation without requiring extreme temperatures or pressures. This two-stage approach—building the skeleton first, then fusing the rings—provides exceptional control over the molecular structure. It enables the facile variation of the peripheral alkyl chains, allowing manufacturers to customize the material's solubility and self-assembly behavior without altering the core electronic properties.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Cyclization
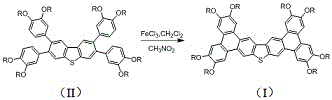
The transformation of the precursor compound (II) into the final fused product (I) is a critical step that defines the electronic character of the material. This intramolecular oxidative cyclization is mediated by ferric chloride (FeCl3) in a mixed solvent system of dichloromethane and nitromethane. The mechanism involves the generation of radical cations on the aromatic rings, which subsequently undergo coupling to form new carbon-carbon bonds, effectively fusing the phenyl rings to the central thiophene core. The use of nitromethane is particularly strategic; as a polar solvent, it helps solubilize the polar intermediates and the iron salts, ensuring a homogeneous reaction environment that promotes efficient cyclization. This step converts the flexible biphenyl linkages into a rigid, planar polycyclic system, which is essential for the formation of the columnar liquid crystal phase.
Impurity control is paramount in electronic materials, and this oxidative step is designed to minimize defect formation. The reaction conditions are carefully monitored, with TLC tracking every 15 minutes to prevent over-oxidation or degradation of the sensitive thiophene moiety. Quenching with methanol effectively terminates the reaction and reduces residual iron species, facilitating downstream purification. The resulting product exhibits a highly ordered structure, as evidenced by its ability to form stable gels and distinct liquid crystalline textures. This level of structural integrity is crucial for achieving the high charge carrier mobilities required in organic field-effect transistors and solar cells. The process demonstrates how careful selection of oxidants and solvents can drive complex molecular rearrangements with high fidelity.
How to Synthesize Bis-benzophenanthrene Thiophene Efficiently
The synthesis of these advanced electronic materials follows a logical progression from simple commodity chemicals to complex functional architectures. The process begins with the preparation of alkoxy-substituted phenyl boronic esters, which serve as the building blocks for the peripheral arms. These are then coupled to a tetrabrominated dibenzothiophene core using palladium catalysis. The final step involves the oxidative ring closure to lock the structure into its planar, conjugated form. This route is designed for reproducibility and scalability, utilizing standard organic synthesis techniques that are well-understood in industrial settings. For detailed operational parameters and safety considerations, please refer to the standardized guide below.
- Preparation of alkoxy-substituted phenyl boronic esters via bromination and borylation.
- Suzuki coupling of the boronic ester with tetrabromodibenzothiophene to form the precursor.
- Intramolecular oxidative cyclization using FeCl3 to close the rings and form the final discotic liquid crystal.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits in terms of cost efficiency and supply reliability. The reliance on Suzuki coupling means that the process utilizes widely available catalysts and reagents, reducing dependency on exotic or single-source materials. The modular nature of the synthesis allows for bulk purchasing of common intermediates like dibenzothiophene and bromoalkanes, leveraging economies of scale to drive down raw material costs. Furthermore, the high yields reported in the patent examples suggest a material-efficient process that minimizes waste generation, aligning with modern green chemistry principles and reducing disposal costs.
- Cost Reduction in Manufacturing: The elimination of complex, multi-step protection and deprotection sequences significantly streamlines the production workflow. By using direct oxidative cyclization with inexpensive iron salts instead of precious metal catalysts for the ring-closing step, the process achieves substantial cost savings. The ability to purify intermediates via standard silica gel chromatography and recrystallization avoids the need for specialized preparative HPLC, further lowering capital and operational expenditures associated with purification infrastructure.
- Enhanced Supply Chain Reliability: The starting materials, including 5-bromosalicylaldehyde and various long-chain bromoalkanes, are commodity chemicals with established global supply chains. This reduces the risk of supply disruptions compared to routes relying on bespoke, hard-to-source precursors. The robustness of the Suzuki coupling reaction also means that the process is less sensitive to minor variations in reagent quality, ensuring consistent batch-to-batch production even when sourcing from multiple suppliers.
- Scalability and Environmental Compliance: The reaction conditions, primarily occurring in common solvents like THF, dichloromethane, and dioxane, are compatible with existing large-scale reactor setups. The process avoids the use of highly toxic reagents or extreme high-pressure conditions, simplifying safety compliance and facility requirements. Additionally, the formation of gel phases suggests that the material can be processed from solution, potentially enabling roll-to-roll manufacturing techniques that are far more cost-effective than vacuum deposition methods for large-area electronics.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for integrating these materials into your product pipeline. The following questions address common concerns regarding the scalability, purity, and application of these discotic liquid crystals. The answers are derived directly from the technical disclosures in the patent, providing a reliable foundation for your feasibility assessments.
Q: What are the primary applications of bis-benzophenanthrene thiophene compounds?
A: These compounds function as discotic liquid crystals capable of self-assembling into hexagonal columnar phases, making them ideal for organic semiconductors, field-effect transistors, and photovoltaic devices due to their efficient charge carrier mobility.
Q: How does the synthetic route ensure high purity for electronic grade materials?
A: The route utilizes Suzuki coupling for precise carbon-carbon bond formation followed by a controlled oxidative cyclization, minimizing regioisomers and allowing for rigorous purification via column chromatography and recrystallization.
Q: Can the alkyl chain length be modified for specific processing requirements?
A: Yes, the synthetic protocol allows for the variation of the alkoxy side chains (C8 to C16), enabling manufacturers to tune the solubility, gelation properties, and liquid crystalline phase transition temperatures to suit specific device fabrication processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis-benzophenanthrene Thiophene Supplier
As the demand for high-performance organic semiconductors grows, having a partner with deep technical expertise is essential. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. Our stringent purity specifications and rigorous QC labs guarantee that every batch of bis-benzophenanthrene thiophene meets the exacting standards required for electronic applications. We understand that consistency is key in the electronics industry, and our optimized processes are designed to deliver uniform material properties batch after batch.
We invite you to collaborate with us to optimize your supply chain for next-generation display and energy materials. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and purity requirements. Contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate the commercialization of your organic electronic devices.
