Advanced Gold-Catalyzed Synthesis of Z-Type Halogenated Chalcones for Commercial Scale-Up
Advanced Gold-Catalyzed Synthesis of Z-Type Halogenated Chalcones for Commercial Scale-Up
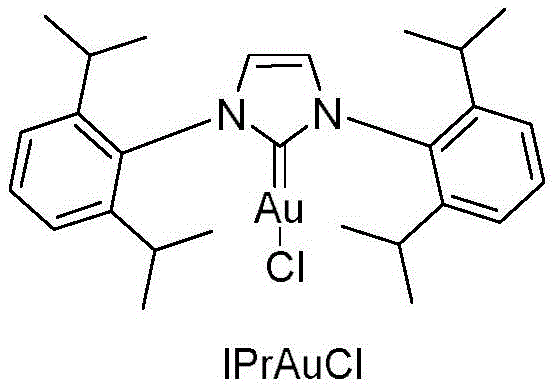
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex molecular scaffolds, particularly those with specific stereochemical requirements. A significant breakthrough in this domain is detailed in patent CN108863751B, which discloses a novel synthetic method for Z-type halogenated chalcone compounds. These compounds serve as critical precursors for flavonoids and possess diverse biological activities, including anti-inflammatory and anticancer properties. The patented technology utilizes a sophisticated gold-catalyzed rearrangement strategy that operates under remarkably mild conditions, specifically at room temperature, offering a stark contrast to energy-intensive traditional processes. By employing IPrAuCl as a catalyst and AgSbF6 as an additive in 1,2-dichloroethane, the method achieves rapid conversion rates, often completing within minutes. This innovation represents a paradigm shift for manufacturers aiming to produce high-purity intermediates with precise regioselectivity.
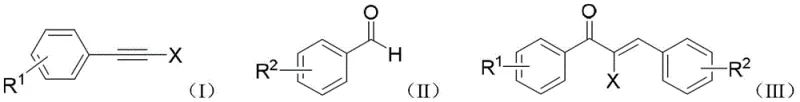
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of halogenated chalcones has been plagued by inefficiencies and environmental hazards that burden both R&D budgets and supply chain logistics. Traditional routes typically involve a multi-step sequence starting with a Claisen-Schmidt condensation between arylethanone and an aldehyde under alkaline conditions to form the chalcone backbone. Subsequently, hazardous halogenating reagents such as liquid bromine or chlorine gas are employed to add across the alpha-beta unsaturated double bonds, creating alpha-beta dihalo ketones. The final step requires an elimination reaction under alkaline conditions to regenerate the double bond, often resulting in a mixture of stereoisomers. This conventional approach suffers from poor regioselectivity, frequently yielding the thermodynamically more stable E-type (trans) products alongside the desired Z-type isomers, necessitating difficult and costly separation processes. Furthermore, the use of corrosive and toxic reagents generates substantial hazardous waste, complicating disposal and increasing the overall environmental compliance costs for manufacturing facilities.
The Novel Approach
In sharp contrast, the method disclosed in CN108863751B streamlines the entire synthesis into a single, elegant step that bypasses the need for pre-functionalized ketones or harsh halogenation conditions. By directly reacting a haloalkyne (Formula I) with an aromatic aldehyde (Formula II) in the presence of a gold catalyst, the process constructs the Z-type halogenated chalcone (Formula III) with exceptional precision. The reaction proceeds at ambient temperatures ranging from 20°C to 30°C, eliminating the energy costs associated with heating or cooling systems required by older methodologies. Most critically, this gold-catalyzed pathway demonstrates 100% regioselectivity for the Z-isomer, completely suppressing the formation of the unwanted E-type byproducts. This high level of stereocontrol simplifies downstream purification, reduces raw material waste, and significantly shortens the overall production timeline, making it an ideal candidate for cost reduction in fine chemical manufacturing.
Mechanistic Insights into IPrAuCl-Catalyzed Rearrangement
The success of this synthetic route lies in the unique activation mode provided by the cationic gold species generated in situ. The reaction mechanism initiates with the coordination of the gold catalyst, derived from IPrAuCl and AgSbF6, to the triple bond of the haloalkyne substrate. This coordination activates the alkyne towards nucleophilic attack by the carbonyl oxygen of the aromatic aldehyde. Following this initial attack, the intermediate undergoes a series of intricate isomerizations and rearrangements, passing through a gold carbene epoxy intermediate. This highly reactive species subsequently evolves into a four-membered ring intermediate, during which the gold catalyst is regenerated and released back into the cycle. The final step involves an intramolecular rearrangement of this four-membered ring to yield the cis-halogenated chalcone product. Understanding this mechanistic pathway is crucial for R&D directors, as it highlights the specificity of the catalyst-substrate interaction that drives the exclusive formation of the Z-configuration.
From an impurity control perspective, the mechanism inherently prevents the formation of trans-isomers, which are common contaminants in base-catalyzed eliminations. The rigid geometric constraints imposed by the gold-carbene intermediate and the subsequent four-membered ring transition state dictate the stereochemical outcome of the reaction. This means that the impurity profile of the crude product is significantly cleaner compared to traditional methods, reducing the burden on analytical quality control teams. The ability to predictably generate the Z-isomer without detectable amounts of the E-isomer ensures that the final API intermediate meets stringent purity specifications required by regulatory bodies. This level of control is particularly valuable when scaling up reactions where minor impurities can accumulate and compromise the safety profile of the final drug substance.
How to Synthesize Z-Type Halogenated Chalcone Efficiently
Implementing this gold-catalyzed protocol in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and mixing protocols to maximize yield and selectivity. The process is designed to be operationally simple, utilizing readily available solvents and commercially sourced catalysts. The reaction is typically monitored using thin-layer chromatography (TLC) to determine the precise endpoint, which usually occurs within a very short timeframe of 5 to 30 minutes depending on the electronic nature of the substrates. Post-reaction workup involves a straightforward filtration to remove silver salts, followed by solvent evaporation and column chromatography purification. For a detailed breakdown of the specific operational parameters, reagent quantities, and safety precautions necessary for execution, please refer to the standardized synthesis guide below.
- Prepare the reaction system by dissolving the IPrAuCl catalyst and AgSbF6 additive in 1,2-dichloroethane solvent under stirring conditions.
- Add the aryl aldehyde substrate to the catalyst solution, followed by the slow addition of the haloalkyne reactant at room temperature.
- Monitor reaction progress via TLC until completion (typically 5-30 minutes), then filter, wash, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers tangible benefits that extend beyond mere chemical elegance. The shift from a multi-step, hazardous process to a one-pot, room-temperature reaction fundamentally alters the cost structure and risk profile of producing halogenated chalcones. By eliminating the need for expensive and dangerous halogenating agents like bromine or chlorine gas, the process reduces the requirement for specialized corrosion-resistant equipment and complex waste treatment infrastructure. This simplification translates directly into lower capital expenditure (CAPEX) and operational expenditure (OPEX) for manufacturing partners. Furthermore, the dramatic reduction in reaction time—from hours or days in traditional methods to mere minutes—enhances facility throughput, allowing for faster turnaround times on customer orders and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic viability of this process is bolstered by the high efficiency of the gold catalyst, which is effective at loadings as low as 1 mol%. Although gold is a precious metal, the low loading combined with the potential for catalyst recovery or the use of heterogeneous variants in future iterations ensures that the raw material cost remains competitive. Additionally, the elimination of multiple isolation and purification steps between reaction stages significantly reduces solvent consumption and labor costs. The high yields reported, often exceeding 90%, minimize the loss of valuable starting materials, further driving down the cost per kilogram of the final product. This efficiency makes the process highly attractive for cost reduction in pharmaceutical intermediate manufacturing where margin pressure is constant.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by the mild reaction conditions and the stability of the reagents involved. Unlike processes requiring cryogenic temperatures or high-pressure reactors, this method can be executed in standard glass-lined or stainless steel vessels available in most contract manufacturing organizations (CMOs). The use of stable, commercially available aldehydes and haloalkynes ensures a robust supply of starting materials, reducing the risk of bottlenecks caused by scarce reagents. The short reaction time also means that production slots can be turned over rapidly, increasing the overall capacity of the supply chain to handle large-volume orders without the need for extensive reactor farm expansion. This agility is critical for maintaining continuity of supply in the fast-paced pharmaceutical sector.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but the simplicity of this gold-catalyzed route mitigates many typical scale-up risks. The absence of exothermic hazards associated with strong acid-base neutralizations or halogen additions makes the process safer to operate at the 100 kg to 100 MT scale. From an environmental standpoint, the process aligns perfectly with green chemistry principles by avoiding toxic reagents and minimizing waste generation. This compliance reduces the regulatory burden and disposal costs associated with hazardous waste, making it easier to obtain necessary environmental permits for production facilities. The cleaner process also supports corporate sustainability goals, a factor increasingly weighted in vendor selection by major multinational corporations.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries regarding the patent's scope and practical implementation. These insights address concerns about catalyst availability, substrate compatibility, and the comparative advantages over legacy synthetic routes. Understanding these details is essential for assessing the potential integration of this method into existing production workflows.
Q: What is the primary advantage of this gold-catalyzed method over traditional chalcone synthesis?
A: Unlike traditional multi-step methods requiring harsh halogenating agents and strong bases, this patent describes a one-pot reaction at room temperature that achieves 100% Z-selectivity without generating E-isomers.
Q: Is the IPrAuCl catalyst commercially available for large-scale production?
A: Yes, the catalyst IPrAuCl (CAS 852445-83-1) is commercially available or can be synthesized via literature methods, and it is used in low loadings (0.1-5%) to ensure cost-effectiveness.
Q: How does this process impact environmental compliance in manufacturing?
A: The process eliminates the need for toxic halogenating reagents like liquid bromine or chlorine gas and avoids strong acid/base waste streams, significantly reducing the environmental footprint and disposal costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Z-Type Halogenated Chalcone Supplier
The technological potential of the gold-catalyzed synthesis of Z-type halogenated chalcones is immense, offering a pathway to high-value intermediates with unmatched efficiency and selectivity. At NINGBO INNO PHARMCHEM, we pride ourselves on our ability to translate such cutting-edge academic and patent literature into robust, commercial-scale manufacturing processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical output. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of Z-type halogenated chalcone meets the exacting standards required for pharmaceutical applications.
We invite you to explore how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data for our reference standards and to discuss route feasibility assessments for your target molecules. Let us partner with you to bring this efficient, green, and highly selective chemistry to your commercial portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
