Advanced Sulfuryl Fluoride-Promoted Fluorination for Scalable Pharmaceutical Intermediate Production
Advanced Sulfuryl Fluoride-Promoted Fluorination for Scalable Pharmaceutical Intermediate Production
The landscape of fine chemical synthesis is constantly evolving, driven by the urgent need for more sustainable, cost-effective, and scalable methodologies, particularly in the realm of organofluorine chemistry. Patent CN114105722A introduces a groundbreaking approach to preparing organic fluorine compounds, specifically targeting the efficient conversion of alcohol precursors into valuable fluoride derivatives. This technology leverages sulfuryl fluoride ($SO_2F_2$) gas as a potent promoter in conjunction with standard fluoride sources and organic bases, operating under remarkably mild conditions ranging from 20°C to 80°C. For R&D directors and procurement strategists in the pharmaceutical and agrochemical sectors, this represents a significant paradigm shift away from hazardous or expensive stoichiometric fluorinating agents. The method not only simplifies the operational workflow but also ensures high purity and yield across a diverse range of substrates, making it an ideal candidate for the reliable supply of complex intermediates required in modern drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of organofluorine compounds has been plagued by significant technical and economic hurdles that hinder efficient commercial production. Traditional nucleophilic substitution reactions often require harsh conditions, such as high temperatures and polar aprotic solvents, which can lead to decomposition of sensitive functional groups commonly found in pharmaceutical intermediates. Furthermore, methods relying on transition metal catalysis, while effective for specific transformations, introduce the risk of heavy metal contamination, necessitating costly and time-consuming purification steps to meet stringent regulatory limits for residual metals in active pharmaceutical ingredients (APIs). Additionally, specialized deoxyfluorinating reagents like DAST or Deoxo-Fluor, although powerful, are notoriously unstable, potentially explosive, and prohibitively expensive for large-scale applications, creating a bottleneck for cost reduction in pharmaceutical intermediates manufacturing. These legacy processes often suffer from narrow substrate scope, failing to accommodate the diverse electronic environments present in complex molecular architectures.
The Novel Approach
In stark contrast to these conventional limitations, the novel methodology disclosed in the patent utilizes sulfuryl fluoride gas to efficiently promote the reaction between alcohols and fluorinating reagents, offering a robust and economically viable alternative. This gas-promoted system operates under neutral to mildly basic conditions without the need for transition metal catalysts, thereby inherently eliminating the risk of metal impurities and simplifying the downstream purification process significantly. The use of $SO_2F_2$, a chemically stable and inexpensive commodity gas, drastically reduces the raw material costs compared to proprietary liquid fluorinating agents. As illustrated in the general reaction scheme below, the process accommodates a wide variety of benzyl alcohol derivatives, including those bearing electron-withdrawing groups like nitro, cyano, and ester moieties, achieving excellent yields that demonstrate the versatility of this new synthetic route.
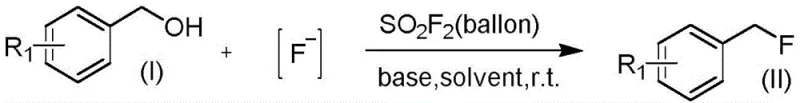
The practical efficacy of this approach is further evidenced by specific examples such as the synthesis of 4-nitrobenzyl fluoride, where the reaction proceeds smoothly to afford the product in high purity and yield. This capability to handle diverse functional groups without compromising efficiency makes the process exceptionally suitable for the commercial scale-up of complex fluorinated intermediates. By replacing hazardous liquids with a manageable gas flow and utilizing common inorganic fluoride salts like potassium fluoride, the method aligns perfectly with green chemistry principles while delivering the high performance required by industrial applications.
Mechanistic Insights into Sulfuryl Fluoride-Promoted Deoxyfluorination
The mechanistic underpinning of this transformation relies on the unique ability of sulfuryl fluoride to activate the hydroxyl group of the alcohol substrate in situ, effectively converting a poor leaving group into a highly reactive intermediate. In the presence of an organic base such as triethylamine or DBU, the $SO_2F_2$ gas likely reacts with the alcohol to form an alkyl fluorosulfonate species, which serves as an excellent electrophile for subsequent nucleophilic attack by the fluoride ion. This activation strategy bypasses the high energy barriers associated with direct displacement of the hydroxyl group, allowing the reaction to proceed at ambient or moderately elevated temperatures. The choice of base plays a critical role in scavenging the acidic byproducts generated during the activation phase, ensuring that the reaction equilibrium is driven towards the formation of the desired carbon-fluorine bond. This mechanistic pathway is distinct from radical or metal-catalyzed processes, offering a cleaner reaction profile that minimizes the formation of side products typically associated with oxidative or reductive conditions.
From an impurity control perspective, the absence of transition metals is a paramount advantage for the production of high-purity organic fluorine compounds intended for therapeutic use. Metal-catalyzed reactions often generate trace amounts of palladium, copper, or nickel that are difficult to remove and can catalyze degradation pathways in the final drug product. By employing a metal-free protocol, this method inherently produces a cleaner crude reaction mixture, reducing the burden on purification teams and lowering the overall cost of goods sold (COGS). Furthermore, the mild reaction conditions prevent the thermal degradation of sensitive substrates, such as those containing aldehyde or ester functionalities, ensuring that the structural integrity of the molecule is preserved throughout the synthesis. This level of control over the reaction environment is essential for maintaining consistent quality attributes in a GMP manufacturing setting.
How to Synthesize Benzyl Fluoride Derivatives Efficiently
To implement this innovative fluorination strategy in a laboratory or pilot plant setting, operators must follow a precise sequence of mixing and gas introduction to maximize safety and yield. The process begins with the dissolution of the alcohol substrate and the fluoride source in a suitable solvent, followed by the addition of the organic base to establish the necessary reaction environment. Detailed standardized synthesis steps see the guide below.
- Mix the alcohol substrate, fluoride source (e.g., KF, NH4F), and organic base in a suitable solvent like acetonitrile or dichloromethane.
- Introduce sulfuryl fluoride (SO2F2) gas into the reaction vessel and maintain the atmosphere while stirring at 20-80°C for 2-8 hours.
- Perform post-treatment by diluting with water, extracting with ethyl acetate, washing, drying, and concentrating to isolate the pure fluorinated product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this sulfuryl fluoride-promoted technology offers transformative benefits that extend far beyond simple reaction efficiency. The shift from expensive, specialized fluorinating reagents to commodity-grade gases and salts fundamentally alters the cost structure of fluorine chemistry, enabling significant cost savings in raw material procurement. Moreover, the simplified workup procedure, which involves standard aqueous extraction and drying, reduces the consumption of solvents and processing time, leading to a more streamlined manufacturing operation. These factors collectively contribute to a more resilient supply chain capable of responding rapidly to market demands for fluorinated building blocks without the bottlenecks associated with sourcing hazardous or controlled reagents.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and expensive proprietary fluorinating agents results in a drastic reduction in direct material costs. By utilizing potassium fluoride or ammonium fluoride alongside sulfuryl fluoride gas, manufacturers can achieve substantial economic advantages compared to traditional methods that rely on precious metals or unstable liquids. The simplified purification process further reduces operational expenditures by minimizing the need for specialized scavenging resins or extensive chromatography, allowing for a more lean and efficient production model that enhances overall profit margins.
- Enhanced Supply Chain Reliability: The reliance on widely available and stable reagents such as $SO_2F_2$ gas and common organic bases ensures a robust and continuous supply of critical inputs. Unlike specialized reagents that may face supply disruptions or long lead times due to limited global production capacity, the components of this reaction are commodity chemicals with established supply chains. This stability mitigates the risk of production delays and allows for better inventory planning, ensuring that the delivery of high-purity organic fluorine compounds remains consistent even in volatile market conditions.
- Scalability and Environmental Compliance: The mild reaction conditions and gas-phase promotion mechanism make this process highly scalable from kilogram to multi-ton quantities without significant re-engineering. The absence of heavy metals simplifies waste treatment protocols, reducing the environmental footprint and ensuring compliance with increasingly stringent regulatory standards for industrial effluent. This scalability, combined with the safety benefits of avoiding explosive liquid reagents, positions the technology as a sustainable solution for the long-term commercial production of fluorinated intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sulfuryl fluoride-based fluorination method. These insights are derived directly from the patent data to provide clarity on the process capabilities and limitations for potential adopters.
Q: What are the advantages of using sulfuryl fluoride gas over traditional fluorinating agents?
A: Sulfuryl fluoride gas is significantly cheaper and more stable than specialized reagents like DAST. It eliminates the need for transition metal catalysts, reducing impurity profiles and downstream purification costs.
Q: Is this fluorination method suitable for large-scale manufacturing?
A: Yes, the process operates under mild temperatures (20-80°C) and uses commodity reagents. The simple workup procedure involving standard extraction makes it highly amenable to commercial scale-up.
Q: What types of substrates are compatible with this SO2F2-promoted reaction?
A: The method demonstrates wide substrate applicability, successfully converting benzyl alcohols with electron-withdrawing groups such as nitro, cyano, ester, and aldehyde functionalities into their corresponding fluorides with high yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Organic Fluorine Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to maintain a competitive edge in the global fine chemicals market. Our team of expert chemists has extensively evaluated the sulfuryl fluoride-promoted fluorination technology and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity organic fluorine compounds that meet the most rigorous quality standards, supported by our state-of-the-art rigorous QC labs and comprehensive analytical capabilities. Our facility is equipped to handle gas-phase reactions safely and efficiently, ensuring that the benefits of this novel patent are fully realized in our commercial offerings.
We invite pharmaceutical and agrochemical companies to collaborate with us to leverage this advanced technology for their specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your target molecules, demonstrating exactly how this method can optimize your budget. We encourage you to contact us today to obtain specific COA data and route feasibility assessments for your next fluorination project, ensuring a seamless transition from development to commercial supply.
