Advanced Ruthenium-Catalyzed Synthesis of Trisubstituted Olefin Tertiary Phosphine Compounds for Industrial Scale-up
The landscape of organophosphorus chemistry is continually evolving, driven by the demand for more efficient and versatile ligands in transition metal catalysis. A significant breakthrough in this domain is documented in Chinese Patent CN112876507B, which introduces a novel and robust methodology for the preparation of trisubstituted olefin tertiary phosphine compounds. These compounds are critical building blocks in the synthesis of advanced catalysts used across the pharmaceutical and agrochemical industries. The patent outlines a transition metal ruthenium-catalyzed hydroarylation strategy that effectively couples 2-diphenylphosphine-biphenyl compounds with internal alkynes. This approach represents a paradigm shift from traditional methods, offering a streamlined pathway to access structurally diverse phosphine ligands that were previously difficult or prohibitively expensive to synthesize. By leveraging this technology, manufacturers can achieve superior control over regioselectivity and stereochemistry, essential factors for developing high-performance catalytic systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of trisubstituted olefin-biaromatic monophosphine compounds was fraught with significant challenges that hindered widespread adoption and commercial viability. The existing literature, such as reports in Organic Letters, primarily relied on monovalent rhodium-catalyzed coupling reactions. While effective in specific contexts, these legacy methods suffered from severe limitations, most notably the requirement for expensive rhodium catalysts which drastically inflated production costs. Furthermore, the substrate scope was inherently restricted, often necessitating the use of symmetrical alkynes as coupling reagents. This limitation severely curtailed the structural diversity of the resulting phosphine ligands, preventing chemists from fine-tuning electronic and steric properties for specialized applications. Additionally, the complexity of the reaction workflows often involved multiple steps or harsh conditions, leading to lower atom economy and increased environmental burden through waste generation.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112876507B offers a concise and highly efficient alternative that directly addresses the shortcomings of rhodium-based systems. By employing a ruthenium catalyst in conjunction with specific ligands and bases, this new process enables the direct hydroarylation of internal alkynes with 2-diphenylphosphine-biphenyl derivatives. The reaction is remarkably versatile, accommodating a wide range of substituents on both the alkyne and the biphenyl backbone, including alkyl, aryl, heteroaryl, and various functional groups like halogens and alkoxy groups. This broad substrate tolerance allows for the rapid generation of diverse chemical libraries, accelerating the discovery of new catalytic activities. Moreover, the reaction proceeds under relatively mild thermal conditions, typically between 80°C and 120°C, eliminating the need for extreme temperatures or pressures that complicate reactor design and operation.

Mechanistic Insights into Ruthenium-Catalyzed Hydroarylation
The core of this technological advancement lies in the sophisticated catalytic cycle mediated by the ruthenium complex. The reaction initiates with the activation of the C-H bond on the biphenyl phosphine substrate by the ruthenium center, a process facilitated by the presence of a carboxylic acid ligand such as phenylglycine or Boc-protected amino acids. This C-H activation step is crucial as it generates a reactive ruthenacycle intermediate that is poised for subsequent alkyne insertion. The internal alkyne then coordinates to the metal center and undergoes migratory insertion into the Ru-C bond. The choice of ligand plays a pivotal role in stabilizing the active catalytic species and directing the regioselectivity of the alkyne insertion, ensuring that the trisubstituted olefin is formed with high fidelity. The presence of a base, such as potassium acetate, assists in the regeneration of the active catalyst by neutralizing acidic byproducts and facilitating the final reductive elimination step that releases the desired trisubstituted olefin tertiary phosphine product.
From an impurity control perspective, the mechanism is inherently clean due to the high specificity of the ruthenium catalyst for the targeted C-H activation site. Unlike non-catalytic methods that might generate numerous side products through radical pathways or uncontrolled electrophilic substitutions, this coordinated process minimizes the formation of regioisomers and oligomeric byproducts. The use of an inert atmosphere, typically argon, further protects the sensitive phosphine moiety from oxidation, preserving the integrity of the P-C bonds throughout the reaction duration. Post-reaction processing involves straightforward techniques such as suction filtration to remove metal residues, followed by reduced pressure distillation and column chromatography. These purification steps are highly effective at removing trace catalyst and unreacted starting materials, ensuring that the final product meets the stringent purity specifications required for use in sensitive cross-coupling reactions like Suzuki-Miyaura or Heck couplings.
How to Synthesize Trisubstituted Olefin Tertiary Phosphine Efficiently
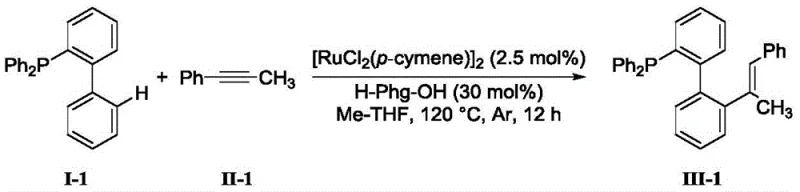
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and reproducibility. The general protocol involves charging a reactor with the 2-diphenylphosphine-biphenyl compound and the internal alkyne in a molar ratio ranging from 1.0:1 to 3.0:1. A ruthenium catalyst, such as dichlorobis(p-cymene)ruthenium(II) dimer, is added at a loading of 0.1 to 10 mol%, along with a suitable amino acid ligand and an inorganic base like potassium acetate. The reaction mixture is suspended in an organic solvent, with methyl tetrahydrofuran (Me-THF) being a preferred choice due to its stability and solvating power. The system is purged with argon to establish an oxygen-free environment before heating to 120°C for approximately 12 hours. Upon completion, the mixture is cooled, filtered, and purified to isolate the target compound. For a visual representation of a specific high-yielding example, refer to the reaction scheme below which demonstrates the synthesis of a methoxy-substituted derivative.
- Mix 2-diphenylphosphine-biphenyl compound, internal alkyne compound, ruthenium catalyst, ligand, base, and organic solvent in a reactor under inert atmosphere.
- Heat the reaction mixture to 80-120°C and maintain constant temperature for approximately 12 hours to ensure complete conversion.
- Cool to room temperature, perform suction filtration, reduced pressure distillation, and column chromatography to isolate the pure trisubstituted olefin tertiary phosphine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ruthenium-catalyzed process translates into tangible strategic benefits that extend beyond simple chemical transformation. The shift from precious rhodium catalysts to more abundant and affordable ruthenium alternatives results in a substantial reduction in raw material costs, directly impacting the bottom line of manufacturing operations. This cost efficiency is compounded by the high atom utilization rate of the reaction, which minimizes waste disposal fees and aligns with increasingly strict environmental regulations. Furthermore, the simplicity of the one-step synthesis eliminates the need for complex multi-stage workflows, thereby reducing labor costs and shortening the overall production cycle time. The robustness of the method also implies fewer batch failures and less downtime for equipment cleaning, enhancing overall operational efficiency.
- Cost Reduction in Manufacturing: The replacement of expensive rhodium catalysts with cost-effective ruthenium systems significantly lowers the direct material costs associated with ligand production. Additionally, the mild reaction conditions reduce energy consumption compared to high-temperature or high-pressure alternatives. The high yields reported, often exceeding 90%, mean that less raw material is wasted, further optimizing the cost per kilogram of the final product. This economic advantage allows for more competitive pricing in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including various substituted biphenyl phosphines and internal alkynes, are readily available from established chemical suppliers. This accessibility mitigates the risk of supply disruptions that can occur with exotic or proprietary reagents. The scalability of the process ensures that production volumes can be ramped up quickly to meet surging demand without the need for specialized or hard-to-source equipment. This reliability is crucial for maintaining continuous production schedules in downstream pharmaceutical manufacturing.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, featuring low equipment requirements and generating minimal three wastes. The absence of strong acids or bases simplifies waste treatment protocols and reduces the environmental footprint of the facility. This compliance with green chemistry principles not only avoids regulatory penalties but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational corporations focused on ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of the technology. Understanding these details is essential for R&D teams evaluating the process for pilot plant studies and for procurement teams assessing the long-term viability of the supply source.
Q: What are the advantages of using Ruthenium over Rhodium for this synthesis?
A: The patented method utilizes Ruthenium catalysts which are significantly more cost-effective than the expensive Rhodium catalysts required in prior art methods. Additionally, the Ruthenium system allows for a broader substrate scope, including unsymmetrical internal alkynes, whereas previous methods were limited to symmetrical alkynes.
Q: What is the typical yield and purity achievable with this process?
A: According to the patent data, the synthesis yields can reach up to 90% or higher depending on the specific substrates used. The process includes standard purification steps like column chromatography to ensure high-purity specifications suitable for catalytic applications.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method is designed for scalability. It operates under mild conditions (80-120°C) without requiring strong acids or bases, has low equipment requirements, and demonstrates high atom utilization with minimal waste generation, making it ideal for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trisubstituted Olefin Tertiary Phosphine Supplier
As the demand for high-performance phosphine ligands continues to grow in the pharmaceutical and fine chemical sectors, partnering with an experienced CDMO is essential for successful commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from laboratory bench to full-scale manufacturing. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of trisubstituted olefin tertiary phosphine meets the highest industry standards, minimizing risks in your downstream catalytic applications. We understand the critical nature of supply continuity and are equipped to handle complex synthetic routes with precision and efficiency.
We invite you to engage with our technical procurement team to discuss how this innovative ruthenium-catalyzed technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and innovation in your organization.
