Revolutionizing Benzazepine Core Synthesis: A Technical Breakthrough for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and efficient pathways to access complex heterocyclic scaffolds that serve as the backbone for life-saving medications. Patent CN107674085B introduces a transformative synthetic methodology for constructing 1,5-epoxy-2,3,4,5-tetrahydro-1H-1-benzazepine derivatives, a privileged structural motif found in numerous bioactive compounds. This innovation addresses long-standing challenges in medicinal chemistry by utilizing a Lewis acid-promoted [3+4] cycloaddition between benzisoxazoles and substituted cyclopropanes. Unlike traditional approaches that often suffer from low atom economy and harsh conditions, this novel route operates under mild parameters with exceptional diastereoselectivity. For R&D directors and process chemists, this represents a significant leap forward in accessing high-purity pharmaceutical intermediates with reduced operational complexity. The ability to construct this seven-membered nitrogen-containing ring system in a single catalytic step not only accelerates lead optimization but also lays a solid foundation for cost-effective commercial manufacturing.
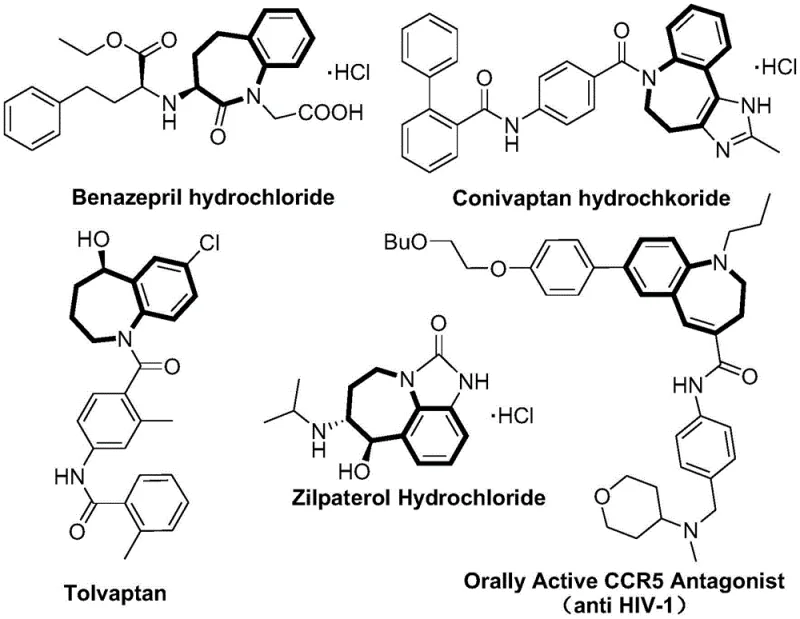
The strategic importance of the 2,3,4,5-tetrahydro-1H-1-benzazepine core cannot be overstated, as it serves as the central pharmacophore in a wide array of clinically approved drugs. As illustrated in the structural diversity of existing medications, this scaffold is integral to angiotensin-converting enzyme inhibitors like Benazepril, vasopressin receptor antagonists such as Conivaptan and Tolvaptan, and bronchodilators like Zilpaterol. Furthermore, ongoing research continues to uncover new therapeutic applications, including potent CCR5 antagonists with anti-HIV activity. The versatility of this chemical architecture drives immense demand for reliable synthetic routes that can deliver these intermediates with high fidelity. By mastering the synthesis of the 1,5-epoxy precursor described in this patent, manufacturers can unlock access to this entire class of high-value therapeutics, positioning themselves as a critical link in the global pharmaceutical supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of epoxy-fused benzazepine systems has been fraught with synthetic difficulties, often relying on convoluted multi-step sequences that hinder scalability. A prominent example of prior art involves the intramolecular cycloaddition of nitrones with alkenes, a strategy reported by Alirio Palma and colleagues in 2010. While scientifically valid, this legacy approach necessitates the pre-functionalization of substrates to install the nitrone moiety, followed by rigorous thermal conditions to drive the cyclization. Such methods typically result in long reaction times, poor stereocontrol, and significant material loss due to side reactions and decomposition. From a process chemistry perspective, the requirement for harsh conditions and the generation of complex impurity profiles make these conventional routes economically unviable for large-scale production. The inability to easily tune the substitution pattern without redesigning the entire synthetic sequence further limits the utility of these older methodologies in modern drug discovery.
The Novel Approach
In stark contrast, the methodology disclosed in CN107674085B offers a streamlined, direct, and highly efficient alternative that fundamentally reshapes the synthetic landscape for these targets. By leveraging the inherent strain energy of donor-acceptor cyclopropanes, the new process enables a direct [3+4] annulation with benzisoxazoles under the promotion of catalytic Lewis acids. This approach eliminates the need for pre-formed nitrones and avoids the high-energy barriers associated with thermal cycloadditions. The reaction proceeds smoothly at room temperature in common aprotic solvents, demonstrating remarkable tolerance for various functional groups on both the cyclopropane and the benzisoxazole components. This operational simplicity translates directly into reduced processing time and lower energy consumption, key metrics for any sustainable manufacturing process. Moreover, the high diastereoselectivity observed ensures that the desired isomer is formed predominantly, minimizing the burden on downstream purification units and maximizing overall throughput.
Mechanistic Insights into Lewis Acid-Catalyzed [3+4] Cycloaddition
The success of this transformation hinges on the precise activation of the cyclopropane ring by the Lewis acid catalyst, such as Scandium Triflate (Sc(OTf)3) or Ytterbium Triflate (Yb(OTf)3). Upon coordination with the electron-withdrawing groups on the cyclopropane, the Lewis acid enhances the electrophilicity of the three-membered ring, facilitating a regioselective ring-opening event. This generates a reactive 1,3-dipole equivalent that subsequently attacks the nucleophilic nitrogen or carbon center of the benzisoxazole, depending on the specific electronic properties of the substrates. The resulting zwitterionic intermediate undergoes a rapid cyclization to forge the new C-N and C-C bonds, establishing the seven-membered benzazepine framework with the embedded 1,5-epoxy bridge. The stereochemical outcome is governed by the spatial arrangement of substituents during this concerted or stepwise cascade, leading to the high diastereomeric ratios reported in the patent examples. Understanding this mechanistic pathway allows chemists to fine-tune reaction parameters, such as catalyst loading and solvent polarity, to optimize yields for specific substrate combinations.
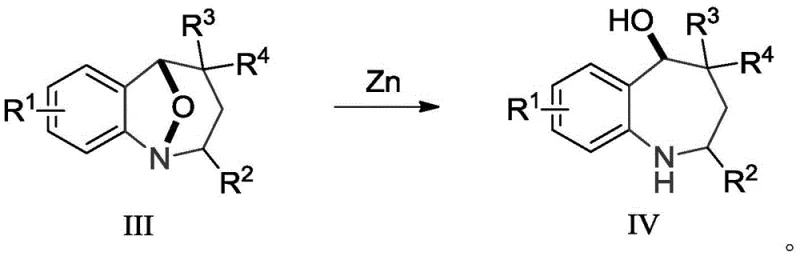
Beyond the initial cyclization, the utility of the resulting 1,5-epoxy-benzazepine lies in its potential for further derivatization, particularly the cleavage of the N-O bond. The patent highlights that the N-O bridge, while stable enough to survive the reaction conditions, can be selectively reduced using metals like Zinc to yield the corresponding 2,3,4,5-tetrahydro-1H-1-benzazepine amines. This reductive step is crucial because it converts the protected epoxy intermediate into the free amine scaffold found in the final active pharmaceutical ingredients. The ability to perform this cleavage cleanly without affecting other sensitive functional groups underscores the robustness of the overall synthetic design. For process developers, this two-stage sequence (cycloaddition followed by reduction) provides a modular platform for generating diverse libraries of benzazepine analogs, facilitating rapid structure-activity relationship (SAR) studies and accelerating the path to clinical candidates.
How to Synthesize 1,5-Epoxy-Benzazepine Efficiently
The experimental protocols outlined in the patent provide a clear roadmap for implementing this chemistry in a laboratory or pilot plant setting. The procedure typically involves suspending activated molecular sieves in an anhydrous solvent like dichloromethane or toluene to maintain strictly dry conditions, which are essential for the stability of the Lewis acid catalyst. Substrates are added sequentially, followed by the catalyst, and the mixture is stirred at ambient temperature. Workup is straightforward, involving filtration to remove the drying agent and catalyst residues, followed by standard chromatographic purification. This simplicity stands in sharp contrast to the cumbersome workups often required for older methods. For detailed operational parameters, stoichiometry, and specific purification techniques tailored to your specific substrate variants, please refer to the standardized synthesis guide below.
- Prepare the reaction vessel with activated molecular sieves and anhydrous aprotic solvent such as dichloromethane or toluene under inert atmosphere.
- Add the substituted cyclopropane donor and benzisoxazole acceptor to the mixture, followed by the addition of a catalytic amount of Lewis acid like Scandium Triflate.
- Stir the reaction at room temperature until completion, filter off the molecular sieves, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route offers compelling economic and logistical benefits that extend far beyond the laboratory bench. The shift from multi-step thermal processes to a single-step catalytic cycle dramatically reduces the number of unit operations required, which directly correlates to lower capital expenditure and reduced manufacturing lead times. By eliminating the need for specialized high-temperature reactors and complex substrate pre-functionalization, facilities can leverage existing infrastructure to produce these high-value intermediates. This flexibility enhances supply chain resilience, allowing manufacturers to respond more agilely to fluctuations in market demand without the bottleneck of lengthy campaign schedules. Furthermore, the use of earth-abundant or recoverable Lewis acid catalysts minimizes reliance on expensive precious metals, contributing to a more stable and predictable cost structure for raw materials.
- Cost Reduction in Manufacturing: The implementation of this catalytic methodology drives substantial cost savings by significantly improving atom economy and reducing waste generation. Traditional routes often involve stoichiometric reagents and generate large volumes of saline or organic waste during extensive workup procedures, whereas this new method utilizes catalytic amounts of promoter and simple filtration for purification. The elimination of multiple isolation steps between intermediates reduces solvent consumption and labor costs, while the high yields reported ensure that raw material inputs are converted efficiently into saleable product. Additionally, the mild reaction conditions lower energy utility costs, as there is no need for prolonged heating or cryogenic cooling, making the process inherently more economical on a metric ton scale.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit greatly from the use of readily available starting materials such as substituted benzisoxazoles and cyclopropanes, which are commodity chemicals produced by a broad base of global suppliers. This diversification of the supply base mitigates the risk of shortages that often plague proprietary or highly specialized reagents required by older synthetic paths. The robustness of the reaction also means that production campaigns are less susceptible to batch failures caused by sensitive reaction parameters, ensuring a consistent and reliable flow of intermediates to downstream API manufacturers. This stability is critical for maintaining uninterrupted production schedules for essential medicines, thereby strengthening the partnership between intermediate suppliers and pharmaceutical clients.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns perfectly with modern green chemistry principles, facilitating easier regulatory approval and permitting. The absence of hazardous reagents and the operation at ambient temperature reduce the facility's safety footprint, lowering insurance premiums and compliance overheads. The simplified waste stream, characterized primarily by spent molecular sieves and common organic solvents, is easier to treat and dispose of compared to the heavy metal or toxic byproduct-laden wastes of conventional methods. This environmental compatibility not only supports corporate sustainability goals but also future-proofs the manufacturing asset against tightening global environmental regulations, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their existing portfolios, we have compiled answers to common inquiries regarding the patent's scope and practical application. These insights address critical concerns about scalability, substrate compatibility, and downstream processing, providing a clear picture of what to expect during technology transfer. Understanding these nuances is essential for making informed decisions about process adoption and resource allocation. We encourage stakeholders to review these points carefully as they assess the potential impact on their specific project timelines and quality targets.
Q: What are the primary advantages of this Lewis acid-catalyzed method over traditional nitrone cycloadditions?
A: The primary advantages include significantly milder reaction conditions (room temperature vs. harsh thermal conditions), a drastically reduced number of synthetic steps, and superior diastereoselectivity, which simplifies downstream purification and improves overall yield.
Q: Can the N-O bridge in the resulting benzazepine derivative be easily removed for further functionalization?
A: Yes, the patent explicitly demonstrates that the N-O bond in the 1,5-epoxy structure can be efficiently cleaved via reduction reactions, such as using Zinc, to generate valuable 2,3,4,5-tetrahydro-1H-1-benzazepine scaffolds found in various active pharmaceutical ingredients.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Absolutely. The use of catalytic amounts of commercially available Lewis acids, combined with simple workup procedures like filtration and standard chromatography, makes this process highly scalable and cost-effective for industrial manufacturing compared to multi-step legacy routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Epoxy-Benzazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a promising patent to a commercial reality requires more than just chemical knowledge; it demands deep engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies demonstrated in the lab are faithfully reproduced on an industrial scale. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 1,5-epoxy-2,3,4,5-tetrahydro-1H-1-benzazepine derivative meets the exacting standards required for pharmaceutical applications. Our state-of-the-art facilities are equipped to handle the specific solvent and catalyst requirements of this Lewis acid-catalyzed process, delivering a product that is ready for immediate downstream conversion into active pharmaceutical ingredients.
We invite you to collaborate with us to leverage this cutting-edge synthetic technology for your next drug development program. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this route for your target molecule. We encourage you to reach out today to request specific COA data and route feasibility assessments tailored to your unique chemical requirements. Let us help you optimize your supply chain and accelerate your time to market with a reliable, cost-effective, and scalable solution for benzazepine core synthesis.
