Scalable Synthesis of Chiral Aryl Oxime Ethers via Asymmetric Copper Catalysis for Pharmaceutical Applications
Scalable Synthesis of Chiral Aryl Oxime Ethers via Asymmetric Copper Catalysis for Pharmaceutical Applications
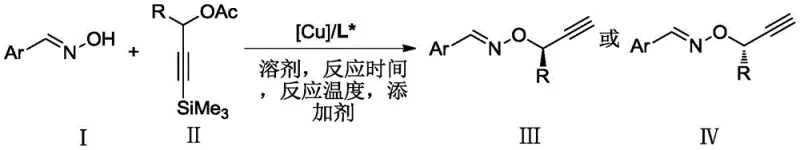
The landscape of asymmetric synthesis is continually evolving, driven by the demand for high-purity chiral intermediates essential for modern drug discovery. Patent CN112279779B introduces a significant advancement in this field by detailing a robust preparation method for chiral aryl oxime ether compounds. This technology leverages a chiral copper catalyst to facilitate the asymmetric oxime etherification reaction between aryl oximes and propargyl compounds. Unlike traditional methods that often struggle with oxygen nucleophiles, this invention achieves high enantioselectivity under remarkably mild conditions, specifically at temperatures as low as -20°C using common solvents like methanol or ethanol. For R&D directors and process chemists, this represents a pivotal shift towards more efficient and stereoselective pathways for constructing complex molecular architectures found in bioactive molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, copper-catalyzed propargyl substitution has been extensively explored, particularly since the seminal reports by van Maarseveen and Nishibayashi in 2008 regarding asymmetric propargyl amination. While nitrogen and carbon nucleophiles have been widely utilized in these transformations, the incorporation of oxygen nucleophiles has remained a significant challenge. Prior art, such as the work by the Nishibayashi group in 2015 using Cu/pybox catalysts, successfully demonstrated asymmetric propargyl etherification but was largely restricted to aliphatic propargyl alcohol esters reacting with simple alcohols or phenols. Furthermore, these conventional protocols often suffered from prolonged reaction times and limited substrate scope, failing to accommodate the structural diversity required for complex pharmaceutical intermediate synthesis. The inability to effectively utilize oximes as oxygen nucleophiles in these catalytic cycles left a critical gap in the synthetic toolbox for accessing chiral oxime ethers, which are valuable motifs in medicinal chemistry.
The Novel Approach
The methodology disclosed in CN112279779B overcomes these historical barriers by employing a metal complex generated in situ from a copper metal precursor and a chiral P,N,N-ligand. This novel catalytic system enables the direct asymmetric oxime etherification of aryl oximes with propargyl compounds, a transformation that was previously unachieved with high stereocontrol. The reaction operates efficiently at -20°C, significantly reducing energy consumption compared to high-temperature alternatives, and utilizes readily available solvents like methanol. By expanding the nucleophile scope to include aryl oximes, this approach unlocks access to a new series of chiral aryl oxime ether compounds with excellent yields and enantiomeric excess values. The versatility of this method is underscored by its compatibility with a wide array of substituents, allowing for the rapid generation of diverse chemical libraries.
Mechanistic Insights into Copper-Catalyzed Asymmetric Oxime Etherification
The core of this technological breakthrough lies in the precise design of the catalytic species. The active catalyst is formed by the coordination of a copper salt, preferably Cu(OTf)2, CuCl, CuBr, or CuI, with a specialized chiral P,N,N-ligand. These ligands, characterized by a phosphine group and two nitrogen-containing moieties, create a chiral environment around the copper center that dictates the stereochemical outcome of the nucleophilic attack. The mechanism likely involves the activation of the propargyl ester by the copper complex to form a reactive pi-allyl copper intermediate, which is then attacked by the oxime oxygen nucleophile in a highly enantioselective manner. The use of P,N,N-ligands allows for fine-tuning of the steric and electronic properties of the catalyst, which is crucial for achieving the observed high levels of stereoselectivity (up to 95% ee in certain substrates).
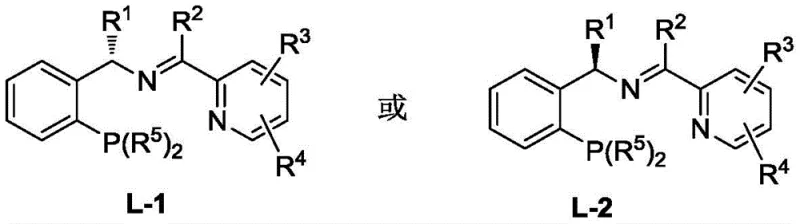
From an impurity control perspective, the mild reaction conditions play a vital role. Conducting the reaction at -20°C minimizes side reactions such as polymerization of the alkyne or decomposition of sensitive functional groups on the aryl ring. The protocol specifies the use of alkali additives like iPr2NEt, NEt3, or Cs2CO3 to facilitate the deprotonation of the oxime or stabilize the transition state, ensuring clean conversion to the desired ether product. The broad tolerance for substituents—including halogens, nitro groups, esters, and cyano groups on the phenyl ring of the oxime, as well as diverse R groups on the propargyl component—demonstrates the robustness of the catalytic cycle. This mechanistic stability ensures that the process remains reliable even when scaling up or modifying the molecular scaffold for SAR studies.
How to Synthesize Chiral Aryl Oxime Ether Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for manufacturing these valuable intermediates. The process begins with the in situ generation of the chiral copper catalyst under an inert nitrogen atmosphere, ensuring the exclusion of moisture and oxygen which could deactivate the catalyst. Subsequently, the substrates—aryl oxime and propargyl compound—are combined with a base in a suitable solvent and introduced to the catalyst solution. The reaction is allowed to proceed at low temperature for a defined period, typically around 24 hours, to ensure complete conversion. Following the reaction, standard workup procedures involving concentration and silica gel column chromatography yield the pure chiral product. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Prepare the chiral copper catalyst by stirring a copper salt (e.g., Cu(OTf)2) and a chiral P,N,N-ligand in a reaction medium under nitrogen protection for 0.5 to 2 hours.
- Dissolve the propargyl compound, aryl oxime, and an alkali additive (e.g., iPr2NEt) in a solvent like methanol, then add this mixture to the catalyst solution.
- Stir the reaction mixture at -20°C for at least 12 hours, followed by concentration, silica gel column chromatography separation, and vacuum drying to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this copper-catalyzed technology offers distinct strategic advantages over traditional precious metal catalysis or resolution-based methods. The primary benefit stems from the use of copper, an abundant and inexpensive base metal, which drastically reduces the raw material cost compared to processes relying on rhodium, palladium, or iridium. Furthermore, the ligands employed are described as simple and convenient to synthesize, implying a stable and cost-effective supply chain for the catalytic system itself. The ability to run reactions at -20°C rather than requiring cryogenic conditions (e.g., -78°C) or elevated temperatures translates to significant energy savings and reduced complexity in reactor management, directly impacting the cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The elimination of expensive precious metal catalysts is a major driver for cost optimization. Copper salts are orders of magnitude cheaper than their noble metal counterparts, and the catalyst loading can be as low as 0.001:1 molar ratio in optimized scenarios, although typical examples use around 5 mol%. Additionally, the use of common solvents like methanol and ethanol avoids the need for specialized, high-cost anhydrous solvents often required in sensitive organometallic chemistry. The high stereoselectivity achieved (often >85% ee) reduces or eliminates the need for costly downstream chiral resolution steps, such as preparative HPLC or recrystallization of diastereomeric salts, thereby streamlining the entire production workflow and maximizing overall yield.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more reliable supply chain. The tolerance for a wide range of functional groups means that a single standardized protocol can be applied to synthesize a diverse portfolio of intermediates without extensive re-optimization for each new derivative. This flexibility allows manufacturers to respond quickly to changing demands for different API precursors. Moreover, the starting materials—aryl oximes and propargyl alcohol esters—are generally commercially available or easily synthesized from commodity chemicals, reducing the risk of supply bottlenecks associated with exotic or proprietary reagents.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, the process is highly favorable. The mild temperatures and ambient pressure operations reduce the thermal load on manufacturing facilities, enhancing safety profiles. The use of copper, while requiring appropriate waste management, is generally less toxic and easier to handle than heavy precious metals. The high atom economy of the substitution reaction, coupled with the high yields reported (up to 89% in some examples), minimizes waste generation. This aligns well with green chemistry principles and simplifies regulatory compliance for waste disposal, making the scale-up from laboratory grams to multi-ton commercial production a smoother and more sustainable endeavor.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy for technical evaluation.
Q: What represents the key innovation in this copper-catalyzed oxime etherification?
A: The key innovation is the successful use of aryl oximes as oxygen nucleophiles in copper-catalyzed asymmetric propargyl substitution, a reaction type previously rarely reported compared to nitrogen or carbon nucleophiles. This method achieves high enantioselectivity (up to 95% ee) under mild conditions (-20°C).
Q: What are the optimal reaction conditions for this synthesis?
A: The optimal conditions involve using Cu(OTf)2 or Cu(OAc)2·H2O with a chiral P,N,N-ligand in methanol or ethanol at -20°C. The reaction typically requires an alkali additive like iPr2NEt and proceeds effectively with a catalyst loading as low as 0.001:1 molar ratio relative to the substrate.
Q: Does this method support a wide range of substrates?
A: Yes, the method demonstrates broad substrate applicability. It tolerates various substituents on the aryl oxime (such as halogens, alkyl, alkoxy, nitro groups) and diverse groups on the propargyl compound (including phenyl, substituted phenyl, naphthyl, and cycloalkyl groups), making it highly versatile for generating libraries of chiral intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Aryl Oxime Ether Supplier
The development of efficient asymmetric synthesis routes like the one described in CN112279779B underscores the importance of partnering with a CDMO that possesses deep technical expertise in catalytic processes. NINGBO INNO PHARMCHEM stands at the forefront of this capability, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and stringent purity specifications to ensure that every batch of chiral aryl oxime ether meets the exacting standards required for pharmaceutical applications. We understand the critical nature of enantiomeric purity and are committed to delivering high-quality intermediates that accelerate your drug development timelines.
We invite you to leverage our technical proficiency to optimize your supply chain for these complex molecules. Whether you require custom synthesis of specific derivatives or scale-up of the copper-catalyzed process, our team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your long-term commercial goals.
