Advanced Asymmetric Synthesis of Dexchlorpheniramine for Commercial API Production
Advanced Asymmetric Synthesis of Dexchlorpheniramine for Commercial API Production
The pharmaceutical industry continuously seeks more efficient pathways for producing high-value antihistamine intermediates, and the technology disclosed in patent CN111138350B represents a significant leap forward in this domain. This patent details a novel asymmetric synthesis method for dextro-halogenated pheniramines, specifically targeting dexchlorpheniramine and dexbrompheniramine, which are critical active pharmaceutical ingredients (APIs) for treating allergic conditions. Unlike traditional methods that rely on the resolution of racemates, this innovative approach utilizes a chiral rhodium-catalyzed conjugate addition to establish the stereocenter with exceptional precision. The process achieves single-step yields of up to 96 percent and enantiomeric excess values reaching 96 percent, demonstrating a level of efficiency that is highly attractive for large-scale manufacturing. By shifting from resolution to direct asymmetric synthesis, manufacturers can drastically reduce raw material consumption and waste generation, aligning with modern green chemistry principles while securing a more reliable supply of high-purity intermediates.
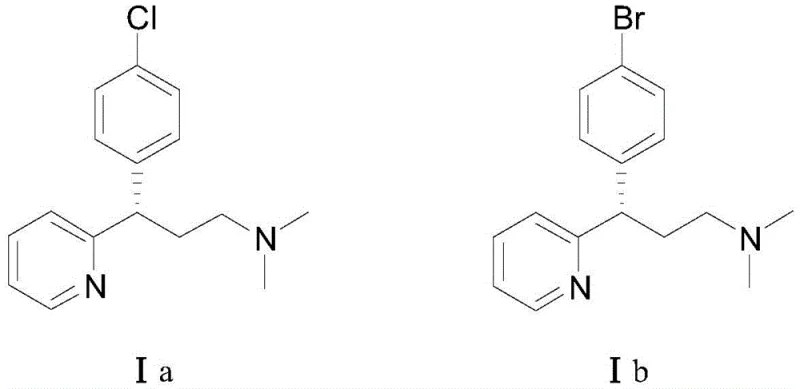
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of single-enantiomer antihistamines like dexchlorpheniramine has been dominated by chiral resolution techniques, which suffer from inherent thermodynamic and economic inefficiencies. In a typical resolution process, a racemic mixture is synthesized first, requiring the subsequent separation of enantiomers using chiral resolving agents. This methodology fundamentally limits the maximum theoretical yield to 50 percent, as the unwanted enantiomer is often discarded or requires complex recycling processes that add significant cost and environmental burden. Furthermore, the use of stoichiometric amounts of expensive resolving agents increases the overall cost of goods sold (COGS) and complicates the purification workflow. The generation of substantial solid waste from resolving salts poses a significant challenge for environmental compliance, creating pressure on supply chain managers to find cleaner alternatives. Additionally, the multiple crystallization steps required to achieve high optical purity often lead to prolonged production cycles, increasing lead times and reducing the agility of the supply chain in responding to market demand fluctuations.
The Novel Approach
The technology outlined in patent CN111138350B introduces a streamlined four-step synthetic route that bypasses the need for resolution entirely by constructing the chiral center at the outset. The core of this innovation is the rhodium-catalyzed asymmetric 1,4-addition of arylboronic acids to ethyl 3-(2-pyridyl)acrylate. This reaction not only forms the carbon-carbon bond but also sets the stereochemistry with high fidelity in a single operation. Following the addition, the sequence proceeds through hydrolysis, amidation, and reduction to deliver the final amine product. This convergent strategy significantly shortens the synthetic timeline compared to multi-step resolution protocols. The use of readily available starting materials, such as substituted phenylboronic acids and pyridyl acrylates, ensures that the supply chain remains robust and less susceptible to raw material shortages. The high atom economy of the conjugate addition step means that nearly all input materials are incorporated into the product, minimizing waste and maximizing resource efficiency.
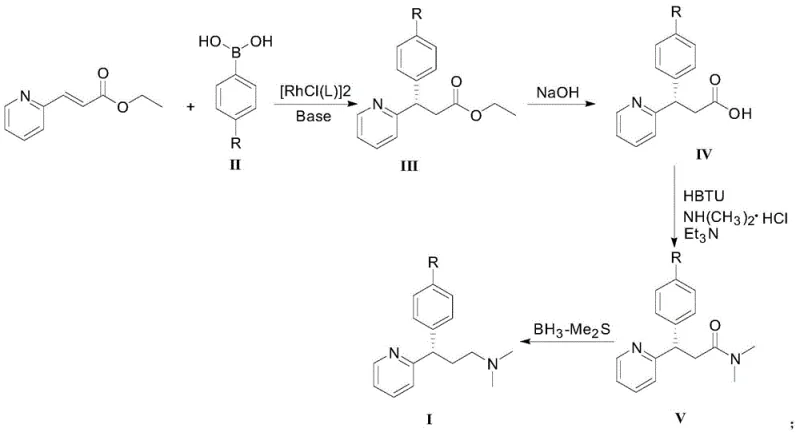
Mechanistic Insights into Rhodium-Catalyzed Asymmetric Conjugate Addition
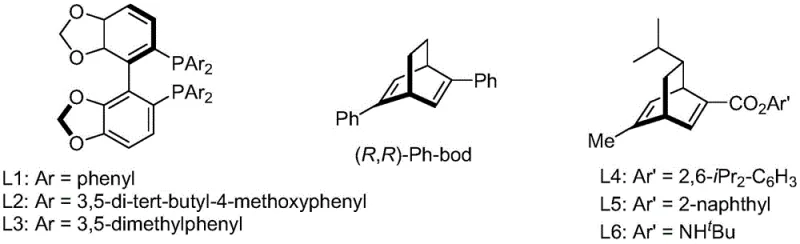
The success of this synthesis hinges on the precise engineering of the catalytic cycle, where a chiral rhodium complex facilitates the enantioselective transfer of the aryl group to the activated alkene. The catalyst system typically comprises a rhodium chloride precursor coordinated with a chiral diene or phosphine ligand. Among the various ligands screened, the patent highlights the superiority of specific chiral diene ligands, such as L3 (where Ar is 3,5-dimethylphenyl), in inducing high levels of stereocontrol. The steric and electronic properties of these ligands create a chiral environment around the metal center that favors the formation of one enantiomer over the other during the migratory insertion step. This mechanistic precision is critical for pharmaceutical applications, where regulatory agencies mandate strict limits on chiral impurities. By achieving 96 percent ee in the initial step, the downstream purification burden is significantly lightened, ensuring that the final API meets stringent quality specifications without the need for extensive chromatographic separations.
Impurity control is further enhanced by the mild reaction conditions employed throughout the sequence. The conjugate addition is conducted in mixed solvent systems like tetrahydrofuran and water at moderate temperatures, which minimizes side reactions such as polymerization or over-reduction. The subsequent hydrolysis and amidation steps utilize standard reagents like sodium hydroxide and HBTU, which are well-understood in process chemistry and generate predictable byproduct profiles. The final reduction step using borane-dimethyl sulfide complex is highly chemoselective for the amide functionality, leaving the sensitive pyridine and aryl halide moieties intact. This chemoselectivity is vital for maintaining the structural integrity of the molecule and preventing the formation of difficult-to-remove impurities. The combination of high catalyst turnover and selective reactivity results in a clean crude product profile, facilitating easier isolation and higher overall recovery rates.
How to Synthesize Dexchlorpheniramine Efficiently
The synthesis of dexchlorpheniramine via this patented route involves a logical sequence of transformations designed for operational simplicity and high yield. The process begins with the catalytic coupling of the boronic acid and acrylate, followed by functional group manipulations to install the amine tail. Detailed standard operating procedures for each step, including specific molar ratios, solvent choices, and workup protocols, are essential for reproducing the high yields reported in the patent literature. Process chemists should pay close attention to the catalyst loading and ligand selection, as these parameters directly influence the enantiomeric purity of the intermediate. For a comprehensive guide on executing this synthesis with optimal results, please refer to the standardized protocol below.
- Perform asymmetric conjugate addition of 4-chlorophenylboronic acid to ethyl 3-(2-pyridyl)acrylate using a chiral rhodium catalyst.
- Hydrolyze the resulting ester intermediate to the corresponding carboxylic acid using aqueous base.
- Condense the acid with dimethylamine hydrochloride using HBTU to form the amide intermediate.
- Reduce the amide to the final amine product using a borane reducing agent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this asymmetric synthesis route offers compelling economic and logistical benefits over legacy technologies. The elimination of chiral resolution agents removes a significant cost driver from the bill of materials, directly contributing to margin improvement. Furthermore, the doubling of theoretical yield compared to resolution methods means that manufacturers can produce twice the amount of product from the same quantity of starting materials, effectively halving the raw material intensity per kilogram of output. This efficiency gain translates into substantial cost savings and reduces the environmental footprint associated with raw material extraction and transport. The robustness of the reaction conditions also implies lower energy consumption and reduced equipment wear, further enhancing the economic viability of the process.
- Cost Reduction in Manufacturing: The transition to a direct asymmetric synthesis eliminates the need for expensive chiral resolving agents and the associated recycling infrastructure. By avoiding the 50 percent yield loss inherent in resolution, the effective cost of the active moiety is drastically reduced. The use of commodity chemicals like arylboronic acids and simple esters ensures that raw material costs remain stable and predictable. Additionally, the high selectivity of the process reduces the volume of solvent and consumables required for purification, leading to lower waste disposal fees and operational expenditures.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials mitigates the risk of supply disruptions caused by specialized reagent shortages. Arylboronic acids and pyridyl acrylates are produced by numerous global suppliers, fostering a competitive sourcing environment that protects against price volatility. The shortened synthetic route reduces the number of unit operations and intermediate handlings, which decreases the probability of batch failures and delays. This streamlined workflow enables faster turnaround times from order to delivery, allowing pharmaceutical companies to maintain leaner inventory levels while ensuring continuity of supply for their finished dosage forms.
- Scalability and Environmental Compliance: The process has been demonstrated to scale effectively from gram to multi-gram quantities without loss of performance, indicating strong potential for tonnage production. The absence of heavy metal waste streams associated with some alternative catalytic methods simplifies effluent treatment and regulatory compliance. The high atom economy aligns with sustainability goals, making it easier for manufacturers to meet corporate social responsibility targets. The robust nature of the chemistry allows for flexible manufacturing strategies, including campaign production, which optimizes asset utilization and supports long-term supply agreements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the primary advantage of this rhodium-catalyzed route over traditional resolution methods?
A: The primary advantage is the elimination of the 50% material loss inherent in racemic resolution. This asymmetric synthesis builds the chiral center directly with high enantiomeric excess (up to 96% ee), significantly improving atom economy and reducing waste disposal costs.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the patent data demonstrates successful scale-up experiments. The use of robust reagents like arylboronic acids and standard reduction conditions allows for stable production from laboratory to commercial tonnage scales without compromising enantioselectivity.
Q: What specific ligand system provides the optimal enantioselectivity?
A: The patent identifies chiral diene ligands, specifically the L3 ligand (Ar = 3,5-dimethylphenyl), as providing superior performance. This ligand system, when complexed with rhodium chloride, achieves yields up to 96% with 96% ee in the key conjugate addition step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dexchlorpheniramine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in patent CN111138350B for securing a competitive edge in the pharmaceutical market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into robust industrial processes. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging our expertise in asymmetric catalysis and process optimization, we help our partners navigate the complexities of chiral synthesis to achieve consistent quality and supply stability.
We invite you to collaborate with us to explore how this efficient synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can support your long-term business goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
