Scalable Anhydrous Synthesis of Aripiprazole Lauroxil for High-Purity Antipsychotic Prodrugs
Introduction to Advanced Prodrug Manufacturing
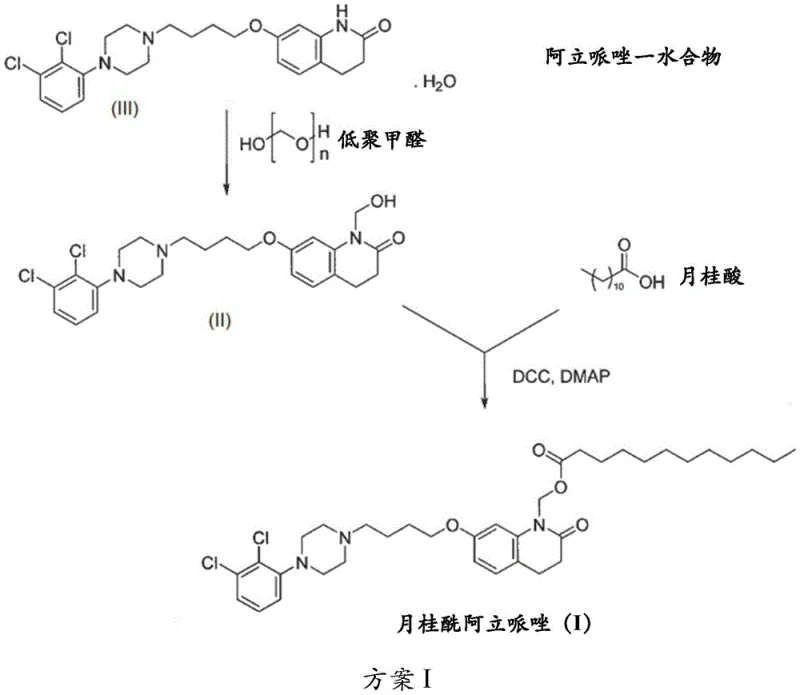
The pharmaceutical landscape for long-acting injectable antipsychotics has been significantly advanced by the disclosure in patent CN111315722B, which details a robust method for preparing Aripiprazole Lauroxil. This specific prodrug is critical for treating schizophrenia and bipolar disorder, offering extended therapeutic release profiles that improve patient compliance. The core innovation lies in shifting from traditional aqueous formaldehyde methodologies to an anhydrous paraformaldehyde approach, fundamentally altering the reaction kinetics and impurity profile. By strictly controlling water content to below 1% by weight during the initial hydroxymethylation step, manufacturers can achieve conversion rates exceeding 77%, a substantial improvement over the 65% conversion typically observed with 37% aqueous formaldehyde solutions. This technical breakthrough not only enhances the overall yield but also simplifies the downstream purification processes, making it a highly attractive route for industrial scale-up.
For R&D directors and process chemists, the significance of this patent extends beyond mere yield improvements; it addresses the critical challenge of impurity management in complex ester synthesis. The document explicitly identifies the formation of a specific formate ester impurity (Compound IV) resulting from side reactions with formic acid generated in situ. By providing a validated method to synthesize this impurity as a reference standard, the patent empowers quality control teams to implement precise HPLC monitoring protocols. This level of analytical rigor ensures that the final Active Pharmaceutical Ingredient (API) meets stringent regulatory specifications, thereby reducing the risk of batch failures during commercial production. Consequently, this process represents a paradigm shift towards more predictable and controllable manufacturing of long-acting injectable formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of hydroxymethylated intermediates for prodrugs like Aripiprazole Lauroxil relied heavily on the use of aqueous formaldehyde, typically in concentrations around 37%. While chemically straightforward, this conventional approach introduces a significant volume of water into the reaction matrix, which acts as a thermodynamic sink that hinders the forward reaction. As documented in prior art such as WO2010151689, the presence of excess water limits the conversion of the starting aripiprazole to merely 65%, leaving a substantial 25% of unreacted starting material in the crude mixture. This low conversion rate creates a bottleneck for purification, requiring extensive chromatographic separation or multiple recrystallization steps to remove the persistent aripiprazole impurity, which drastically increases processing time and solvent consumption. Furthermore, the aqueous environment promotes hydrolysis and other side reactions that generate difficult-to-remove byproducts, compromising the overall purity profile of the intermediate before it even reaches the esterification stage.
The Novel Approach
The novel methodology disclosed in CN111315722B circumvents these limitations by utilizing paraformaldehyde as the source of the hydroxymethyl group in conjunction with strictly anhydrous or near-anhydrous conditions. By employing organic solvents such as toluene, ethyl acetate, or acetone, and limiting the total water content to less than 1% by weight, the reaction equilibrium is driven decisively towards the formation of 1-(hydroxymethyl)aripiprazole (Compound II). This strategic modification allows for conversion rates to surpass 77%, significantly reducing the burden on downstream purification units. The process utilizes bases like DBU or potassium carbonate to facilitate the depolymerization of paraformaldehyde and the subsequent nucleophilic attack on the aripiprazole nitrogen. This results in a cleaner reaction profile where the primary impurity is easily managed, enabling a more streamlined isolation of the intermediate via simple filtration or slurring, thus enhancing the overall efficiency of the manufacturing workflow.
Mechanistic Insights into Anhydrous Hydroxymethylation and Esterification
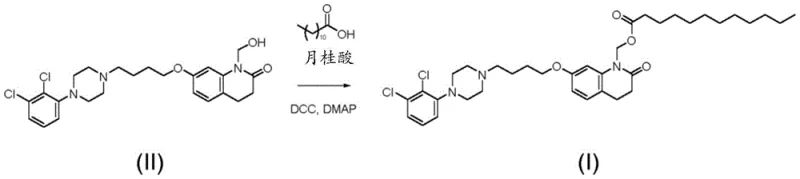
The mechanistic pathway for this synthesis involves two distinct catalytic phases that require precise control to maximize yield and minimize impurities. In the first phase, the hydroxymethylation of aripiprazole, the choice of base is critical; 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) acts as a non-nucleophilic base that effectively deprotonates the secondary amine of aripiprazole without inducing unwanted side reactions. The paraformaldehyde depolymerizes in situ to release formaldehyde monomers, which then react with the activated amine to form the N-hydroxymethyl intermediate. The absence of water is paramount here, as water would compete for the formaldehyde, forming methylene glycol and reversing the desired reaction. In the second phase, the esterification with lauric acid, the mechanism shifts to a carbodiimide-mediated coupling. Here, dicyclohexylcarbodiimide (DCC) activates the carboxylic acid of lauric acid to form an O-acylisourea intermediate, which is highly susceptible to nucleophilic attack by the hydroxyl group of the intermediate.
Impurity control is deeply embedded in the mechanistic understanding of this process, particularly regarding the formation of the formate ester impurity (Compound IV). The patent reveals that formic acid can be generated as a byproduct during the reaction or exist as a contaminant in reagents, which then reacts with the hydroxymethyl intermediate to form the undesired formate ester. To mitigate this, the process incorporates a purification step for the intermediate (Compound II) involving slurring in aqueous media with mild bases like sodium bicarbonate or triethylamine. This step effectively neutralizes and removes any trace formic acid before the esterification begins. Additionally, the final recrystallization in isopropanol leverages solubility differences to exclude the formate impurity, ensuring the final API meets the rigorous purity standard of greater than 99.5% HPLC area. This multi-layered approach to impurity management demonstrates a sophisticated understanding of reaction kinetics and thermodynamics.
How to Synthesize Aripiprazole Lauroxil Efficiently
The synthesis of Aripiprazole Lauroxil via this patented route offers a clear, scalable pathway that balances reaction efficiency with ease of operation. The process begins with the suspension of aripiprazole monohydrate in toluene, followed by the addition of paraformaldehyde and a catalytic amount of DBU. Heating this mixture to 30-40°C under a nitrogen atmosphere for approximately 16 hours ensures complete conversion while maintaining the integrity of the sensitive hydroxymethyl group. Once the reaction is complete, cooling the mixture induces precipitation of the intermediate, which is then isolated by filtration. This solid intermediate can be further purified by slurring in water to remove residual base and formic acid, a crucial step for preventing downstream impurity formation. The dried intermediate is then dissolved in dichloromethane for the subsequent esterification step.
- React aripiprazole monohydrate with paraformaldehyde in toluene using DBU base at 30-40°C under anhydrous conditions to form 1-(hydroxymethyl)aripiprazole.
- Purify the intermediate via aqueous slurry and filtration to remove unreacted starting materials and ensure low water content.
- Perform esterification with lauric acid using DCC and DMAP in dichloromethane, followed by recrystallization in isopropanol to achieve >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this anhydrous synthesis route presents compelling economic and logistical advantages that directly impact the bottom line. The shift from aqueous formaldehyde to paraformaldehyde eliminates the need for handling hazardous, high-water-content liquids, thereby reducing shipping weights and associated logistics costs. Paraformaldehyde is a stable, solid polymer that is easier to store and transport, enhancing supply chain reliability and reducing the risk of degradation during warehousing. Furthermore, the significant improvement in reaction conversion rates means that less raw material is wasted as unreacted starting material, leading to a more efficient utilization of expensive aripiprazole feedstock. This efficiency translates into substantial cost savings in raw material procurement, as the overall yield from starting material to final API is markedly higher than conventional methods.
- Cost Reduction in Manufacturing: The elimination of water from the reaction system drastically simplifies the workup and purification stages. Conventional aqueous processes often require energy-intensive drying steps and large volumes of solvents to extract products from water, whereas this anhydrous process allows for direct filtration and crystallization. By reducing the number of unit operations and the volume of solvents required for purification, the manufacturing facility can achieve significant reductions in utility consumption and waste disposal costs. Additionally, the higher yield per batch means that fewer batches are required to meet production targets, optimizing the utilization of reactor capacity and labor resources without compromising quality standards.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route against moisture variations ensures consistent batch-to-batch quality, which is critical for maintaining a reliable supply of API to formulation partners. The use of commercially available and stable reagents like paraformaldehyde and lauric acid minimizes the risk of supply disruptions caused by the scarcity of specialized reagents. Moreover, the ability to purify the intermediate effectively before the final coupling step acts as a quality gate, preventing the propagation of impurities that could lead to batch rejections later in the process. This predictability allows supply chain planners to forecast production timelines with greater accuracy, ensuring timely delivery to downstream customers.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process aligns well with green chemistry principles by minimizing solvent usage and avoiding the generation of large volumes of aqueous waste streams. The use of dichloromethane in the esterification step is managed through efficient recovery and recycling protocols, and the solid byproducts like dicyclohexylurea are easily filtered and disposed of. The scalability of the process is evidenced by the use of standard reaction conditions (room temperature to 40°C) that do not require extreme heating or cooling, making it suitable for transfer from pilot plant to multi-ton commercial production. This ease of scale-up reduces the time-to-market for generic versions of the drug, providing a competitive edge in the pharmaceutical marketplace.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the manufacturing of Aripiprazole Lauroxil using this advanced anhydrous methodology. These insights are derived directly from the experimental data and claims presented in patent CN111315722B, providing a factual basis for decision-making. Understanding these nuances is essential for stakeholders evaluating the feasibility of technology transfer or sourcing strategies for this high-value antipsychotic prodrug.
Q: Why is paraformaldehyde preferred over aqueous formaldehyde in this synthesis?
A: Using paraformaldehyde under anhydrous conditions prevents the formation of excessive water, which significantly reduces the conversion rate of the hydroxymethylation step. This method achieves conversions higher than 77% compared to only 65% with aqueous formaldehyde, minimizing residual aripiprazole impurities.
Q: How is the formate ester impurity controlled during manufacturing?
A: The formate ester impurity forms when the intermediate reacts with formic acid byproducts. The process controls this by removing formic acid during the purification of the intermediate or by recrystallizing the final product in isopropanol, reducing impurity levels to below 0.1%.
Q: What represents the critical quality attribute for the final API?
A: The critical quality attribute is the chemical purity, specifically ensuring HPLC purity of at least 99.5%. This is achieved through a rigorous recrystallization process in alcoholic solvents like isopropanol after the initial esterification reaction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aripiprazole Lauroxil Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates and APIs in the development of effective CNS therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the complex anhydrous synthesis of Aripiprazole Lauroxil is executed with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art HPLC and MS instrumentation to detect and quantify trace impurities like the formate ester down to 0.1% levels. Our commitment to quality assurance guarantees that every batch of Aripiprazole Lauroxil we supply meets the highest international regulatory standards, providing our partners with the confidence they need to advance their clinical and commercial programs.
We invite pharmaceutical companies and contract manufacturing organizations to collaborate with us to leverage this superior synthetic route for their supply needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that demonstrates how our optimized process can reduce your overall cost of goods sold. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation. Let us help you secure a stable, high-quality supply of Aripiprazole Lauroxil that supports your mission to deliver life-changing medications to patients worldwide.
