Advanced Synthesis of Poly-substituted 5-Acyl-2-Iminothiazolines for Commercial Scale-up
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient routes to bioactive scaffolds. Patent CN102127032B introduces a groundbreaking methodology for the synthesis of poly-substituted 5-acyl-2-iminothiazolines, a class of compounds renowned for their potent biological activities ranging from insecticidal and antibacterial properties in agriculture to kinase inhibition in medicine. This innovation addresses critical bottlenecks in traditional organic synthesis by leveraging a multicomponent strategy that utilizes commodity chemicals such as carbodiimides, acid chlorides, and terminal alkynes. By shifting away from complex, hard-to-source precursors, this technology offers a streamlined pathway that significantly enhances the feasibility of producing these valuable intermediates on an industrial scale. For R&D teams and procurement strategists alike, this represents a pivotal opportunity to optimize supply chains for high-value agrochemical and pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-acyl-2-imine thiazoline derivatives has been plagued by significant operational challenges that hinder commercial viability. Traditional routes often rely on starting materials that are synthetically difficult to access, requiring multi-step preparations that drastically inflate costs and extend lead times. Furthermore, existing methodologies frequently suffer from a narrow substrate scope, meaning that slight modifications to the molecular structure can cause the reaction to fail or yield unacceptable levels of impurities. These limitations create a fragile supply chain where the availability of key intermediates is contingent upon the successful execution of low-yielding, complex transformations. For procurement managers, this translates to volatile pricing and unreliable delivery schedules, while R&D directors face constant hurdles in scaling these processes from the bench to the pilot plant without compromising on purity or throughput.
The Novel Approach
In stark contrast, the method disclosed in CN102127032B revolutionizes the production landscape by employing a robust, convergent synthesis strategy. This novel approach utilizes a sequential addition of readily available industrial reagents, including carbodiimides, acyl chlorides, and terminal alkynes, in the presence of elemental sulfur. The process is designed to be operationally simple, utilizing common ether solvents and avoiding the need for exotic transition metal catalysts that often require expensive removal steps. By integrating the formation of the thiazoline ring with the installation of the acyl and imino substituents in a cohesive sequence, the method achieves high isolated yields, often exceeding 80%, with exceptional purity profiles. This shift not only simplifies the chemical logic but also creates a resilient manufacturing protocol that is inherently more cost-effective and scalable.
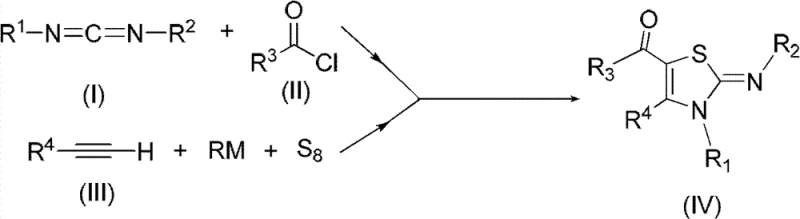
Mechanistic Insights into Multicomponent Thiazoline Cyclization
The chemical elegance of this synthesis lies in its precise orchestration of reactive intermediates to construct the thiazoline core. The mechanism initiates with the activation of the carbodiimide by the acid chloride in an ether solvent, generating a highly reactive ketene imine or similar electrophilic species. Concurrently, the terminal alkyne undergoes lithiation using an organolithium reagent like n-Butyl Lithium at cryogenic temperatures, typically between -78°C and 0°C, to form a nucleophilic acetylide. The subsequent introduction of elemental sulfur allows for the formation of a lithium thiolate or polysulfide intermediate, which serves as the sulfur source for the heterocycle. When these two distinct reaction streams are combined and heated to 60-100°C, a cascade of cyclization events occurs, efficiently closing the ring to form the 5-acyl-2-iminothiazoline scaffold. This mechanistic pathway minimizes side reactions and ensures that the diverse functional groups tolerated by the substrates remain intact throughout the transformation.
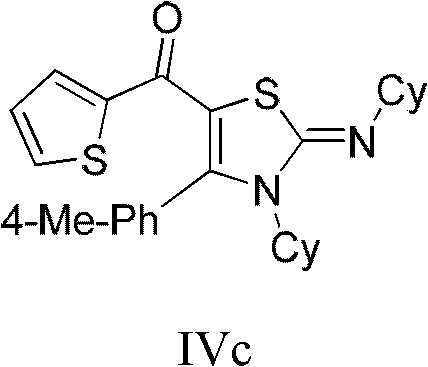
From an impurity control perspective, the specificity of this reaction sequence is paramount for producing high-purity intermediates required in regulated industries. The use of stoichiometric ratios, such as a 1:1.2 ratio of acyl chloride to carbodiimide, ensures complete consumption of the limiting reagents, thereby reducing the burden on downstream purification. The reaction conditions are mild enough to prevent the decomposition of sensitive functional groups, such as the trifluoromethyl or heteroaryl moieties often found in bioactive molecules. For instance, the synthesis of derivative IVc demonstrates the compatibility of thiophene rings and cyclohexyl groups, yielding a product with purity greater than 98%. This level of control is critical for R&D directors who must ensure that the impurity profile of their API intermediates remains within strict regulatory limits, avoiding the formation of genotoxic impurities or difficult-to-remove byproducts that could derail clinical development.
How to Synthesize Poly-substituted 5-Acyl-2-Iminothiazolines Efficiently
Implementing this synthesis route requires careful attention to temperature control and reagent addition sequences to maximize yield and safety. The process is divided into distinct stages: the preparation of the activated carbodiimide species, the generation of the sulfur-containing alkyne intermediate, and the final thermal cyclization. Operators must maintain an inert atmosphere, typically nitrogen, to prevent the quenching of the organolithium reagent by moisture or oxygen. The detailed标准化 synthesis steps见下方的指南。
- React carbodiimide with acid chloride in an ether solvent at temperatures between 25°C and the solvent's boiling point to form the activated intermediate.
- Treat a terminal alkyne compound with an organolithium reagent (such as n-Butyl Lithium) in a second ether solvent at cryogenic temperatures (-78°C to 0°C), followed by the addition of elemental sulfur.
- Combine the two reaction mixtures and raise the temperature to 60-100°C to facilitate the cyclization and formation of the thiazoline ring.
- Perform post-processing workup including concentration and chromatographic purification using petroleum ether and dichloromethane to isolate the final product with >98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology offers profound strategic benefits that extend beyond simple chemical efficiency. By relying on commodity chemicals such as acid chlorides and carbodiimides, manufacturers can decouple their production from the volatility associated with specialized, low-volume reagents. This shift significantly reduces the risk of supply disruptions and allows for more accurate forecasting of raw material costs. Furthermore, the elimination of complex catalytic systems removes the need for expensive metal scavenging processes, which are often a hidden cost driver in fine chemical manufacturing. The result is a leaner, more agile supply chain capable of responding rapidly to market demands for agrochemical and pharmaceutical intermediates without compromising on quality or compliance.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the use of inexpensive, bulk-available starting materials and the avoidance of precious metal catalysts. By streamlining the synthesis into fewer operational steps with high atom economy, the overall cost of goods sold (COGS) is drastically lowered. Additionally, the high isolated yields reduce the amount of raw material waste, further enhancing the financial efficiency of the production line. This cost structure allows suppliers to offer competitive pricing for high-purity intermediates, providing a distinct advantage in price-sensitive markets like generic pharmaceuticals and crop protection.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents like elemental sulfur and common ether solvents ensures that production can continue uninterrupted even during global supply chain fluctuations. The robustness of the reaction conditions means that the process is less susceptible to minor variations in raw material quality, reducing the rate of batch failures. For supply chain planners, this reliability translates to shorter lead times and the ability to maintain consistent inventory levels, ensuring that downstream customers receive their orders on schedule every time.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of hazardous high-pressure or high-temperature requirements. The use of standard ether solvents facilitates easy recovery and recycling, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing site. This ease of scale-up enables rapid capacity expansion to meet surging demand, while the simplified waste profile ensures compliance with increasingly stringent environmental regulations regarding solvent discharge and heavy metal contamination.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thiazoline synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of this synthesis method over conventional routes?
A: The method utilizes readily available industrial chemicals like carbodiimides and acid chlorides, avoiding complex starting materials and offering higher isolated yields up to 82% compared to existing methods which often suffer from restricted substrate scope.
Q: Can this process be scaled for commercial production of agrochemical intermediates?
A: Yes, the reaction conditions utilize common ether solvents and manageable temperature ranges (25°C to 100°C), making it highly suitable for large-scale manufacturing without requiring exotic catalysts or extreme pressures.
Q: What is the purity profile of the resulting thiazoline derivatives?
A: The process consistently delivers products with purity greater than 98% after standard silica gel column purification, meeting stringent requirements for pharmaceutical applications and reducing the need for extensive recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Poly-substituted 5-Acyl-2-Iminothiazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthesis route for the next generation of bioactive molecules. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from concept to market. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of poly-substituted thiazoline meets the highest standards of quality required for pharmaceutical and agrochemical applications. We are committed to being a reliable partner who understands the complexities of heterocyclic chemistry and the critical nature of your supply chain.
We invite you to collaborate with us to unlock the full commercial potential of this technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can drive efficiency and reduce costs for your organization.
