Advanced Synthesis of Multi-Substituted 2-Imidoimidazoline-4,5-Diketones for Global Supply Chains
Advanced Synthesis of Multi-Substituted 2-Imidoimidazoline-4,5-Diketones for Global Supply Chains
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for greener, more efficient synthetic routes for high-value intermediates. A pivotal advancement in this domain is detailed in patent CN102532030A, which discloses a robust and universal synthesis method for multi-substituted 2-iminoimidazoline-4,5-dione derivatives. These compounds serve as critical scaffolds in the development of next-generation agrochemicals, functioning as potent insecticides and herbicides, while also holding significant promise in the pharmaceutical sector as thrombin inhibitors and anti-inflammatory agents. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding the mechanistic superiority and operational simplicity of this patent is essential. The technology replaces hazardous traditional oxidation steps with a streamlined acylation-cyclization sequence, utilizing readily available carbodiimides and oxalyl chloride. This shift not only enhances the safety profile of the manufacturing process but also ensures a consistent supply of high-purity OLED material precursors and pharmaceutical building blocks, addressing the growing demand for environmentally compliant chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-iminoimidazoline-4,5-dione derivatives has been plagued by significant technical and environmental hurdles that hindered their widespread commercial adoption. Traditional protocols often relied on the oxidation of 5-methylene groups starting from 1-methylhydantoin-2-imide, utilizing toxic heavy metal oxidants such as mercury acetate. This reliance on mercury-based reagents introduces severe complications for modern supply chain heads, including stringent regulatory compliance burdens, high costs associated with hazardous waste disposal, and potential contamination of the final product with residual heavy metals. Furthermore, these conventional methods exhibited poor substrate tolerance, severely limiting the diversity of substituents that could be introduced at the R1, R2, and R3 positions. The inability to easily vary these functional groups restricted the chemical space available for drug discovery and agrochemical optimization, forcing R&D teams to work with a narrow library of compounds. Additionally, the harsh reaction conditions often led to lower yields and complex purification challenges, making the cost reduction in pharmaceutical intermediates manufacturing difficult to achieve using legacy technologies.
The Novel Approach
In stark contrast, the methodology outlined in the patent data presents a transformative approach that circumvents these historical bottlenecks through a clever two-step sequence involving carbodiimides and oxalyl chloride. By initiating the reaction between a substituted carbodiimide and oxalyl chloride in an ether solvent under anhydrous conditions, the process generates a reactive intermediate that is subsequently trapped by a primary amine. This strategy eliminates the need for toxic mercury oxidants entirely, replacing them with benign organic bases like triethylamine or diisopropylethylamine to neutralize byproduct hydrogen chloride. The result is a universally applicable synthetic route that accommodates a vast array of substituents, including alkyl, cycloalkyl, aryl, and heteroaryl groups, without compromising yield or purity. For a reliable pharma intermediate supplier, this versatility is paramount, as it allows for the rapid generation of diverse compound libraries for biological screening. The operational simplicity, often proceeding at room temperature or mild heating, further underscores the commercial viability of this method, enabling seamless scale-up from laboratory grams to multi-ton production batches.
Mechanistic Insights into Carbodiimide-Mediated Cyclization
The core of this synthetic breakthrough lies in the unique reactivity of the carbodiimide functionality when exposed to electrophilic acylating agents like oxalyl chloride. The mechanism initiates with the nucleophilic attack of the carbodiimide nitrogen on the carbonyl carbon of oxalyl chloride, forming a highly reactive imidoyl chloride intermediate. This step is critical and must be conducted under strictly anhydrous and oxygen-free conditions, typically under a nitrogen atmosphere, to prevent hydrolysis of the sensitive intermediates. The choice of solvent plays a pivotal role here; while tetrahydrofuran (THF) ensures homogeneity, diethyl ether often promotes faster reaction kinetics, sometimes leading to the precipitation of the intermediate which can be redissolved by adding THF before the next step. This nuanced control over solubility and reaction rate is a key factor in achieving the high isolation yields reported in the experimental examples, ranging significantly based on the steric and electronic nature of the substituents.
Following the formation of the activated intermediate, the addition of a primary amine triggers the cyclization event that constructs the imidazoline-4,5-dione core. The amine attacks the electrophilic center, displacing chloride and facilitating ring closure to form the five-membered heterocyclic system. Simultaneously, the organic base added to the mixture scavenges the stoichiometric amount of hydrogen chloride generated during the acylation and cyclization steps, driving the equilibrium towards product formation. This tandem acylation-cyclization process is remarkably efficient, as evidenced by the structural diversity achieved in the patent examples. For instance, the synthesis of compound IVa demonstrates the compatibility of bulky cyclohexyl and phenyl groups, while other examples showcase the tolerance for electron-withdrawing groups like cyano and acetyl moieties. 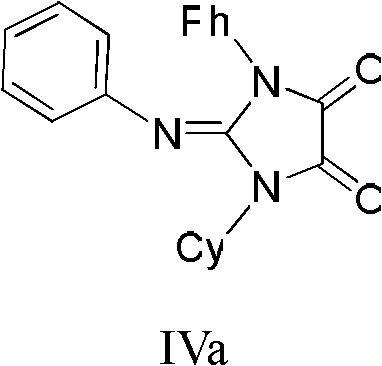 This mechanistic robustness ensures that impurities are minimized, as the reaction pathway is direct and avoids the formation of complex side products often seen in oxidation-based routes. The ability to fine-tune the reaction by selecting specific amines and carbodiimides provides chemists with precise control over the final molecular architecture, essential for optimizing the biological activity of the resulting agrochemical or pharmaceutical candidates.
This mechanistic robustness ensures that impurities are minimized, as the reaction pathway is direct and avoids the formation of complex side products often seen in oxidation-based routes. The ability to fine-tune the reaction by selecting specific amines and carbodiimides provides chemists with precise control over the final molecular architecture, essential for optimizing the biological activity of the resulting agrochemical or pharmaceutical candidates.
How to Synthesize Multi-Substituted 2-Imidoimidazoline-4,5-Diketones Efficiently
Implementing this synthesis protocol requires careful attention to reaction conditions, particularly regarding moisture exclusion and reagent stoichiometry, to ensure optimal outcomes. The process begins by dissolving the substituted carbodiimide in a dry ether solvent, followed by the slow addition of oxalyl chloride, maintaining the temperature between room temperature and the solvent's boiling point. Once the initial activation is complete, monitored by the disappearance of the starting carbodiimide, the amine component and the organic base are introduced to the reaction vessel. The mixture is then stirred for a period typically ranging from 10 to 12 hours to ensure complete conversion to the cyclic diketone. Post-reaction processing is straightforward, involving the concentration of the reaction liquor followed by purification via silica gel chromatography using a petroleum ether and ethyl acetate gradient.  For detailed operational parameters and specific molar ratios tailored to different substrates, please refer to the standardized guide below.
For detailed operational parameters and specific molar ratios tailored to different substrates, please refer to the standardized guide below.
- React substituted carbodiimide with oxalyl chloride in an ether solvent (e.g., diethyl ether or THF) under anhydrous and oxygen-free conditions at room temperature.
- Add a primary amine and an organic base (such as triethylamine) to the reaction mixture to facilitate cyclization and neutralize generated hydrogen chloride.
- Separate the final multi-substituted 2-iminoimidazoline-4,5-dione product via concentration and chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this synthesis method offers profound benefits that extend beyond mere chemical efficiency, directly impacting the bottom line and supply chain resilience. The elimination of mercury-based oxidants removes a major regulatory liability, simplifying the environmental compliance landscape for manufacturing facilities and reducing the overhead costs associated with hazardous waste management. This shift towards greener chemistry aligns perfectly with the sustainability goals of modern multinational corporations, enhancing the marketability of the final products. Furthermore, the use of commodity chemicals such as oxalyl chloride, triethylamine, and various carbodiimides ensures a stable and cost-effective raw material supply base. These reagents are produced on a massive global scale, mitigating the risk of supply disruptions that often plague specialty reagent markets. Consequently, procurement managers can negotiate more favorable pricing terms and secure long-term supply contracts with greater confidence.
- Cost Reduction in Manufacturing: The economic advantages of this route are driven primarily by the simplification of the process workflow and the removal of expensive, toxic reagents. By avoiding the use of mercury acetate, manufacturers eliminate the need for specialized heavy metal removal steps and the associated costly disposal fees, leading to substantial cost savings in the overall production budget. Additionally, the high isolated yields reported across various examples, often exceeding 80% and reaching up to 97% for certain derivatives, mean that less raw material is wasted per unit of product produced. This improved atom economy translates directly into lower variable costs, allowing suppliers to offer more competitive pricing to their downstream clients in the agrochemical and pharmaceutical sectors without sacrificing margin.
- Enhanced Supply Chain Reliability: The reliance on widely available, shelf-stable reagents significantly de-risks the supply chain compared to methods requiring bespoke or unstable intermediates. Carbodiimides and oxalyl chloride are standard inventory items for most fine chemical producers, ensuring that production schedules can be maintained even during periods of market volatility. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require cryogenic cooling or high-pressure equipment, further enhances operational reliability. This flexibility allows manufacturing partners to utilize existing infrastructure without needing capital-intensive retrofits, thereby shortening the lead time for scaling up production to meet surging demand for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process from the bench to the plant is facilitated by the homogeneous nature of the reaction mixture and the simplicity of the workup procedure. The absence of heavy metal sludge simplifies the effluent treatment process, making it easier for facilities to meet strict environmental discharge standards. This environmental compatibility is increasingly becoming a prerequisite for doing business with top-tier global companies who enforce rigorous supplier codes of conduct regarding sustainability. Moreover, the versatility of the method allows for the production of a wide portfolio of derivatives on the same production line, maximizing asset utilization and providing a buffer against fluctuations in demand for specific single products.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this technology into their supply chains, we have compiled answers to common inquiries based on the patent specifications. These responses address critical aspects regarding safety, scalability, and product quality, reflecting the practical realities of implementing this synthesis in a commercial setting. Understanding these details is crucial for R&D directors planning pilot runs and procurement officers assessing vendor capabilities. The following insights are derived directly from the experimental data and technical disclosures within the patent documentation.
Q: What are the environmental advantages of this synthesis method compared to traditional routes?
A: Unlike traditional methods that utilize toxic mercury acetate oxidants, this novel pathway employs oxalyl chloride and carbodiimides, eliminating heavy metal contamination and significantly reducing hazardous waste treatment costs.
Q: What is the typical yield and purity achievable with this protocol?
A: Experimental data indicates isolated yields ranging from 54% to 97% depending on the substituents, with product purity consistently exceeding 98% after standard silica gel column chromatography.
Q: Is this method suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes readily available commercial reagents, operates at mild temperatures (room temperature to solvent boiling point), and avoids complex equipment, making it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Imidoimidazoline-4,5-Diketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis method described in CN102532030A for producing high-value heterocyclic intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market launch is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-iminoimidazoline-4,5-dione derivatives meets the exacting standards required for pharmaceutical and agrochemical applications. We understand that consistency and quality are non-negotiable in your supply chain, and our commitment to process excellence ensures reliable delivery of these critical building blocks.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact our technical procurement team today to request specific COA data for our available catalog compounds or to discuss route feasibility assessments for your proprietary derivatives. Together, we can drive innovation and efficiency in the production of next-generation bioactive molecules.
