Scalable Electrocatalytic Synthesis of C2-Acyloxy-3-Indolinone Derivatives for Oncology Drug Development
Introduction to Advanced Electrocatalytic Synthesis
The pharmaceutical industry is constantly seeking greener, more efficient pathways to construct complex heterocyclic scaffolds essential for modern oncology therapeutics. Patent CN114057625A introduces a groundbreaking methodology for the synthesis of C2-acyloxy-3-indolinone derivatives, a privileged structural motif found in potent antitumor agents such as mitomycin C analogs. This technology leverages electrocatalysis to forge carbon-oxygen bonds directly, bypassing the need for traditional stoichiometric oxidants and expensive transition metal catalysts. By utilizing electrical energy as a clean reagent, this approach aligns perfectly with the principles of green chemistry while delivering high-purity intermediates crucial for drug development. The general structure of these valuable derivatives is depicted below, showcasing the versatile substitution patterns achievable through this novel route.
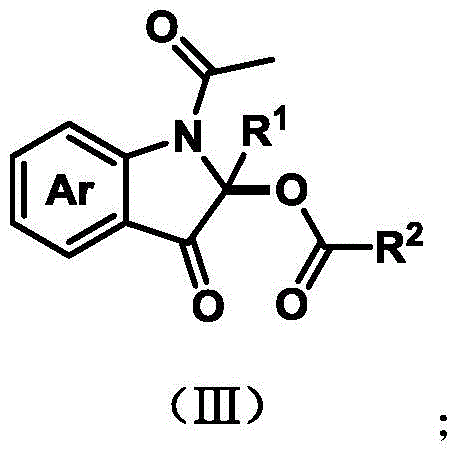
For R&D directors and process chemists, the ability to access these scaffolds without metal contamination is a significant advantage. The patent details a robust protocol where N-acetyl-3-hydroxyindole compounds undergo oxidative coupling with diverse carboxylic acids. This transformation is not merely a laboratory curiosity but represents a viable industrial strategy for producing high-value pharmaceutical intermediates. The method ensures that the resulting compounds maintain stringent purity specifications, which is paramount when synthesizing candidates for biological evaluation against cancer cell lines like HeLa, MCF-7, and HCT116.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of C-O bonds at the C2 position of indolinone systems has relied heavily on cross-dehydrogenative coupling (CDC) reactions that require stoichiometric amounts of harsh chemical oxidants or noble metal catalysts. These conventional pathways often involve toxic reagents, generate substantial hazardous waste, and necessitate rigorous downstream processing to remove trace metal residues that could compromise the safety profile of the final drug substance. Furthermore, the use of external oxidants can lead to over-oxidation side reactions, reducing overall yield and complicating the impurity profile. For procurement managers, the reliance on precious metals like palladium or rhodium introduces volatility in raw material costs and supply chain risks associated with geopolitical sourcing issues.
The Novel Approach
In stark contrast, the electrochemical strategy outlined in the patent utilizes a bromide-mediated anodic oxidation system to drive the coupling reaction. As illustrated in the reaction scheme below, the process combines an N-acetyl-3-hydroxyindole substrate with a carboxylic acid in the presence of a simple bromide salt electrolyte. Electricity serves as the terminal oxidant, regenerating the active bromine species in situ without consuming additional chemical reagents. This eliminates the need for external oxidants entirely and avoids the introduction of transition metals. The reaction proceeds under mild conditions, typically at room temperature, which preserves sensitive functional groups and enhances the overall safety profile of the manufacturing process.
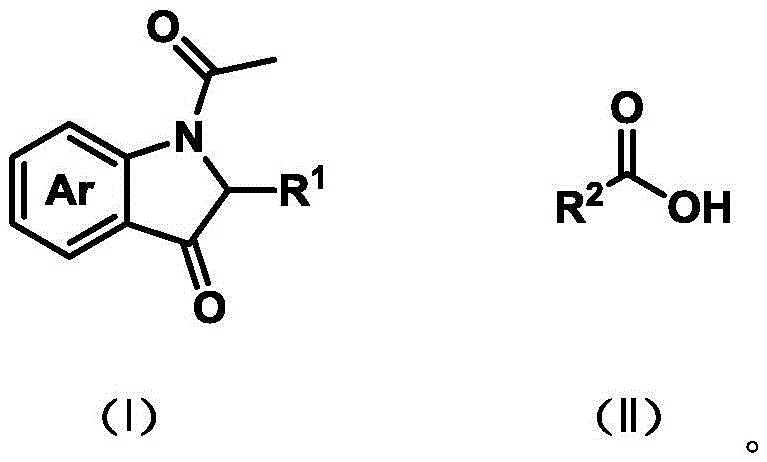
This shift from chemical oxidation to electrochemical oxidation represents a paradigm shift in process efficiency. By removing the dependency on expensive catalysts and hazardous oxidants, the novel approach drastically simplifies the workup procedure. The absence of metal catalysts means that costly scavenging steps are no longer required, leading to significant operational savings. Additionally, the mild reaction conditions allow for a broader substrate scope, enabling the synthesis of complex derivatives that might decompose under the harsh thermal or oxidative conditions of traditional methods. This flexibility is critical for medicinal chemists exploring structure-activity relationships in new drug discovery programs.
Mechanistic Insights into Electrocatalytic C-O Bond Coupling
The core of this technology lies in the elegant electrochemical cycle that facilitates the formation of the C-O bond. At the anode, bromide ions from the electrolyte are oxidized to generate reactive bromine radicals or cationic species. These active bromine intermediates then interact with the N-acetyl-3-hydroxyindole substrate, likely forming a transient radical species at the C2 position. Simultaneously, the carboxylic acid component is activated, allowing for a radical-radical coupling or a nucleophilic attack that establishes the new ester linkage. Crucially, the bromide species is regenerated at the end of the cycle, acting as a redox mediator rather than a consumable reagent. This catalytic turnover ensures that only a small amount of electrolyte is needed to drive the transformation of large quantities of substrate.
From an impurity control perspective, this mechanism offers superior cleanliness compared to metal-catalyzed alternatives. Since no transition metals are introduced into the reaction matrix, the risk of metal-catalyzed side reactions such as homocoupling or decomposition is minimized. The primary byproducts are typically hydrogen gas evolved at the cathode and minor amounts of unreacted starting materials, which are easily separated during the standard aqueous workup. This clean reaction profile translates directly to higher isolated yields, with the patent reporting efficiencies often exceeding 90% for various substrates. For quality assurance teams, this means a more consistent and predictable impurity profile, facilitating faster regulatory approval processes for new drug applications.
How to Synthesize C2-Acyloxy-3-Indolinone Efficiently
Implementing this electrochemical synthesis requires careful attention to cell configuration and current density to maximize efficiency. The patent provides detailed experimental procedures that serve as a robust foundation for scaling up. The process begins with the preparation of the electrolytic solution, followed by the application of a constant current across platinum electrodes. The simplicity of the setup allows for easy adaptation to flow chemistry reactors, which can further enhance mass transfer and reaction rates. Below is a summary of the standardized synthesis steps derived from the patent examples, designed to guide process engineers in replicating these high-yielding transformations.
- Prepare the electrolytic cell by charging a three-necked flask with N-acetyl-3-hydroxyindole, carboxylic acid substrate, and a bromide electrolyte such as tetrabutylammonium bromide in acetonitrile.
- Initiate constant current electrolysis using platinum electrodes at room temperature, maintaining a current density between 5 mA and 20 mA for approximately 7 to 15 hours.
- Quench the reaction mixture with ethyl acetate, perform aqueous workup to separate organic layers, dry over sodium sulfate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical methodology offers tangible strategic benefits beyond mere technical novelty. The elimination of precious metal catalysts removes a major cost driver and supply bottleneck from the manufacturing equation. Traditional palladium or copper catalysts are subject to significant price fluctuations and require specialized handling and disposal protocols. By replacing these with inexpensive bromide salts and electricity, the overall cost of goods sold (COGS) for these intermediates can be substantially reduced. Furthermore, the simplified purification process reduces solvent consumption and waste generation, aligning with corporate sustainability goals and reducing environmental compliance costs.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and stoichiometric oxidants leads to direct material cost savings. Additionally, the simplified workup procedure reduces the consumption of solvents and adsorbents typically used for metal scavenging. This streamlined process lowers the operational expenditure associated with waste treatment and disposal, contributing to a more economically viable production model for high-volume pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reagents required for this electrochemical process, such as acetonitrile, bromide salts, and simple carboxylic acids, are commodity chemicals with stable and robust global supply chains. Unlike specialized ligands or organometallic complexes that may have single-source suppliers, these basic materials are readily available from multiple vendors. This diversification of supply sources mitigates the risk of production delays caused by raw material shortages, ensuring consistent delivery schedules for downstream drug manufacturers.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of hazardous oxidants make this process inherently safer and easier to scale. Electrochemical reactors can be efficiently scaled out by adding more cells or scaled up using continuous flow technology, which offers precise control over reaction parameters. The green nature of the process, characterized by atom economy and reduced waste, simplifies regulatory compliance regarding environmental emissions and worker safety, facilitating smoother audits and inspections.
Frequently Asked Questions (FAQ)
Understanding the practical implications of this technology is essential for stakeholders evaluating its adoption. The following questions address common concerns regarding the scalability, safety, and versatility of the electrochemical synthesis method. These answers are derived directly from the technical disclosures in the patent, providing a reliable basis for decision-making. Whether you are concerned about equipment requirements or substrate compatibility, the following insights clarify the operational realities of implementing this green chemistry solution.
Q: Does this electrochemical method eliminate heavy metal contamination risks?
A: Yes, the process described in patent CN114057625A operates without transition metal catalysts like palladium or copper, significantly reducing the risk of heavy metal residues in the final API intermediate and simplifying purification protocols.
Q: What is the substrate scope for the carboxylic acid component?
A: The method demonstrates excellent functional group tolerance, successfully coupling with various substituted benzoic acids, heteroaryl carboxylic acids, and even complex drug molecules like ibuprofen and naproxen derivatives.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The reaction utilizes inexpensive reagents, mild room temperature conditions, and avoids hazardous stoichiometric oxidants, making it highly adaptable for commercial scale-up and continuous flow electrochemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C2-Acyloxy-3-Indolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modern pharmaceutical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle electrochemical processes safely and efficiently, ensuring that your project benefits from the latest advancements in green chemistry. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of C2-acyloxy-3-indolinone intermediate meets the highest quality standards required for clinical and commercial applications.
We invite you to collaborate with us to leverage this innovative technology for your drug development pipeline. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how this metal-free route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your timeline to market with a sustainable, cost-effective, and reliable supply of critical pharmaceutical intermediates.
