Revolutionizing Peptide Synthesis with Diphenylmethane Carriers for Commercial Scale-up
The landscape of peptide manufacturing is undergoing a significant transformation driven by the innovations disclosed in patent CN110194724B, which introduces a novel class of compounds containing a diphenylmethane structure designed specifically for use as C-terminal protecting reagents. This technological breakthrough addresses the longstanding inefficiencies associated with traditional solid-phase and conventional liquid-phase synthesis methods by offering a versatile carrier system that operates effectively in both homogeneous and heterogeneous solvent environments. For research and development directors overseeing complex molecule production, the ability to utilize a protective carrier that facilitates rapid reaction speeds and high reagent utilization rates represents a critical advancement in process chemistry. The core innovation lies in the molecular architecture which incorporates hydroxyl, amino, and active groups alongside lipophilic chains, enabling precise control over solubility profiles during different stages of the synthesis cycle. By leveraging this patented technology, manufacturers can achieve higher purity products through simple liquid-liquid extraction separation, thereby eliminating the need for costly and time-consuming preparative chromatography steps that often bottleneck production timelines. This report analyzes the technical merits and commercial implications of adopting these diphenylmethane-based carriers for the reliable supply of high-purity pharmaceutical intermediates.
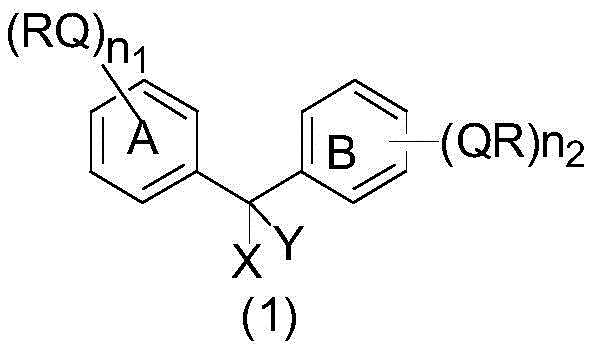
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional solid-phase carrier synthesis methods, while universally applicable, suffer from inherent mass transfer limitations due to the poor affinity between the solvent and the solid phase carrier, which restricts reaction speed and necessitates the use of reactants in large excess to compensate for low reactivity. Furthermore, intermediates carried on a solid phase are difficult to analyze using conventional methods and cannot be purified until the final cleavage step, which typically requires reverse-phase liquid chromatography that is expensive and difficult to scale for commercial production. On the other hand, conventional liquid-phase synthesis offers good reactivity and homogeneous reaction conditions but lacks a universal method for separation and purification because the properties of products and impurities vary significantly at each step. Existing liquid-phase carrier technologies, such as those utilizing long-chain alkoxybenzyl alcohols, often encounter problems with low solubility in medium to low polarity solvents, requiring complex quantitative distillation operations to change solvent composition for precipitation. Additionally, these older methods frequently result in the formation of amorphous solids that are difficult to filter and wash, leading to poor impurity removal effects and prolonged processing times that hinder reproducibility and scalability.
The Novel Approach
The novel approach presented in the patent data utilizes a diphenylmethane structure that is engineered to overcome these solubility and purification challenges by incorporating specific nonpolar and polar structural components that allow dissolution in both nonpolar organic solvents and polar solvents. This dual-solubility characteristic enables the synthesis reaction to proceed in a heterogeneous system composed of hydrocarbons mixed with esters, ethers, or halogenated hydrocarbons alongside an amide polar solvent, facilitating a unique separation mechanism. During the reaction, the system remains homogeneous or manageable heterogeneous, but upon addition of a high-polarity solvent such as water or alcohols during post-treatment, the target product and impurities separate cleanly into different phases. This mechanism allows for effective purification through simple liquid-liquid extraction, drastically simplifying the operation steps and reducing the generation of three wastes compared to traditional chromatographic methods. The result is a universal production method that maintains small solubility changes throughout the synthesis process, ensuring strong versatility and high repeatability for the commercial scale-up of complex peptide intermediates.
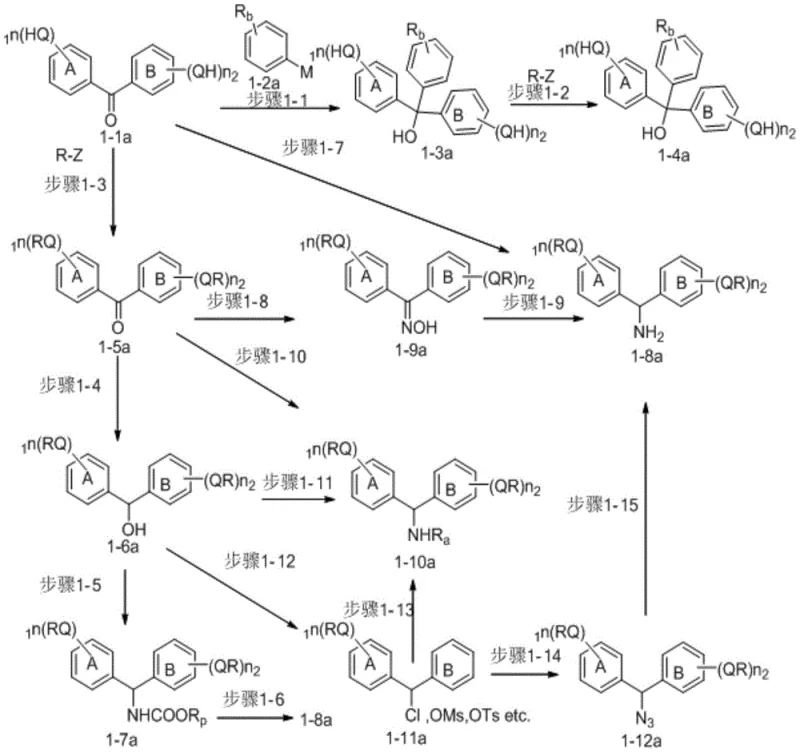
Mechanistic Insights into Diphenylmethane-Catalyzed Peptide Elongation
The mechanistic advantage of this technology stems from the specific substitution patterns on the diphenylmethane core, where groups such as long-chain alkoxy substituents provide the necessary lipophilicity to keep the growing peptide chain soluble in organic media like heptane or isopropyl acetate. As the peptide chain elongates, the carrier prevents the aggregation and gelation phenomena that typically plague liquid-phase synthesis when the amino acid count exceeds four residues, maintaining a consistent reaction environment. The presence of active groups such as hydroxyl or amino functionalities allows for robust coupling with N-protected amino acids using standard condensation reagents like HBTU or EDCI without significant loss of efficiency. Crucially, the solubility switch mechanism is triggered by the polarity difference between the reaction solvent system and the extraction solvent, allowing the carrier-bound peptide to remain in the organic phase while reagents and byproducts partition into the polar phase. This selective partitioning ensures that the intermediate compound experiences minimal solubility changes during the synthetic process, which is vital for maintaining high reagent utilization rates and minimizing the formation of deletion sequences or side products.
Impurity control is inherently built into the solvent system design, where the use of heterogeneous conditions during specific deprotection and coupling steps enhances the kinetic selectivity of the reactions. The patent data indicates that the carrier can be used in amounts that allow for near-stoichiometric usage of protected amino acids, typically ranging from 1 to 1.1 equivalents, which is a significant improvement over the large excesses required in solid-phase synthesis. By avoiding the use of transition metal catalysts or complex resin linkers, the process eliminates the risk of heavy metal contamination, thereby simplifying the downstream purification requirements and ensuring compliance with stringent pharmaceutical quality standards. The stability of the diphenylmethane linkage under the reaction conditions ensures that the C-terminal protection remains intact throughout the elongation cycles, only being cleaved under specific final treatment conditions to release the target peptide with high structural integrity. This level of control over the chemical environment translates directly into a more predictable impurity profile, which is a key concern for regulatory approval and quality assurance in drug substance manufacturing.
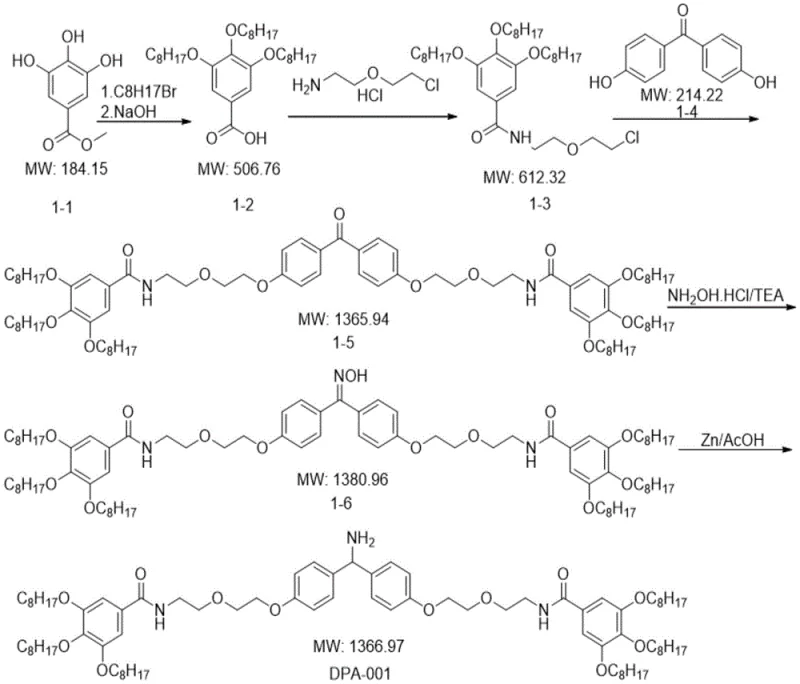
How to Synthesize Diphenylmethane Carriers Efficiently
The synthesis of these advanced carriers begins with the preparation of the core diphenylmethane structure followed by the attachment of lipophilic chains through amidation or etherification reactions to tune the solubility properties. Detailed standardized synthesis steps for specific derivatives such as DPA-001 involve multi-step sequences including alkylation of hydroxybenzoates, amidation with amino-ether linkers, and final reduction of benzophenone intermediates to the amine functionality.
- Preparation of the diphenylmethane core structure through alkylation and amidation reactions using specific lipophilic chains to ensure solubility.
- Coupling the carrier with N-protected amino acids in a homogeneous or heterogeneous solvent system depending on the specific derivative used.
- Executing iterative deprotection and elongation cycles followed by liquid-liquid extraction for purification instead of chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this diphenylmethane carrier technology offers substantial cost savings and operational efficiencies by fundamentally changing the economics of peptide manufacturing. The elimination of expensive solid-phase resins and the reduction in solvent consumption due to the efficiency of liquid-liquid extraction directly contribute to a lower cost of goods sold, making the production of complex peptides more economically viable. The process simplifies the supply chain by reducing reliance on specialized chromatography columns and equipment, allowing for production in standard chemical reactors that are more readily available and easier to maintain. This universality means that manufacturing capacity can be scaled up rapidly without the need for significant capital investment in new infrastructure, ensuring supply continuity even during periods of high market demand. Furthermore, the high purity achieved through this method reduces the risk of batch failures and reprocessing, which are major sources of delay and expense in the pharmaceutical supply chain.
- Cost Reduction in Manufacturing: The process achieves cost reduction in peptide manufacturing by eliminating the need for expensive chromatographic purification steps and reducing the consumption of solvents and reagents through higher utilization rates. By enabling the use of stoichiometric amounts of amino acids rather than large excesses, the raw material costs are significantly optimized, and the waste disposal costs are lowered due to the reduced volume of hazardous waste generated. The simplified workup procedure involving extraction rather than filtration of gels or resins also reduces labor hours and energy consumption associated with drying and solvent recovery. These cumulative efficiencies result in a more competitive pricing structure for the final active pharmaceutical ingredients without compromising on quality or regulatory compliance standards.
- Enhanced Supply Chain Reliability: Supply chain reliability is enhanced because the raw materials required for synthesizing the carriers and the peptides are commercially available and do not rely on single-source proprietary resins that can cause bottlenecks. The robustness of the chemical process against variations in reaction conditions means that batch-to-batch consistency is high, reducing the likelihood of supply disruptions caused by quality deviations. Additionally, the ability to use common industrial solvents like heptane and isopropyl acetate ensures that solvent supply risks are minimized compared to specialized solvent systems required by other technologies. This stability allows for better long-term planning and inventory management, ensuring that critical peptide intermediates are available when needed for downstream drug product formulation.
- Scalability and Environmental Compliance: Scalability is a core strength of this technology as the liquid-phase nature of the reaction allows for easy transfer from laboratory glassware to large-scale stainless steel reactors without the mass transfer limitations of solid supports. The environmental compliance aspect is improved through the reduction of solid waste associated with spent resins and the ability to recycle organic solvents more effectively in a liquid-liquid extraction workflow. The process aligns with green chemistry principles by minimizing the use of hazardous reagents and reducing the overall environmental footprint of the manufacturing facility. This makes it an attractive option for companies looking to meet increasingly stringent environmental regulations while expanding their production capacity for high-value peptide therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of diphenylmethane carriers in peptide synthesis workflows based on the patent specifications.
Q: How does the diphenylmethane carrier improve purification compared to solid-phase synthesis?
A: Unlike solid-phase synthesis which requires extensive washing and often preparative chromatography, this carrier allows for simple liquid-liquid extraction due to its tunable solubility in hydrocarbon and polar solvent systems, significantly reducing processing time and waste.
Q: What are the solubility characteristics of these carriers during peptide elongation?
A: The compounds maintain consistent solubility throughout the synthesis process, avoiding the gelation phenomena often seen in other liquid-phase carriers when the peptide chain exceeds four amino acids, thus ensuring reproducible reaction kinetics.
Q: Is this technology suitable for large-scale commercial production?
A: Yes, the universal production method described in patent CN110194724B is designed for scalability, utilizing common organic solvents and standard extraction techniques that are easily adaptable from laboratory to industrial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diphenylmethane Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production for complex pharmaceutical intermediates. Our technical team is fully equipped to leverage the diphenylmethane carrier technology to deliver high-purity peptide fragments with stringent purity specifications verified by our rigorous QC labs. We understand the critical nature of supply chain continuity and are committed to providing a reliable diphenylmethane derivatives supplier partnership that ensures your project timelines are met with precision. Our facilities are designed to handle the specific solvent systems and extraction processes required by this technology, guaranteeing that the commercial advantages identified in the patent are fully realized in our production outputs.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific peptide sequence requirements. By engaging with us, you can obtain specific COA data and route feasibility assessments that demonstrate how this innovative chemistry can be integrated into your existing supply chain. Let us help you optimize your manufacturing process and secure a competitive edge in the global pharmaceutical market through advanced chemical solutions.
