Revolutionizing Peptide Manufacturing with Novel Benzyl Structure Liquid Phase Carriers
Revolutionizing Peptide Manufacturing with Novel Benzyl Structure Liquid Phase Carriers
The landscape of peptide synthesis is undergoing a significant transformation driven by the innovations disclosed in patent CN110183347B. This pivotal intellectual property introduces a novel class of compounds containing a benzyl structure that function as highly efficient C-terminal protective reagents for liquid phase peptide synthesis. Unlike traditional methods that struggle with mass transfer limitations or complex purification workflows, these benzyl-based carriers offer a unique balance of solubility and reactivity. They are engineered to maintain high reaction speeds and reagent utilization rates within appropriate solvent systems, effectively bridging the gap between the convenience of solid-phase synthesis and the analytical control of conventional liquid-phase methods. By incorporating hydroxyl, amino, or substituted amino active groups, these compounds facilitate robust coupling reactions while enabling simplified post-treatment through liquid-liquid extraction. This technological breakthrough allows manufacturers to achieve superior product purity with reduced operational complexity, marking a substantial leap forward for the production of high-value pharmaceutical intermediates and therapeutic peptides.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthetic production of peptides has been dominated by solid-phase carrier synthesis and conventional liquid-phase synthesis, both of which present distinct operational bottlenecks for large-scale manufacturing. Solid-phase synthesis, while universal, suffers from serious mass transfer problems due to poor affinity between solvents and the solid carrier, often requiring reactants to be used in large excess to compensate for low reactivity. Furthermore, intermediates attached to solid supports are difficult to analyze using conventional techniques, and final purification typically relies on expensive and time-consuming reverse-phase preparative chromatography. On the other hand, existing liquid-phase carrier technologies, such as those utilizing long-chain alkoxybenzyl alcohols, often face challenges with low solubility in medium-low polarity solvents like acetate or toluene. This limitation restricts reaction concentrations and hinders amplification production. Additionally, previous liquid-phase methods frequently require complex quantitative distillation to change solvent composition for precipitation, and the resulting amorphous solids are notoriously difficult to filter and wash, leading to poor impurity removal and extended processing times.
The Novel Approach
The novel approach presented in this patent overcomes these historical hurdles by introducing benzyl structure compounds specifically designed for versatile solvent compatibility. These carriers possess a unique molecular architecture that ensures high solubility in hydrocarbon organic solvents, aromatic hydrocarbons, esters, and ethers, while remaining compatible with polar aprotic solvents. This dual-solubility characteristic enables the synthesis to proceed in either homogeneous or heterogeneous solvent systems, with a particular advantage in heterogeneous systems formed by mixing hydrocarbons with amide polar solvents. In this configuration, the reaction speed is significantly enhanced, and by-product formation is minimized. Crucially, the post-treatment process is drastically simplified; target products and impurities can be effectively separated merely by adding a high-polarity solvent for liquid-liquid extraction, eliminating the need for complex precipitation or filtration steps. This universality in operation flow allows for the development of a general production method that maintains consistent solubility and reaction rates throughout the peptide chain elongation process, avoiding the gelation issues that plague other liquid-phase carriers when synthesizing longer chains.
Mechanistic Insights into Benzyl Structure Carrier Synthesis and Function
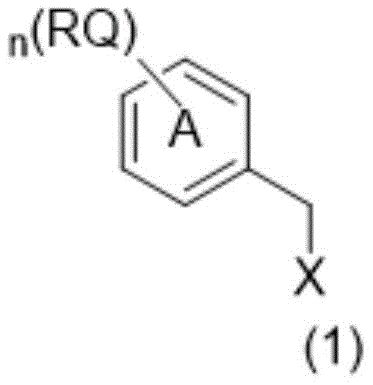
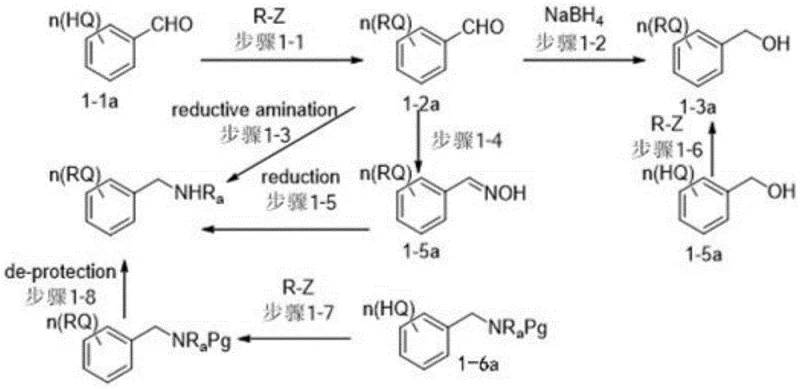
The chemical efficacy of these benzyl structure carriers stems from their carefully balanced amphiphilic properties and reactive functional groups. The core structure, as defined by general formula (1), incorporates a benzyl moiety linked to various active groups (X) such as hydroxyl, halogen, sulfonate, or amino derivatives. The presence of long-chain alkoxy substituents (R groups) on the aromatic ring provides the necessary lipophilicity to ensure dissolution in non-polar solvents like heptane or isopropyl acetate. This solubility profile is critical for creating the heterogeneous reaction systems that drive the efficiency of the process. When used as a C-terminal protective reagent, the carrier forms an ester or amide bond with the incoming amino acid. The mechanistic advantage lies in the stability of this linkage during the repetitive deprotection and coupling cycles, combined with the ease of cleavage at the final stage. The synthesis of the carriers themselves involves straightforward alkylation and condensation reactions, starting from hydroxybenzaldehydes or benzoic acid derivatives. For instance, the conversion of aldehyde intermediates to the final alcohol carriers via reduction with sodium borohydride ensures high yields and purity. This robust synthetic pathway allows for the precise tuning of the alkyl chain lengths (e.g., isooctyl, isononyl, isotridecyl) to optimize solubility parameters for specific peptide sequences.
Impurity control is inherently built into the physical chemistry of these carriers. During the peptide synthesis cycle, specifically the N-terminal deprotection and chain elongation steps, the reaction mixture can be manipulated to form a biphasic system. By selecting a solvent system where the carrier-bound peptide resides primarily in the organic phase while protecting group by-products and excess reagents partition into the polar phase, effective purification is achieved without chromatography. The patent data indicates that compounds like BM-001 and BM-002 maintain their solubility characteristics even as the peptide chain grows, preventing the precipitation or gelation that leads to incomplete reactions and truncated sequences in other methods. This consistent physical state ensures that every coupling step proceeds with high efficiency, minimizing the accumulation of deletion sequences and other impurities. The result is a final crude peptide with significantly higher purity, often exceeding 90% even before final global deprotection and purification, which translates to reduced burden on downstream processing units.
How to Synthesize Benzyl Structure Carriers Efficiently
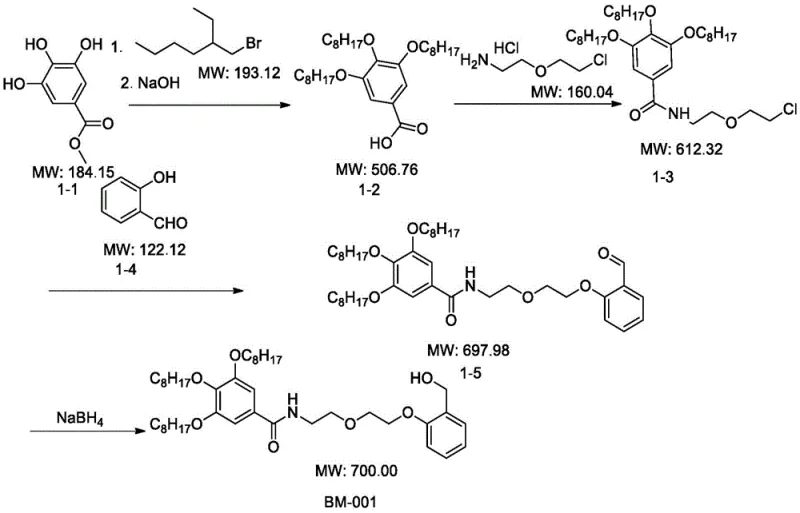
The preparation of these advanced peptide synthesis carriers follows a logical and scalable chemical pathway that leverages readily available starting materials. The process typically begins with the alkylation of hydroxybenzoic acid derivatives or hydroxybenzaldehydes using long-chain alkyl halides to introduce the necessary lipophilic tails. Subsequent functional group transformations, such as amidation with amino-alcohol linkers and reduction of aldehyde groups to alcohols, yield the final active carriers. The versatility of the synthetic route allows for the creation of a diverse library of carriers (e.g., BM-001 through BM-036) by varying the alkyl chain length and the nature of the linker. This modularity is essential for tailoring the carrier properties to specific peptide synthesis requirements. For detailed operational parameters, stoichiometry, and specific reaction conditions tailored to your target molecule, please refer to the standardized synthesis guide below.
- Carrier Access: Connect the benzyl structure compound with an N-protected amino acid or peptide via conventional condensation reactions to form the C-terminal protected intermediate.
- N-Terminal Deprotection: Dissolve the protected intermediate in a hydrocarbon solvent, add a deprotection reagent solution to form a heterogeneous system, and extract impurities using a high-polarity solvent.
- Peptide Chain Elongation: Add the next N-protected amino acid and condensation reagent to the solution, react, and purify via liquid-liquid extraction before repeating the cycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of these benzyl structure carriers represents a strategic opportunity to optimize manufacturing economics and operational reliability. The primary economic driver is the significant reduction in processing complexity and resource consumption. By enabling purification through simple liquid-liquid extraction rather than costly chromatography or difficult filtration of amorphous solids, the technology drastically lowers the cost of goods sold (COGS). The elimination of complex solvent switching procedures and the ability to operate at higher concentrations in cost-effective hydrocarbon solvents further contribute to substantial cost savings in pharmaceutical intermediate manufacturing. Moreover, the high reagent utilization rate means that expensive protected amino acids and coupling agents are not wasted in large excess, directly improving material efficiency. From a supply chain perspective, the robustness of the synthesis method enhances reliability. The avoidance of gelation and precipitation issues ensures consistent batch-to-batch performance, reducing the risk of production delays caused by failed runs or difficult workups. This consistency is vital for maintaining continuous supply lines for critical peptide therapeutics.
- Cost Reduction in Manufacturing: The implementation of this technology eliminates the need for expensive transition metal catalysts and complex purification equipment associated with traditional methods. The shift towards liquid-liquid extraction as the primary purification method reduces solvent consumption and waste disposal costs significantly. Furthermore, the ability to recycle solvents more effectively in a heterogeneous system contributes to a leaner manufacturing process. The overall simplification of the workflow reduces labor hours and facility occupancy time, leading to a more favorable cost structure for high-volume production.
- Enhanced Supply Chain Reliability: The chemical stability and solubility profile of these carriers ensure that raw material availability is not a bottleneck, as the precursors are commodity chemicals. The process is less sensitive to minor variations in reaction conditions compared to solid-phase synthesis, which enhances the robustness of the supply chain against operational fluctuations. This reliability allows for more accurate forecasting and inventory management, ensuring that downstream customers receive their orders on time without quality disputes.
- Scalability and Environmental Compliance: The technology is inherently designed for scale-up, moving seamlessly from laboratory gram-scale to multi-ton commercial production without fundamental changes to the process logic. The use of greener solvent systems, such as hydrocarbons and esters, aligns with increasingly stringent environmental regulations regarding volatile organic compounds (VOCs) and hazardous waste. The reduction in three wastes (waste water, waste gas, waste residue) due to higher atom economy and simpler workups supports corporate sustainability goals and regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of benzyl structure carriers in peptide synthesis workflows. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these aspects is crucial for R&D teams evaluating the feasibility of integrating this technology into their existing manufacturing platforms. The answers provide clarity on performance metrics, compatibility with standard protocols, and the tangible benefits observed during pilot-scale evaluations.
Q: How do benzyl structure carriers improve peptide synthesis purity compared to solid phase methods?
A: Unlike solid phase synthesis which suffers from mass transfer limitations and difficult intermediate analysis, benzyl structure carriers operate in liquid phase systems allowing for homogeneous or heterogeneous reactions. This enables simple liquid-liquid extraction for purification at every step, significantly removing impurities and yielding higher final purity without complex chromatography.
Q: What are the solubility characteristics of these benzyl carriers in common organic solvents?
A: These compounds exhibit excellent solubility in low to medium polarity solvents such as heptane, isopropyl acetate, and methyl tert-butyl ether, while maintaining sufficient solubility in polar aprotic solvents like DMF. This dual solubility profile facilitates flexible reaction conditions and efficient phase separation during workup.
Q: Can these carriers be scaled for industrial production of long peptide chains?
A: Yes, the design prevents the gelation phenomena often seen in other liquid phase carriers when peptide chains exceed four amino acids. The consistent solubility throughout the synthesis process ensures robust scalability and reproducibility for commercial manufacturing of complex peptides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzyl Structure Compound Supplier
As the demand for high-purity peptide therapeutics continues to surge, partnering with a knowledgeable and capable CDMO is essential for translating innovative chemistry into commercial reality. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team possesses deep expertise in the synthesis and application of complex benzyl structure carriers, ensuring that your peptide projects benefit from the highest standards of quality and efficiency. We understand that stringent purity specifications are non-negotiable in the pharmaceutical industry, which is why our rigorous QC labs employ state-of-the-art analytical methods to verify the identity and purity of every batch. Whether you require custom synthesis of specific carrier variants or full-scale peptide manufacturing services, our infrastructure is designed to support your growth and innovation.
We invite you to explore the potential of this advanced technology for your next project. By leveraging our capabilities, you can accelerate your development timelines and achieve superior cost efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific peptide sequence. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our benzyl structure carriers can transform your manufacturing process. Let us collaborate to build a more efficient and reliable supply chain for your critical pharmaceutical intermediates.
