Revolutionizing Peptide Synthesis: Scalable Fluorene Ring Carriers for High-Purity Pharmaceutical Intermediates
Revolutionizing Peptide Synthesis: Scalable Fluorene Ring Carriers for High-Purity Pharmaceutical Intermediates
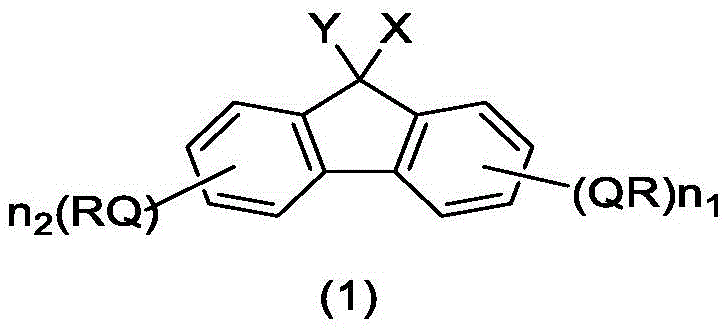
The landscape of peptide manufacturing is undergoing a significant transformation driven by the innovations disclosed in patent CN110256277B. This pivotal intellectual property introduces a novel class of compounds containing a fluorene ring structure, specifically engineered to serve as superior liquid-phase carriers for peptide synthesis. Unlike traditional methods that struggle with the trade-off between reaction efficiency and purification ease, these fluorene-based carriers offer a unique dual-solubility profile. By incorporating specific hydroxyl, amino, and substituted amino groups alongside tailored lipophilic chains, the technology enables high-speed reactions in homogeneous systems while allowing for straightforward product isolation via liquid-liquid extraction in heterogeneous systems. This breakthrough addresses critical bottlenecks in the production of high-purity pharmaceutical intermediates, offering a robust pathway for the commercial scale-up of complex peptide drugs.
For procurement managers and supply chain directors, the implications of this technology extend far beyond mere chemical novelty. The ability to utilize common, cost-effective solvents such as heptane, isopropyl acetate, and methyl tert-butyl ether drastically simplifies the manufacturing infrastructure required. Traditional liquid-phase carriers often necessitate complex solvent switching or quantitative distillation to induce precipitation, operations that are energy-intensive and difficult to control on a multi-ton scale. In contrast, the fluorene carriers described in this patent maintain stable solubility characteristics throughout the synthesis process, preventing the gelation phenomena that plague earlier generations of liquid supports. This stability ensures a reliable peptide synthesis carrier supplier can deliver consistent batch-to-batch quality, minimizing the risk of production delays and ensuring a steady flow of critical API intermediates to the market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The historical reliance on solid-phase peptide synthesis (SPPS) has long been challenged by inherent mass transfer limitations. In solid-liquid two-phase reactions, the affinity between the solvent and the solid carrier is often poor, leading to serious diffusion barriers that cap reaction speeds. To compensate, manufacturers are forced to use reactants in large excess, driving up raw material costs and generating substantial chemical waste. Furthermore, intermediates bound to solid supports are notoriously difficult to analyze using conventional techniques, and final purification typically requires expensive reverse-phase preparative chromatography. On the other hand, earlier liquid-phase carrier technologies, such as those utilizing long-chain alkoxybenzyl alcohols, introduced their own set of operational headaches. These carriers frequently exhibited low solubility in medium-to-low polarity solvents, forcing reactions to proceed at low concentrations that hindered amplification production. Additionally, the separation processes often involved changing solvent compositions via distillation, a complex procedure that increased cycle times and operational risks.
The Novel Approach
The fluorene ring structure compounds presented in this patent represent a paradigm shift by effectively merging the kinetic advantages of solution chemistry with the separation benefits of solid-phase methods. The core innovation lies in the molecular design, which balances polar and non-polar domains to ensure compatibility with a wide range of solvent systems. This design allows the synthesis to proceed in a homogeneous phase for maximum reaction efficiency, followed by a transition to a heterogeneous system simply by adding a high-polarity solvent or adjusting the solvent ratio. This triggers a clean phase separation where the product remains in the organic layer while impurities are washed away into the polar phase. Consequently, the post-treatment operation is vastly simplified, eliminating the need for complex precipitation protocols or difficult filtration of amorphous solids. The result is a universal production method that delivers products with significantly higher purity and improved universality across different peptide sequences.
Mechanistic Insights into Fluorene-Mediated Liquid Phase Synthesis
The efficacy of these fluorene-based carriers is rooted in their sophisticated amphiphilic architecture. The molecule features a rigid fluorene core that provides structural stability, linked to flexible, long-chain alkyl or alkoxy groups (ranging from C8 to C25) that dictate solubility behavior. This structural arrangement allows the carrier to dissolve readily in non-polar hydrocarbon solvents like heptane during the coupling and deprotection steps, ensuring that the growing peptide chain remains accessible to reagents. Crucially, the presence of polar functional groups such as hydroxyl or amino moieties at the 9-position of the fluorene ring facilitates interaction with polar solvents when needed for separation. This duality prevents the "gelation" effect observed in other liquid carriers, where the growing peptide chain causes the support to aggregate and become insoluble, halting the reaction. By maintaining a consistent solution state or forming a manageable heterogeneous mixture, the carrier ensures that reaction kinetics remain fast and reproducible throughout the entire elongation of the peptide chain.
From an impurity control perspective, the mechanism leverages the distinct partition coefficients of the carrier-bound peptide versus small molecule by-products. In the heterogeneous solvent system described, typically comprising a mixture of hydrocarbons and amide polar solvents, the lipophilic carrier-bound peptide preferentially resides in the organic phase. Meanwhile, excess reagents, urea by-products from condensation agents, and cleaved protecting groups are efficiently extracted into the polar aqueous or alcoholic phase. This continuous "wash-and-react" capability inherent to the liquid carrier system means that impurities do not accumulate on the support as they might in solid-phase synthesis. The ability to perform rigorous liquid-liquid extractions after every coupling and deprotection step results in intermediates of exceptional purity before the final cleavage. This mechanistic advantage translates directly to a cleaner final product, reducing the burden on downstream purification processes and increasing the overall yield of the target pharmaceutical intermediate.
How to Synthesize Fluorene-Based Carriers Efficiently
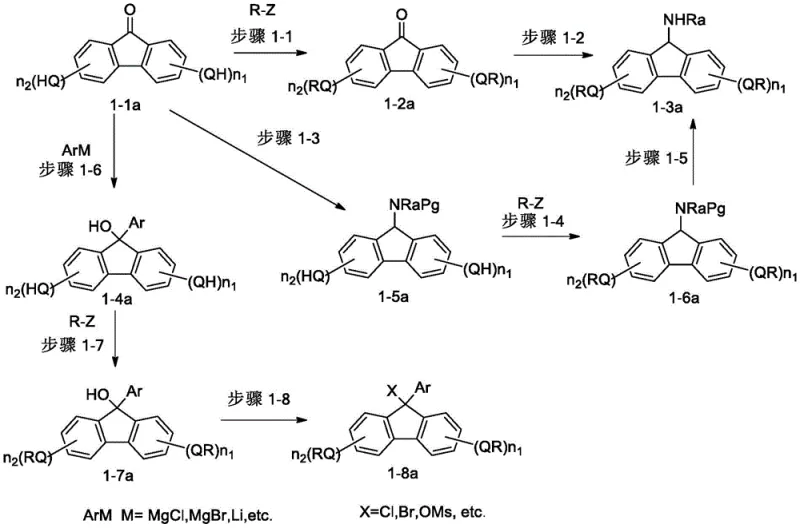
The synthesis of these advanced carriers follows a modular approach that is highly amenable to optimization and scale-up. The general strategy involves the functionalization of a fluorenone precursor, followed by the attachment of the lipophilic tail and subsequent conversion to the active amine form. For instance, the preparation of specific embodiments like FL-001 involves the alkylation of methyl 3,4,5-trihydroxybenzoate with isooctyl bromide to create the lipophilic acid component. This acid is then coupled to an amino-alcohol linker, which is subsequently attached to a hydroxy-fluorenone core. The final step typically involves the reduction of a ketone or oxime intermediate to generate the crucial 9-amino group required for peptide attachment. This pathway utilizes robust, well-understood chemical transformations such as nucleophilic substitution, amide coupling, and reduction, all of which can be performed using standard industrial equipment. The versatility of the route allows for the easy variation of the alkyl chain length and the substitution pattern on the fluorene ring to fine-tune solubility for specific peptide targets.
- Preparation of the fluorenone precursor via alkylation or Grignard reaction to introduce specific hydroxyl or amino groups at the 9-position.
- Coupling the fluorene derivative with long-chain alkoxy-benzoic acids or activated esters to establish the lipophilic carrier tail.
- Final deprotection or functionalization steps to yield the active amino-fluorene carrier suitable for heterogeneous peptide assembly.
Commercial Advantages for Procurement and Supply Chain Teams
For organizations focused on cost reduction in pharmaceutical intermediate manufacturing, the adoption of fluorene-based liquid carriers offers compelling economic benefits derived from process intensification. The primary driver of cost savings is the drastic simplification of the purification workflow. By replacing complex chromatographic separations or difficult filtrations with simple liquid-liquid extractions, manufacturers can significantly reduce solvent consumption, labor hours, and equipment downtime. The ability to operate at higher concentrations due to improved solubility also means that reactor volumes can be utilized more efficiently, effectively increasing throughput without the need for capital expansion. Furthermore, the high reagent utilization rate minimizes the amount of expensive protected amino acids and coupling agents required, directly lowering the bill of materials for each batch. These factors combine to create a leaner, more cost-effective production model that enhances competitiveness in the global peptide market.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reduction in solvent exchange steps lead to substantial cost savings. The process avoids the need for quantitative distillation to change solvent systems, which is energy-intensive and time-consuming. Instead, the system relies on simple phase separation, which reduces utility costs and accelerates the overall cycle time. Additionally, the high purity of intermediates reduces the load on final purification steps, further driving down the cost per gram of the active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The raw materials required for synthesizing these fluorene carriers, such as fluorenone derivatives and long-chain alkyl halides, are commercially available and stable. This ensures a secure supply chain that is not dependent on exotic or hard-to-source reagents. The robustness of the synthesis method also means that production schedules are more predictable, with fewer batches rejected due to gelation or solubility failures. This reliability is critical for maintaining continuous supply to downstream drug formulation facilities.
- Scalability and Environmental Compliance: The universal nature of the production method facilitates seamless scale-up from laboratory grams to commercial tons. The use of greener solvent systems, such as heptane and isopropyl acetate, aligns with modern environmental regulations and sustainability goals. The reduction in chemical waste generated from excess reagents and simplified workups contributes to a lower environmental footprint, making the process attractive for companies aiming to improve their green chemistry metrics while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of fluorene ring structure compounds in peptide synthesis workflows. These insights are derived directly from the experimental data and beneficial effects detailed in the patent documentation, providing a clear understanding of the technology's practical application. Understanding these nuances is essential for R&D teams evaluating the feasibility of transitioning from solid-phase to this advanced liquid-phase platform.
Q: What are the primary advantages of fluorene-based liquid carriers over solid-phase synthesis?
A: Fluorene-based liquid carriers overcome mass transfer limitations inherent in solid-phase synthesis. They allow for homogeneous reaction conditions which significantly increase reaction speed and reagent utilization, while still enabling simple liquid-liquid extraction for purification, combining the best aspects of both solution and solid-phase methods.
Q: How does this technology address the solubility issues found in previous liquid-phase carriers?
A: Unlike earlier carriers that suffered from rapid solubility drops or gelation as the peptide chain grew, these fluorene derivatives maintain consistent solubility profiles. Their unique amphiphilic structure allows them to remain soluble in non-polar solvents during reaction and facilitates separation in heterogeneous systems without precipitating amorphous solids that are difficult to filter.
Q: Can these carriers be scaled for industrial production of complex peptides?
A: Yes, the universal production method described supports commercial scale-up. The process utilizes standard solvent systems like heptane and isopropyl acetate, avoiding complex distillation steps. This universality and the ability to achieve high purity (>85-95%) through simple extraction make it highly suitable for large-scale manufacturing of API intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorene Ring Compound Supplier
As the demand for complex peptide therapeutics continues to surge, the need for efficient, scalable synthesis technologies has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging the principles of patent CN110256277B to deliver high-performance peptide synthesis carriers. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition to liquid-phase synthesis is smooth and successful. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of our fluorene-based intermediates meets the exacting standards required for GMP manufacturing. Our commitment to quality ensures that you receive a reliable fluorene ring compound supplier partner capable of supporting your most challenging synthesis projects.
We invite you to explore how our advanced carrier technology can optimize your peptide manufacturing processes. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific product portfolio. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you unlock the full potential of liquid-phase peptide synthesis, driving down costs and accelerating your time to market with superior quality intermediates.
