Advanced Synthesis of Somaglutide Side Chain Intermediates for Scalable Peptide Production
The pharmaceutical landscape for Type 2 diabetes and obesity treatment has been revolutionized by GLP-1 receptor agonists, with Somaglutide standing out as a market leader due to its dual efficacy in glycemic control and weight management. The commercial viability of such complex polypeptides relies heavily on the efficient synthesis of their specialized side chains, particularly the modification at the Lys26 position. Patent CN111454180A discloses a groundbreaking preparation method for a Somalutide side chain intermediate, specifically the PG-Lys(AEEA-AEEA-K)-OH structure, which serves as a critical building block in the total synthesis of the drug. This innovation addresses the longstanding challenges of steric hindrance and low coupling yields associated with traditional methods, offering a robust pathway for high-purity intermediate production. By introducing a modular approach where the side chain fragment is pre-assembled with orthogonal protecting groups, manufacturers can achieve superior control over the final peptide architecture.
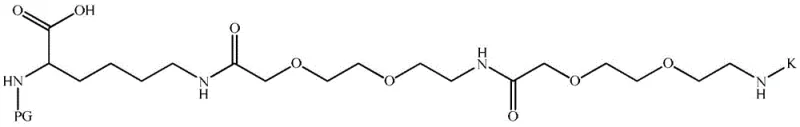
The strategic design of this intermediate allows for the sequential introduction of the fatty acid diacid moiety only after the core peptide backbone is established, thereby optimizing the overall synthetic efficiency. For research and development teams, understanding the precise structural nuances of this intermediate is paramount, as it dictates the success of the subsequent solid-phase peptide synthesis (SPPS) steps. The versatility of the protecting groups (PG and K) described in the patent ensures compatibility with various SPPS protocols, making this intermediate a universal key for unlocking high-quality Somaglutide production. As a reliable somaglutide intermediate supplier, leveraging such patented methodologies is essential for maintaining a competitive edge in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Somaglutide involved direct coupling strategies that presented significant bottlenecks for industrial scale-up. Previous patents, such as CN109456402A, described methods where the entire long-chain segment, Octadecanedioic Acid-Glu-AEEA-AEEA, was coupled directly to the Lys26 amine group on the growing peptide chain. This approach suffered from severe steric hindrance due to the bulkiness of the long hydrophobic fatty acid chain combined with the peptide spacer, leading to notoriously low coupling yields. Furthermore, the raw materials required for these pre-assembled long fragments were often prohibitively expensive and difficult to purify to the stringent standards required for pharmaceutical applications. The accumulation of deletion sequences and incomplete reactions at this critical junction often resulted in complex impurity profiles that were challenging to resolve during final purification, driving up the cost of goods sold and extending lead times.
The Novel Approach
The methodology outlined in CN111454180A represents a paradigm shift by decoupling the synthesis of the side chain spacer from the fatty acid attachment. Instead of attempting to couple the entire massive fragment at once, this novel approach first introduces a protected lysine derivative, PG-Lys(AEEA-AEEA-K)-OH, onto the polypeptide main chain. The 'K' group acts as a temporary, orthogonal mask for the gamma-amine of the glutamic acid linker, while the 'PG' group protects the alpha-amine of the lysine. This modular strategy allows the difficult coupling of the hydrophobic octadecanedioic acid fragment to occur as a separate, optimized step after the main chain assembly is complete. By breaking down the synthesis into manageable, high-yielding liquid-phase steps for the intermediate, the process drastically simplifies the solid-phase synthesis workflow, ensuring higher overall purity and reducing the risk of batch failure.
Mechanistic Insights into Orthogonal Protection and Fragment Assembly
The core chemical innovation lies in the sophisticated use of orthogonal protecting groups that allow for selective deprotection without disturbing other sensitive functionalities within the molecule. The 'K' group, which can be either Dde (2-(1-hydroxyethylidene)-5,5-dimethyl-1,3-cyclohexanedione) or ivDde (2-(1-hydroxyisopentylidene)-5,5-dimethyl-1,3-cyclohexanedione), is stable under the acidic conditions used to remove the Boc or Fmoc groups from the alpha-amine but can be selectively cleaved using hydrazine or similar nucleophiles later in the synthesis. This orthogonality is crucial because it prevents the premature reaction of the gamma-amine with the fatty acid before the lysine residue is correctly incorporated into the peptide backbone. The 'PG' group, which can range from Fmoc to Msc, Nsc, or Trt derivatives, provides the necessary stability during the initial coupling phases while being removable under specific conditions compatible with the final deprotection strategy.
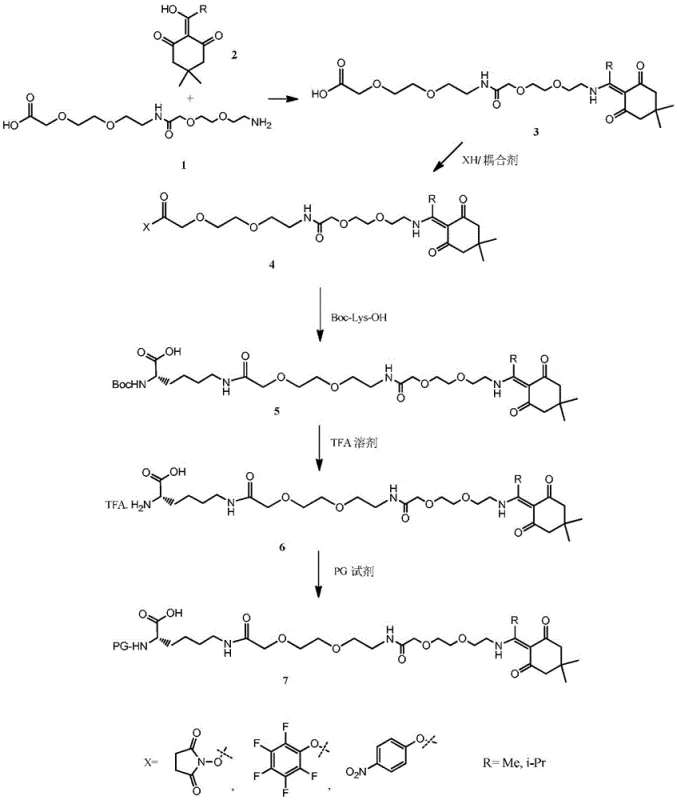
The synthesis begins with the condensation of AEEA-AEEA (2-(2-(2-aminoethoxy)ethoxy)acetic acid dimer) with the K-source under acidic catalysis, forming the K-AEEA-AEEA fragment with high efficiency. This fragment is then activated using standard coupling reagents like DCC or EDC in the presence of HOSu to form a reactive ester, which is subsequently coupled with Boc-Lys-OH in a biphasic organic-water system. This liquid-phase assembly ensures that the critical AEEA-AEEA-Lys junction is formed with high fidelity and purity before the molecule ever touches the solid-phase resin. The subsequent removal of the Boc group with trifluoroacetic acid (TFA) reveals the alpha-amine, which is then capped with the chosen PG group. This meticulous stepwise construction minimizes racemization and side reactions, resulting in an intermediate with a mass purity exceeding 96%, which is a critical specification for ensuring the quality of the final API.
How to Synthesize PG-Lys(AEEA-AEEA-K)-OH Efficiently
The preparation of this high-value intermediate follows a logical sequence of liquid-phase reactions designed to maximize yield and minimize purification complexity. The process leverages common organic solvents and reagents, making it accessible for standard chemical manufacturing facilities without requiring exotic equipment. The initial condensation and activation steps are performed at room temperature, reducing energy consumption, while the final protection steps utilize precise pH control to isolate the product as a sugar-like substance or through extraction, avoiding the need for preparative HPLC at the intermediate stage. For detailed operational parameters, stoichiometry, and specific workup procedures, please refer to the standardized guide below.
- Condense AEEA-AEEA with a K source (Dde or ivDde) using an acid catalyst to form the K-AEEA-AEEA fragment.
- Activate the K-AEEA-AEEA fragment using a coupling agent (e.g., DCC, EDC) and XH (e.g., HOSu) to form the active ester K-AEEA-AEEA-X.
- Couple the active ester with Boc-Lys-OH in an organic-water mixed solvent to obtain Boc-Lys(AEEA-AEEA-K)-OH.
- Remove the Boc protecting group using TFA to yield the amine salt H-Lys(AEEA-AEEA-K)-OH.TFA.
- Protect the alpha-amine with a PG reagent (e.g., Fmoc-Osu, Msc-Osu, Trt-Cl) to finalize the PG-Lys(AEEA-AEEA-K)-OH intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible benefits that extend beyond mere technical feasibility. The shift from direct long-chain coupling to a modular fragment condensation strategy fundamentally alters the cost structure of Somaglutide manufacturing. By synthesizing the side chain spacer independently, manufacturers can procure simpler, lower-cost starting materials like AEEA and cyclic diones rather than expensive pre-assembled long peptides. This decentralization of the synthesis allows for better inventory management and reduces the financial risk associated with holding high-value partially synthesized peptides. Furthermore, the high purity of the intermediate reduces the burden on downstream purification processes, leading to significant cost savings in chromatography resins and solvents.
- Cost Reduction in Manufacturing: The elimination of expensive, pre-assembled long-chain fragments significantly lowers the raw material costs. By utilizing a convergent synthesis strategy where the complex side chain is built from smaller, commodity-grade building blocks, the overall cost of goods is drastically reduced. The high yield of the liquid-phase steps means less material is wasted, and the avoidance of difficult-to-purify byproducts minimizes the loss of valuable intermediates during isolation. Additionally, the use of standard coupling reagents and solvents ensures that the process remains economically viable even at fluctuating market prices for specialty chemicals.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as lysine derivatives and ethoxylated acetic acids ensures a robust and resilient supply chain. Unlike proprietary long-chain fragments that may have limited suppliers, the precursors for this method are produced by multiple chemical manufacturers globally, mitigating the risk of supply disruptions. The simplified synthesis route also shortens the overall production cycle time, allowing for faster response to market demand fluctuations. This agility is crucial for meeting the surging global demand for GLP-1 agonists without compromising on delivery schedules.
- Scalability and Environmental Compliance: The liquid-phase nature of the intermediate synthesis facilitates easy scale-up from laboratory to commercial production volumes. The process avoids the use of hazardous heavy metal catalysts or extreme reaction conditions, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing facility. The ability to isolate intermediates as solids or through simple extractions reduces the volume of organic waste generated compared to continuous solid-phase synthesis of long fragments. This scalability ensures that the supply of high-purity somaglutide intermediates can be expanded seamlessly to support multi-ton annual production requirements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners. Understanding these details is vital for assessing the feasibility of integrating this intermediate into your existing peptide manufacturing workflows.
Q: What is the advantage of using Dde or ivDde protecting groups in this synthesis?
A: Dde and ivDde groups offer orthogonal stability; they remain intact during acidic Boc deprotection but can be selectively removed later under mild hydrazine conditions, preventing premature side reactions during peptide assembly.
Q: How does this method improve the purity of the final Somaglutide product?
A: By synthesizing the side chain fragment separately in the liquid phase with high purity (>96%) before attaching it to the resin, the method minimizes deletion sequences and impurities often associated with direct on-resin coupling of long hydrophobic chains.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process utilizes standard liquid-phase reactions with readily available reagents and avoids complex chromatographic purifications for intermediates, making it highly adaptable for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Somaglutide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex peptides like Somaglutide hinges on the quality and reliability of the key intermediates. Our technical team has extensively analyzed the route disclosed in CN111454180A and possesses the expertise to execute this synthesis with precision. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials that meet stringent purity specifications. Our rigorous QC labs are equipped to verify the orthogonal protection integrity and impurity profiles of every batch, guaranteeing that the PG-Lys(AEEA-AEEA-K)-OH intermediate performs flawlessly in your downstream solid-phase synthesis.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. By leveraging our process optimization capabilities, we can provide a Customized Cost-Saving Analysis tailored to your specific production volumes. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us be your partner in navigating the complexities of peptide intermediate manufacturing and securing your position in the rapidly growing GLP-1 market.
