Scalable Synthesis of High-Purity Anserine Intermediates for Industrial Applications
Introduction to Advanced Anserine Manufacturing
The escalating demand for Anserine, a potent natural histidine dipeptide with significant physiological buffering and antioxidant capabilities, has necessitated a paradigm shift from inefficient extraction methods to robust synthetic pathways. As detailed in patent CN112624977A, the development of a novel intermediate, specifically the compound of formula (II), represents a critical breakthrough in the industrial preparation of this high-value molecule. Unlike traditional extraction from vertebrate muscle tissues, which yields low purity and suffers from supply chain volatility, this synthetic approach offers a stable, controllable, and economically viable route suitable for large-scale production. The technology addresses the urgent need for a reliable pharmaceutical intermediate supplier capable of delivering consistent quality for both food and medical applications, ensuring that the final product meets stringent regulatory standards for purity and safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
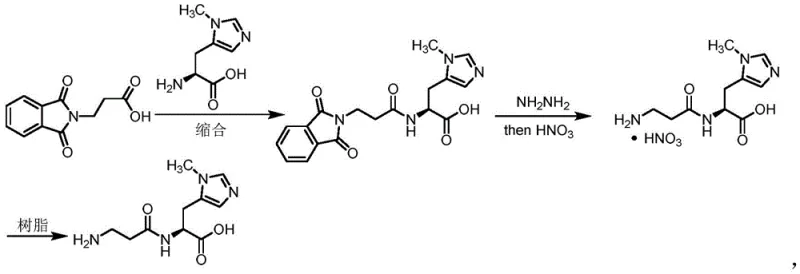
Historically, the synthesis of Anserine has been plagued by significant operational hazards and inefficiencies, as exemplified by the pioneering yet outdated Rinderknecht route reported in 1964. This legacy method relies on the condensation of phthaloyl-protected alanine with N(τ)-methyl-L-histidine benzyl ester, followed by a hazardous hydrazinolysis step to remove the phthaloyl group. The use of hydrazine introduces severe safety risks due to its toxicity and explosive nature, creating substantial barriers to commercial adoption. Furthermore, the deprotection process yields anserine that cannot be directly isolated; it requires a cumbersome sequence of nitrate salt formation followed by ion-exchange resin treatment to obtain the free base. This multi-step purification not only depresses the overall yield to approximately 65 percent but also introduces inorganic contaminants that are difficult to remove, making the process unsuitable for modern GMP manufacturing environments where trace metal and salt control is paramount.
The Novel Approach
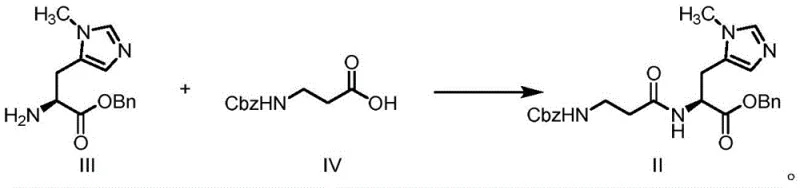
In stark contrast, the methodology disclosed in CN112624977A introduces a streamlined condensation strategy that bypasses these historical bottlenecks by utilizing N(τ)-methyl-L-histidine benzyl ester (Compound III) and Cbz-protected beta-alanine (Compound IV) as key building blocks. This innovative route employs standard peptide coupling techniques or mixed anhydride activation to form the critical amide bond, resulting in the stable intermediate Compound II with significantly improved yields ranging from 78 percent to over 80 percent in optimized examples. The process eliminates the need for toxic hydrazine and complex ion-exchange chromatography, replacing them with straightforward aqueous workups and recrystallization steps that are easily scalable. By shifting to a Cbz/benzyl protection strategy, the synthesis aligns with modern process chemistry principles, facilitating easier handling and purification while maintaining the stereochemical integrity of the histidine residue, which is essential for biological activity.
Mechanistic Insights into Peptide Condensation and Hydrogenolysis
The core of this technological advancement lies in the efficient formation of the peptide bond between the amino group of the histidine derivative and the carboxyl group of the beta-alanine derivative. The patent elucidates several activation mechanisms, including the use of carbodiimides like EDCI in conjunction with additives such as HOBt to suppress racemization and enhance coupling efficiency. Alternatively, the formation of a mixed anhydride using isobutyl chloroformate or pivaloyl chloride provides a highly reactive species that facilitates rapid amidation under mild thermal conditions, typically between -10°C and 30°C. This flexibility in activation chemistry allows manufacturers to tailor the process based on available infrastructure and cost considerations, ensuring that the reaction proceeds with high selectivity to minimize the formation of diastereomeric impurities that could compromise the efficacy of the final dipeptide product.
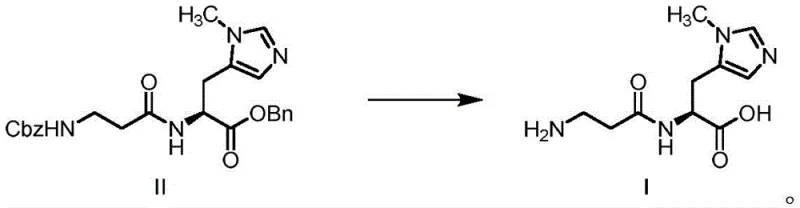
Following the successful synthesis of the fully protected intermediate, the final transformation involves a sophisticated catalytic hydrogenation step that simultaneously removes both the N-terminal Cbz group and the C-terminal benzyl ester. This dual deprotection is achieved using heterogeneous catalysts such as palladium on carbon (Pd/C), palladium black, or Raney nickel under hydrogen pressures ranging from 1 to 3 atmospheres. The mechanistic elegance of this step is its ability to cleave two distinct protecting groups in a single operation without affecting the imidazole ring or the peptide backbone. Experimental data indicates that this hydrogenolysis proceeds with exceptional efficiency, achieving yields upwards of 90 percent with chemical purities exceeding 99.3 percent. The use of solvent systems like methanol/water mixtures further enhances the solubility of the intermediate while providing a medium conducive to efficient gas-liquid-solid mass transfer during the catalytic cycle.
How to Synthesize Anserine Intermediate Efficiently
The practical implementation of this synthesis route requires precise control over reaction parameters to maximize yield and purity while minimizing waste generation. The process begins with the careful selection of solvents such as dichloromethane, acetonitrile, or tetrahydrofuran, which provide optimal solubility for both the histidine and beta-alanine derivatives. Operators must maintain strict temperature control during the activation phase to prevent side reactions, followed by a thorough aqueous workup to remove urea byproducts from carbodiimide coupling or acid salts from mixed anhydride methods. The detailed standardized synthesis steps, including specific molar ratios, agitation speeds, and filtration protocols required to replicate the high yields reported in the patent examples, are outlined in the technical guide below for qualified process engineers.
- Condense N(τ)-methyl-L-histidine benzyl ester (III) with Cbz-beta-alanine (IV) using coupling agents like EDCI/HOBt or mixed anhydride methods to form Compound II.
- Purify the intermediate Compound II via aqueous workup and recrystallization from ethyl acetate/n-heptane systems to ensure high chemical purity.
- Perform catalytic hydrogenation on Compound II using Pd/C or Raney Nickel under mild pressure to simultaneously remove Cbz and benzyl protecting groups, yielding Anserine (I).
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this novel synthesis route offers profound advantages by fundamentally restructuring the cost drivers associated with Anserine production. The elimination of hazardous reagents like hydrazine not only reduces the regulatory burden and safety compliance costs but also simplifies the waste disposal workflow, leading to significant operational expenditure savings. Furthermore, the reliance on commercially available starting materials such as Cbz-beta-alanine and protected histidine esters ensures a stable supply chain that is less susceptible to the fluctuations seen in natural extraction markets. The robustness of the crystallization-based purification method means that production cycles are shorter and more predictable, allowing for better inventory management and reduced lead times for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The transition from ion-exchange purification to simple crystallization drastically reduces the consumption of consumables and resins, which are often expensive and have limited lifecycles. By avoiding the use of transition metals that require complex scavenging steps, the process inherently lowers the cost of goods sold through simplified downstream processing. The high yield of the condensation step minimizes raw material waste, ensuring that the input costs are efficiently converted into saleable product mass, thereby enhancing the overall margin profile for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: Utilizing synthetic precursors decouples production from the seasonal and geographical constraints of animal tissue sourcing, providing a consistent and reliable supply of Anserine intermediates throughout the year. The modular nature of the synthesis allows for production to be scaled up or down based on market demand without the long lead times associated with biological extraction facilities. This flexibility is crucial for meeting the just-in-time delivery requirements of global nutraceutical and pharmaceutical clients who demand uninterrupted supply continuity.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, utilizing solvents that can be readily recovered and recycled, thus minimizing the environmental footprint of the manufacturing operation. The absence of heavy metal catalysts in the final product simplifies the release testing process, accelerating the time to market for finished goods. Additionally, the mild reaction conditions reduce energy consumption compared to high-temperature extraction processes, aligning with corporate sustainability goals and reducing the overall carbon intensity of the production lifecycle.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Anserine synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these nuances is essential for R&D teams planning pilot runs and procurement officers assessing vendor capabilities.
Q: What are the critical impurities in traditional Anserine synthesis?
A: Traditional routes often suffer from incomplete deprotection and salt formation issues, requiring complex ion-exchange steps that introduce inorganic residues and lower overall yield.
Q: How does the new condensation method improve scalability?
A: By utilizing standard peptide coupling reagents or mixed anhydrides in common solvents like DCM or THF, the process eliminates the need for hazardous hydrazine and simplifies downstream purification to basic washing and crystallization.
Q: Is the catalytic hydrogenation step selective enough for industrial use?
A: Yes, the patent demonstrates that using Pd/C or Raney Nickel allows for the simultaneous removal of both Cbz and benzyl ester groups with high selectivity, achieving chemical purities exceeding 99% without racemization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Anserine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we leverage our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this advanced Anserine synthesis technology to the global market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of intermediate meets the exacting standards required for food and pharmaceutical grade applications. We understand the critical importance of impurity profiling and chiral purity in dipeptide synthesis, and our technical team is dedicated to optimizing every step of the condensation and hydrogenation process to deliver superior product quality consistently.
We invite potential partners to engage with our technical procurement team to discuss how this innovative route can drive value in your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with a partner committed to technical excellence and commercial reliability.
