Advanced Copper-Catalyzed Synthesis of 8-Thiaxanthine Intermediates for Commercial Scale-Up
Advanced Copper-Catalyzed Synthesis of 8-Thiaxanthine Intermediates for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds, particularly xanthine derivatives which serve as critical intermediates for adenosine receptor antagonists. Patent CN103254194A discloses a groundbreaking preparation method for 8-thiaxanthine compounds, utilizing a cost-effective copper salt catalyst to achieve direct C-H bond functionalization. This technology represents a significant paradigm shift from traditional multi-step halogenation processes, offering a streamlined one-step synthesis that dramatically improves atom economy and operational safety. By leveraging stable disulfide reagents instead of volatile thiols, this method addresses long-standing challenges in the manufacturing of bioactive molecules like KW-3902 and KW-6002. For R&D directors and procurement specialists, understanding this technological leap is essential for optimizing supply chains and reducing the cost of goods sold for complex pharmaceutical intermediates.
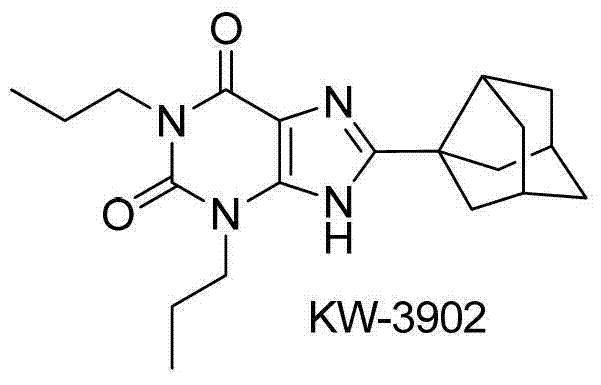
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 8-substituted xanthine compounds has relied heavily on a two-step sequence involving initial halogenation followed by nucleophilic substitution. This conventional pathway typically necessitates the use of excessive amounts of highly corrosive halogens, such as liquid bromine, which presents severe handling risks and requires specialized corrosion-resistant equipment for large-scale production. Furthermore, the subsequent substitution step employs thiol compounds, which are notoriously volatile, possess repulsive odors, and are prone to rapid oxidation into disulfides, leading to significant impurity profiles. To compensate for this oxidation and ensure complete conversion, manufacturers are often forced to use a large excess of thiol reagents, resulting in poor atom economy and generating substantial hazardous waste that complicates downstream purification and environmental compliance.
The Novel Approach
In stark contrast, the innovative method described in the patent utilizes a copper-catalyzed C-H activation strategy that bypasses the need for pre-functionalization entirely. By reacting a xanthine compound of Formula II directly with a disulfide compound of Formula III in the presence of a copper salt catalyst, the 8-thiaxanthine structure (Formula I) is constructed in a single operational step. This approach eliminates the hazardous halogenation stage and replaces unstable thiols with air-stable, easy-to-handle disulfides. The reaction proceeds efficiently in organic solvents like DMF at moderate temperatures, significantly simplifying the workflow. This transition from a linear, hazard-prone sequence to a convergent, catalytic process not only enhances safety but also drastically reduces the number of unit operations required, thereby lowering capital expenditure and operational complexity for commercial manufacturing facilities.

Mechanistic Insights into Copper-Catalyzed C-H Activation
The core of this technological advancement lies in the efficient activation of the inert C-H bond at the 8-position of the xanthine ring. The mechanism involves the coordination of the copper catalyst, specifically copper acetate, which facilitates the cleavage of the C-H bond and the subsequent formation of a carbon-sulfur bond with the disulfide reagent. The addition of silver acetate acts as a crucial promoter, synergistically enhancing the catalytic cycle to accelerate reaction kinetics and improve overall yields. This dual-metal system ensures that the reaction reaches completion within a practical timeframe of 8 to 16 hours at temperatures between 110°C and 130°C. The robustness of this catalytic system allows for high turnover numbers, minimizing the residual metal content in the final product, which is a critical quality attribute for pharmaceutical intermediates intended for further biological evaluation or clinical use.
Furthermore, the resulting 8-thiaxanthine intermediates serve as versatile precursors for downstream diversification via Liebeskind-Srogl cross-coupling reactions. The sulfur moiety installed at the 8-position acts as an effective leaving group when treated with palladium catalysts and boronic acids, enabling the introduction of diverse aryl or alkenyl groups. This two-stage strategy—first installing the sulfur handle via copper catalysis, then exchanging it for a carbon framework via palladium catalysis—provides a modular platform for synthesizing complex 8-aryl or 8-alkenyl xanthine derivatives. This modularity is invaluable for medicinal chemistry campaigns where rapid exploration of structure-activity relationships (SAR) around the xanthine core is required to optimize potency and selectivity against targets like MAO-B or adenosine receptors.
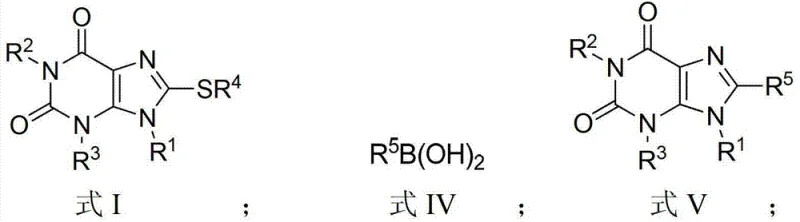
How to Synthesize 8-Thiaxanthine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the use of commercially available reagents and standard processing equipment. The procedure begins with the preparation of the catalytic mixture, followed by the addition of substrates and thermal activation. Detailed standardized operating procedures regarding stoichiometry, solvent selection, and workup techniques are critical for reproducing the high yields reported in the examples. For process chemists looking to implement this route, adherence to the specified molar ratios of copper acetate, silver acetate, and disulfide is essential to maximize efficiency. The following guide summarizes the critical operational parameters derived from the patent data to ensure successful execution of this transformative synthetic methodology.
- Prepare the catalytic system by dissolving copper acetate and silver acetate in DMF solvent within a reaction vessel.
- Introduce the xanthine substrate (Formula II) and the disulfide compound (Formula III) to the mixture under stirring.
- Heat the reaction mixture to 110°C-130°C for 8-16 hours to facilitate C-H activation and thio-functionalization.
- Quench the reaction with water, extract with dichloromethane, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this copper-catalyzed methodology offers profound economic and logistical benefits over legacy synthetic routes. The elimination of hazardous halogenation reagents and volatile thiols significantly reduces the costs associated with specialized containment, waste disposal, and regulatory compliance. By simplifying the synthesis from a multi-step sequence to a one-pot transformation, manufacturers can achieve substantial reductions in production lead times and inventory holding costs. The use of stable disulfide starting materials ensures consistent raw material quality and shelf-life, mitigating the risk of supply disruptions caused by the degradation of sensitive thiol reagents. These factors collectively contribute to a more resilient and cost-effective supply chain for high-value pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The streamlined one-step process inherently lowers manufacturing costs by reducing solvent consumption, energy usage, and labor hours associated with multiple isolation and purification stages. The avoidance of expensive and hazardous halogenating agents further decreases raw material expenses. Additionally, the high atom economy of the C-H activation strategy minimizes waste generation, leading to lower environmental treatment fees. While specific percentage savings depend on local utility and labor rates, the qualitative reduction in process complexity guarantees a lower cost base per kilogram of produced intermediate compared to traditional five-step sequences.
- Enhanced Supply Chain Reliability: Utilizing air-stable disulfides instead of odoriferous and oxidation-prone thiols enhances the reliability of raw material sourcing and storage. This stability reduces the likelihood of batch failures due to reagent degradation, ensuring consistent production output. The robustness of the copper catalytic system also allows for flexibility in scaling, as the reaction conditions are not overly sensitive to minor variations in temperature or mixing, which is crucial for maintaining supply continuity during technology transfer from laboratory to commercial plant.
- Scalability and Environmental Compliance: The reaction conditions, operating at moderate temperatures of 110°C to 130°C in common solvents like DMF, are highly amenable to scale-up in standard stainless steel reactors. This contrasts sharply with older methods requiring extreme conditions or corrosive environments that demand exotic metallurgy. The significant reduction in hazardous waste streams aligns with modern green chemistry principles and stringent environmental regulations, facilitating easier permitting and long-term operational sustainability for manufacturing sites focused on pharmaceutical intermediate production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield improvements, safety profiles, and structural scope. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing production portfolios for xanthine-based therapeutics.
Q: Why is the copper-catalyzed C-H activation method superior to traditional halogenation?
A: Traditional methods require corrosive halogens like liquid bromine and volatile, odorous thiols, posing significant safety and environmental hazards. The novel copper-catalyzed approach utilizes stable disulfides and avoids hazardous halogenation steps, resulting in a safer, more atom-economical, and environmentally friendly process suitable for large-scale manufacturing.
Q: What is the yield advantage for synthesizing clinical candidates like KW-6002?
A: The patented method achieves a yield of 83% for KW-6002 in significantly fewer steps compared to literature routes which often require five steps with a total yield of only 33%. This drastic reduction in synthetic steps directly translates to lower material costs and simplified purification protocols.
Q: Can this method accommodate diverse functional groups for drug discovery?
A: Yes, the methodology demonstrates excellent functional group tolerance. It successfully accommodates various substituents at the N1, N3, and N7 positions (such as methyl, ethyl, benzyl, allyl) and diverse aryl or alkyl groups at the 8-position via disulfide variation, making it highly versatile for generating libraries of bioactive xanthine derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Thiaxanthine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes for complex heterocyclic intermediates like 8-thiaxanthine derivatives. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facility is equipped with rigorous QC labs capable of meeting stringent purity specifications required for clinical-grade materials, guaranteeing that every batch of 8-thiaxanthine intermediate delivered meets the highest standards of quality and consistency demanded by the global pharmaceutical industry.
We invite you to leverage our technical expertise to optimize your supply chain for xanthine-based drug candidates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced copper-catalyzed capabilities can drive down your costs and accelerate your time to market for next-generation therapeutics targeting adenosine receptors and beyond.
