Scalable Visible-Light Synthesis of Arylacetylene Alkyl Sulfones for Advanced Pharmaceutical Intermediates
Introduction to Patent CN110698369B: A Breakthrough in Sulfonylation Chemistry
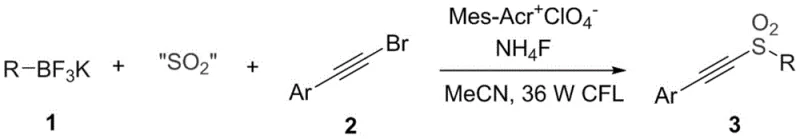
The chemical industry is constantly seeking greener and more efficient pathways to construct complex molecular architectures, particularly for the pharmaceutical sector where sulfonyl motifs are ubiquitous in bioactive molecules. Patent CN110698369B introduces a transformative methodology for synthesizing arylacetylene alkyl sulfone compounds, leveraging the power of visible light photoredox catalysis. This innovation represents a significant departure from classical synthetic routes, utilizing alkyl fluoroborates as stable radical precursors and solid sulfur dioxide surrogates to achieve high-yield transformations under exceptionally mild conditions. By replacing hazardous reagents with benign alternatives and employing ambient temperature protocols, this technology addresses critical pain points in modern organic synthesis, offering a robust platform for the production of high-value pharmaceutical intermediates with superior purity profiles and reduced environmental footprints.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of sulfonyl fragments has relied heavily on traditional sulfonation reagents that pose severe safety and environmental challenges. Conventional processes often necessitate the use of strongly corrosive acids, toxic gaseous sulfur dioxide, or aggressive oxidizing agents, which demand specialized containment infrastructure and rigorous safety protocols. These harsh reaction conditions not only increase the risk of industrial accidents but also limit the functional group tolerance, often leading to decomposition of sensitive substrates or the formation of complex impurity profiles that are difficult to separate. Furthermore, the handling of gaseous reagents requires high-pressure equipment, adding substantial capital expenditure and operational complexity to the manufacturing process, thereby inflating the overall cost of goods for downstream pharmaceutical applications.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a visible-light-driven radical cascade that operates at room temperature in standard glassware. By employing alkyl fluoroborates, which are air-stable and easy to handle, alongside solid SO2 surrogates like sodium metabisulfite or DABSO, the process eliminates the need for handling toxic gases or corrosive liquids. The use of an organic photocatalyst, specifically 9-mesityl-10-methylacridine perchlorate, enables the generation of reactive radical species through a clean single-electron transfer mechanism. This shift to photochemical activation not only enhances the safety profile of the synthesis but also dramatically improves atom economy and step efficiency, providing a streamlined route to diverse arylacetylene alkyl sulfones that is ideally suited for the demands of modern process chemistry.
Mechanistic Insights into Visible Light Photoredox Sulfonylation
The core of this technological advancement lies in its elegant photoredox catalytic cycle, which orchestrates the formation of carbon-sulfur bonds with remarkable precision. Upon irradiation with visible light, the photosensitizer Mes-Acr+ClO4- absorbs photon energy to reach an excited oxidative state capable of engaging in single electron transfer (SET) with the alkyl fluoroborate substrate. This interaction triggers the fragmentation of the boron-carbon bond, releasing a reactive alkyl radical while regenerating the ground state catalyst or entering a subsequent redox cycle. The generated alkyl radical then rapidly intercepts the sulfur dioxide surrogate, forming an alkyl sulfonyl radical intermediate that serves as the key nucleophilic species for the subsequent bond-forming event.
Following the formation of the alkyl sulfonyl radical, the mechanism proceeds through a radical addition to the electron-deficient triple bond of the arylacetylene bromide. This addition step creates a vinyl radical intermediate, which subsequently undergoes a halogen atom transfer or elimination process to finalize the formation of the arylacetylene alkyl sulfone product. This radical pathway ensures high regioselectivity and minimizes side reactions commonly associated with ionic mechanisms. The entire sequence is driven by the continuous absorption of light energy, allowing the reaction to proceed efficiently without the need for external heating or harsh chemical initiators, thus preserving the integrity of sensitive functional groups present on the aryl or alkyl moieties.
How to Synthesize Arylacetylene Alkyl Sulfones Efficiently
The practical implementation of this synthesis involves a straightforward one-pot procedure that combines the arylacetylene bromide, the sulfur dioxide solid substitute, the alkyl fluoroborate, ammonium fluoride, and the photosensitizer in a suitable organic solvent such as acetonitrile. The reaction mixture is subjected to an inert atmosphere to prevent oxygen quenching of the radical species and then irradiated with a standard fluorescent lamp while stirring at room temperature. This simplicity in operation makes the protocol highly accessible for laboratory optimization and subsequent process scaling, with the detailed standardized synthesis steps provided in the technical guide below ensuring reproducibility and consistency across different batches.
- Combine arylacetylene bromide, a sulfur dioxide solid substitute (like sodium metabisulfite), alkyl fluoroborate, ammonium fluoride, and the photosensitizer Mes-Acr+ClO4- in a reaction vessel under an inert atmosphere.
- Add an organic solvent such as acetonitrile and irradiate the mixture with visible light (e.g., 35W fluorescent lamp) at room temperature while stirring for approximately 48 hours.
- Upon completion, concentrate the reaction mixture under reduced pressure and purify the crude product via column chromatography using a petroleum ether and ethyl acetate system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photoredox methodology offers compelling strategic benefits that extend beyond mere technical feasibility. The transition from hazardous traditional reagents to stable, solid-state alternatives significantly de-risks the supply chain by reducing dependency on specialized logistics for toxic gas transport and storage. Moreover, the mild reaction conditions translate directly into lower energy consumption, as the process operates effectively at ambient temperatures without the need for cryogenic cooling or high-temperature heating, resulting in substantial operational cost savings over the lifecycle of the product manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous sulfonation reagents, coupled with the use of inexpensive visible light sources instead of thermal energy, drastically reduces the raw material and utility costs associated with production. By avoiding the need for complex corrosion-resistant reactors and specialized gas handling infrastructure, capital expenditure for new production lines is significantly minimized, while the simplified workup procedures reduce labor and waste treatment expenses, leading to a more economically competitive final product for the pharmaceutical market.
- Enhanced Supply Chain Reliability: The reliance on commercially available and shelf-stable starting materials, such as alkyl fluoroborates and solid SO2 surrogates, ensures a robust and uninterrupted supply of key inputs. Unlike gaseous reagents that may face logistical bottlenecks or regulatory restrictions, these solid reagents can be sourced from multiple global suppliers and stored safely for extended periods, thereby mitigating the risk of production delays and enhancing the overall resilience of the manufacturing schedule against external supply shocks.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this process, including the avoidance of toxic byproducts and the use of benign solvents, facilitate easier regulatory approval and environmental compliance. The scalability of photochemical reactions has improved immensely with modern flow chemistry technologies, allowing for seamless transition from gram-scale discovery to multi-kilogram commercial production without the safety risks associated with scaling up exothermic traditional sulfonation reactions, ensuring a sustainable and compliant manufacturing footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light mediated sulfonylation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, aiming to clarify the operational parameters and potential advantages for industrial partners considering this innovative synthetic route for their intermediate pipelines.
Q: What are the key advantages of this photoredox method over traditional sulfonation?
A: Unlike traditional methods that require highly toxic and corrosive sulfonation reagents under harsh acidic or oxidative conditions, this patented process utilizes mild visible light irradiation at room temperature. It employs stable alkyl fluoroborates and solid SO2 surrogates, significantly improving operational safety and reducing environmental hazards associated with waste disposal.
Q: Can this synthesis method be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the method is designed for scalability. The use of inexpensive and commercially available starting materials, combined with simple reaction conditions (room temperature, visible light), eliminates the need for specialized high-pressure or cryogenic equipment. This facilitates easier transition from laboratory benchtop to kilogram or ton-scale manufacturing for API intermediates.
Q: What is the role of the photosensitizer in this reaction mechanism?
A: The photosensitizer, specifically 9-mesityl-10-methylacridine perchlorate (Mes-Acr+ClO4-), absorbs visible light photons to reach an excited state. In this state, it acts as a strong oxidant to initiate single electron transfer (SET) with the alkyl fluoroborate, generating the crucial alkyl radical species that subsequently reacts with sulfur dioxide to form the sulfonyl radical.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arylacetylene Alkyl Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of photoredox catalysis in accelerating the development of next-generation pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photochemical reactors and stringent purity specifications, supported by rigorous QC labs that guarantee every batch of arylacetylene alkyl sulfones meets the highest international standards for quality and consistency required by top-tier drug manufacturers.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this cutting-edge technology for your specific project needs. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your target molecule, optimizing the synthetic route for maximum efficiency. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, ensuring your supply chain is built on the foundation of the most advanced and reliable chemistry available.
