Advanced Fused-Ring Benzothiadiazole Non-Fullerene Acceptors for High-Efficiency Organic Solar Cells
The rapid evolution of the renewable energy sector has placed organic photovoltaics (OPV) at the forefront of next-generation power solutions, driven by the urgent need for flexible, lightweight, and cost-effective energy harvesting technologies. Patent CN109134513B introduces a groundbreaking class of fused-ring benzothiadiazole non-fullerene acceptor materials that address critical limitations in current organic solar cell architectures. Unlike traditional fullerene-based acceptors which suffer from weak absorption in the visible region and difficult energy level tuning, this novel material utilizes a nitrogen-bridged trapezoidal fused ring structure as its central core. This unique molecular design significantly enhances the electron cloud density through the lone pair participation of nitrogen atoms, thereby improving the electron-donating capability of the core unit. The material is engineered with electron-withdrawing end groups connected to both ends of the central core, creating a push-pull electronic system that effectively broadens the absorption spectrum into the near-infrared region. 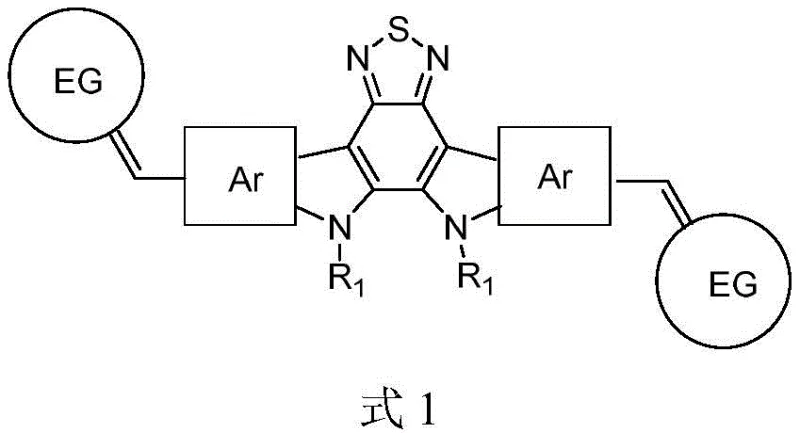
Furthermore, the strategic incorporation of alkyl or alkoxy chains on the nitrogen bridges not only increases the regional planarity of the molecule, potentially boosting charge carrier mobility to values greater than or equal to 10^-4 cm^2·V^-1·s^-1, but also drastically improves solubility in common organic solvents. This dual improvement in electronic properties and processability makes these materials highly suitable for large-area coating techniques required in commercial module manufacturing. The patent data indicates that devices fabricated with these acceptors, such as the specific embodiment ZYQ3, can achieve power conversion efficiencies (PCE) approaching 16%, representing a significant leap forward from the 14% ceiling often observed in earlier single-layer heterojunction systems. For R&D directors seeking reliable electronic chemical suppliers, this technology offers a robust platform for developing high-performance, flexible solar panels that can compete with traditional silicon-based cells in niche applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of organic solar cells has been heavily reliant on fullerene derivatives, such as PCBM, as the standard electron acceptor materials. While these materials possess high electron affinity and isotropic charge transport, they exhibit inherent drawbacks that limit the overall efficiency and commercial viability of OPV devices. One major limitation is their weak and narrow absorption in the visible light spectrum, which restricts the photocurrent generation capability of the device. Additionally, the energy levels of fullerenes are difficult to tune chemically without compromising their stability or solubility, leading to significant energy losses during charge transfer at the donor-acceptor interface. The rigid spherical structure of fullerenes also tends to result in excessive aggregation and poor morphological stability in the active layer blend, which can degrade device performance over time. Moreover, the synthesis of higher-order fullerene derivatives is often complex and expensive, posing a barrier to cost reduction in electronic chemical manufacturing. These factors collectively hinder the ability of conventional fullerene-based systems to reach the theoretical efficiency limits required for widespread commercial adoption.
The Novel Approach
The novel approach detailed in patent CN109134513B overcomes these hurdles by employing a non-fullerene acceptor architecture based on a fused-ring benzothiadiazole core. This design allows for precise modulation of the optical bandgap to values less than 1.5 eV, ensuring better spectral complementarity with common donor polymers like PCE10 and PCE12. By replacing the isotropic fullerene cage with a planar, conjugated ladder-type structure, the new material facilitates stronger intermolecular pi-pi stacking, which enhances charge transport pathways within the active layer. The synthetic strategy involves a modular assembly where the central core is constructed first, followed by the attachment of versatile electron-withdrawing end groups via Knoevenagel condensation. This modularity enables fine-tuning of the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) levels to minimize voltage losses. Consequently, this approach yields materials with superior film-forming properties and higher photoelectric conversion efficiency, providing a clear pathway for the commercial scale-up of complex polymer additives and acceptor materials for the renewable energy market.
Mechanistic Insights into Stille Coupling and Cyclization Reactions
The synthesis of the fused-ring benzothiadiazole core relies heavily on palladium-catalyzed cross-coupling reactions, specifically the Stille coupling, to construct the extended conjugated system. In the initial step, 4,7-dibromo-5,6-dinitrobenzothiadiazole reacts with stannylated thiophene derivatives in the presence of a Pd(PPh3)2Cl2 catalyst. This reaction proceeds through a catalytic cycle involving oxidative addition of the aryl bromide to the palladium center, transmetallation with the organotin species, and reductive elimination to form the new carbon-carbon bond. The use of tetrahydrofuran (THF) as a solvent at temperatures between 80°C and 100°C ensures sufficient kinetic energy for the reaction while maintaining the stability of the sensitive nitro groups. 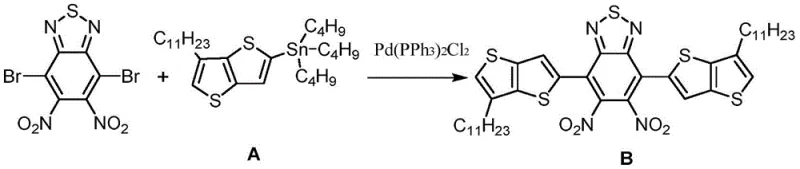
Following the coupling, the formation of the rigid trapezoidal fused ring is achieved through an intramolecular condensation ring-closure reaction mediated by triethyl phosphite. This step is critical as it locks the molecular conformation into a planar geometry, which is essential for high charge mobility. The reaction is typically conducted in o-dichlorobenzene at elevated temperatures of 160°C to 180°C under an inert argon atmosphere to prevent oxidation. The mechanism likely involves the reduction of the nitro groups to intermediate species that subsequently cyclize with the adjacent aromatic rings, eliminating oxygen atoms in the process. This cyclization creates the nitrogen-bridge that defines the core structure, enhancing the conjugation length and electronic communication across the molecule. The subsequent functionalization steps, including nucleophilic substitution to introduce solubilizing alkyl chains and Vilsmeier-Haack formylation to install aldehyde handles, are designed to be high-yielding and scalable, ensuring that the final material can be produced with high purity specifications required for electronic applications.
How to Synthesize Fused-Ring Benzothiadiazole Non-Fullerene Acceptor Efficiently
The preparation of these high-performance acceptor materials follows a well-defined five-step synthetic protocol that balances yield, purity, and operational simplicity. The process begins with the construction of the precursor backbone via Stille coupling, followed by the crucial cyclization step to form the fused core. Subsequent modifications include N-alkylation to improve solubility, formylation to activate the ends for condensation, and finally, the Knoevenagel reaction to attach the electron-withdrawing end groups. Each step has been optimized in the patent examples to minimize side reactions and facilitate purification via standard silica gel column chromatography. For detailed operational parameters and stoichiometric ratios, please refer to the standardized synthesis guide below.
- Perform Stille coupling between 4,7-dibromo-5,6-dinitrobenzothiadiazole and stannylated thiophene derivatives using Pd(PPh3)2Cl2 catalyst in THF at 80-100°C.
- Execute condensation ring-closure reaction using triethyl phosphite in o-dichlorobenzene at 160-180°C to form the nitrogen-bridged trapezoidal fused core.
- Complete the synthesis via Vilsmeier-Haack formylation followed by Knoevenagel condensation with electron-withdrawing end groups to finalize the acceptor structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this fused-ring benzothiadiazole technology offers substantial strategic advantages over legacy fullerene-based systems. The synthetic route utilizes commercially available starting materials such as 4,7-dibromo-5,6-dinitrobenzothiadiazole and common thiophene derivatives, which reduces dependency on exotic or single-source reagents. This availability enhances supply chain reliability and mitigates the risk of production delays caused by raw material shortages. Furthermore, the reaction conditions, while requiring specific catalysts like palladium, operate at moderate temperatures and pressures that are compatible with standard chemical manufacturing infrastructure, avoiding the need for specialized high-pressure reactors or cryogenic equipment.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive and difficult-to-process fullerene cages, which are notoriously costly to produce and purify. By utilizing solution-processable small molecules that can be synthesized through established organic reactions like Stille coupling and Knoevenagel condensation, the overall cost of goods sold (COGS) can be significantly reduced. The high solubility of the final product in common solvents like chloroform and o-dichlorobenzene also simplifies the ink formulation process for printing, reducing solvent consumption and waste disposal costs associated with more recalcitrant materials. Additionally, the high yields reported in the patent examples suggest efficient atom economy, further contributing to cost optimization in large-scale production scenarios.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for flexibility in sourcing; for instance, different alkyl chains or end groups can be swapped without altering the core synthetic strategy, providing resilience against supply disruptions of specific intermediates. The use of robust reaction conditions, such as refluxing in high-boiling solvents, ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with downstream solar module manufacturers. The ability to store intermediates like the formylated compound D without significant degradation also allows for better inventory management and just-in-time production scheduling, reducing the capital tied up in work-in-progress materials.
- Scalability and Environmental Compliance: The process avoids the use of highly toxic reagents often associated with fullerene functionalization, aligning better with increasingly stringent environmental regulations in the chemical industry. While palladium catalysts are used, established protocols for metal scavenging and recycling can be implemented to recover valuable metals and minimize heavy metal contamination in the final product. The scalability of the Stille and cyclization steps has been demonstrated in literature for similar conjugated systems, suggesting that transitioning from gram-scale laboratory synthesis to kilogram or ton-scale commercial production is feasible with standard process engineering adjustments. This scalability ensures that the supply can meet the growing demand for organic photovoltaic materials as the market expands.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology in organic solar cell manufacturing. These answers are derived directly from the experimental data and beneficial effects described in patent CN109134513B, providing a factual basis for decision-making.
Q: What is the photoelectric conversion efficiency of this non-fullerene acceptor?
A: According to patent CN109134513B, the organic solar cell devices fabricated using these fused-ring benzothiadiazole non-fullerene acceptors have demonstrated single-cell conversion efficiencies reaching nearly 16%, with specific examples like ZYQ3 achieving 14.38% when paired with PCE12.
Q: How does the solubility of this material compare to traditional fullerene acceptors?
A: The material exhibits excellent solubility in common organic solvents such as chloroform, o-dichlorobenzene, and tetrahydrofuran. This superior solubility facilitates solution processing and film formation, addressing a common limitation of rigid fullerene derivatives.
Q: What are the key structural features that enhance performance?
A: The core features a nitrogen-bridged trapezoidal fused ring structure which increases electron cloud density and charge mobility. Additionally, the introduction of alkyl chains on the nitrogen atoms improves regional flatness and solubility, while electron-withdrawing end groups broaden the absorption spectrum.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fused-Ring Benzothiadiazole Acceptor Supplier
As the global demand for sustainable energy solutions accelerates, the need for high-performance organic semiconductor materials has never been more critical. NINGBO INNO PHARMCHEM stands ready to support your R&D and production goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for electronic grade materials, ensuring that every batch of fused-ring benzothiadiazole acceptor delivers consistent photoelectric performance. We understand the nuances of handling air-sensitive intermediates and palladium-catalyzed reactions, allowing us to deliver materials that are ready for immediate device fabrication.
We invite you to contact our technical procurement team to discuss how we can tailor our synthesis capabilities to your specific formulation requirements. By requesting a Customized Cost-Saving Analysis, you can gain insights into how our optimized manufacturing processes can lower your overall material costs without compromising quality. We encourage potential partners to reach out for specific COA data and route feasibility assessments to validate the compatibility of our ZYQ3 or ZYQ4 analogues with your existing donor polymer systems. Let us collaborate to bring the next generation of high-efficiency organic solar cells to market.
