Revolutionizing [6.5.5] Indoline Synthesis: A Cost-Effective Cobalt-Catalyzed Strategy for Complex Pharmaceutical Intermediates
Revolutionizing [6.5.5] Indoline Synthesis: A Cost-Effective Cobalt-Catalyzed Strategy for Complex Pharmaceutical Intermediates
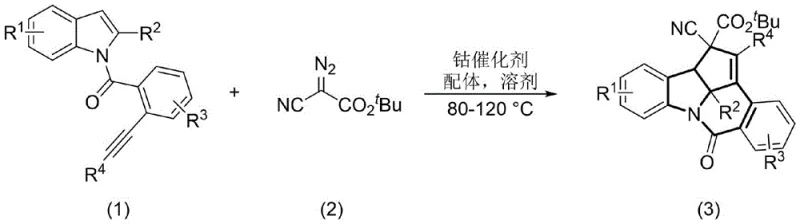
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, sustainable, and cost-effective synthetic methodologies. A recent breakthrough detailed in patent CN116217572A introduces a highly innovative preparation method for multi-condensed ring indoline derivatives, specifically targeting the challenging [6.5.5] aza-condensed ring skeleton. This structural motif is ubiquitous in bioactive natural products and potent drug candidates, including antitumor inhibitors and treatments for rheumatic heart disease. By leveraging a cobalt-catalyzed cycloaddition reaction between alkynyl indole derivatives and tert-butyl diazocyanoacetate, this technology offers a streamlined pathway that addresses many of the bottlenecks associated with traditional heterocycle synthesis. For R&D directors and procurement managers alike, understanding the implications of this cobalt-mediated transformation is crucial for optimizing supply chains and reducing the cost of goods sold (COGS) in complex API manufacturing.
![Chemical structures of [6.5.5] aza-condensed ring skeletons found in natural products like Meloscine and Scandine](/insights/img/655-aza-ring-cobalt-catalysis-pharma-supplier-20260307030612-01.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polycyclic indoline frameworks, particularly those featuring the intricate [6.5.5] fused ring system, has been a formidable challenge in organic synthesis. Conventional strategies often rely on multi-step sequences that involve harsh reaction conditions, expensive transition metal catalysts like palladium or rhodium, and laborious purification processes. These traditional routes frequently suffer from low atom economy and generate significant amounts of chemical waste, which poses both environmental and economic burdens. Furthermore, the sensitivity of intermediates in long synthetic pathways can lead to inconsistent yields and difficulties in scaling up production, creating volatility in the supply of critical pharmaceutical intermediates. The complexity of these molecules often necessitates specialized expertise and extended development timelines, delaying the entry of new therapeutics into the market.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the technology disclosed in CN116217572A presents a paradigm shift through a direct, one-step cycloaddition strategy. By utilizing alkynyl-containing indole derivatives and tert-butyl diazocyanoacetate as readily accessible starting materials, this method achieves the rapid assembly of the [6.5.5] skeleton under relatively mild thermal conditions. The use of earth-abundant cobalt as the catalyst represents a significant strategic advantage, replacing costly precious metals with a more sustainable and economically viable alternative. This approach not only simplifies the operational workflow but also enhances the overall robustness of the synthesis, making it highly attractive for industrial applications where reliability and cost-efficiency are paramount.
Mechanistic Insights into Cobalt-Catalyzed Cycloaddition
The efficacy of this synthetic route lies in the unique reactivity of the cobalt catalyst in conjunction with specific ligands and solvents. Under the optimized reaction conditions, typically ranging from 70°C to 120°C, the cobalt species facilitates the generation of a reactive carbene intermediate from the diazo compound. This electrophilic carbene species then undergoes a highly selective addition across the alkyne triple bond and the indole double bond of the substrate. This concerted or stepwise cyclization process effectively stitches together the three rings of the [6.5.5] system in a single operation, bypassing the need for discrete ring-closing steps that characterize older methodologies. The choice of ligand, such as BINAP or triphenylphosphine, plays a critical role in modulating the electronic environment of the cobalt center, thereby enhancing catalytic turnover and ensuring high regioselectivity.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity intermediates. The direct nature of the transformation minimizes the formation of side products that typically arise from multi-step functional group manipulations. Furthermore, the reaction demonstrates broad substrate tolerance, accommodating various substituents on the indole and phenyl rings without significant loss in efficiency. This versatility implies that the process can be adapted to synthesize a diverse library of analogues, which is invaluable for medicinal chemistry campaigns aiming to optimize structure-activity relationships (SAR). The ability to achieve clean conversions with easily separable products significantly reduces the burden on downstream purification units, contributing to a more streamlined manufacturing process.
How to Synthesize [6.5.5] Aza-Condensed Ring Indoline Derivatives Efficiently
Implementing this cobalt-catalyzed protocol requires careful attention to reaction parameters to maximize yield and purity. The process begins with the preparation of the alkynyl indole precursor, which can be efficiently synthesized via Sonogashira coupling followed by desilication, as illustrated in the patent examples. Once the substrate is secured, the cycloaddition is performed by combining the indole derivative with an excess of tert-butyl diazocyanoacetate in a suitable organic solvent such as toluene or ethyl acetate. The addition of a cobalt salt and a phosphine or nitrogen-based ligand initiates the catalytic cycle upon heating. Following the reaction completion, standard workup procedures involving solvent removal and column chromatography afford the target [6.5.5] fused ring products in good to excellent yields.
- Prepare the alkynyl-containing indole derivative substrate via Sonogashira coupling and desilication.
- Combine the substrate with tert-butyl diazocyanoacetate, a cobalt catalyst (e.g., CoI2), and a ligand (e.g., BINAP) in an organic solvent.
- Heat the mixture to 70-120°C for 7-24 hours, then purify the resulting [6.5.5] skeleton product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cobalt-catalyzed methodology translates into tangible strategic benefits that extend beyond mere chemical novelty. The shift from precious metal catalysts to cobalt represents a direct opportunity for cost reduction in API manufacturing, as the raw material costs for cobalt salts are substantially lower than those for palladium or rhodium complexes. Additionally, the simplification of the synthetic route from multiple steps to a single pot reaction drastically reduces the consumption of solvents and reagents, leading to lower waste disposal costs and a smaller environmental footprint. This efficiency gain allows for more competitive pricing models for the final pharmaceutical intermediates, enhancing the overall value proposition for downstream partners.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and the reduction in synthetic steps significantly lower the direct material costs associated with production. By avoiding complex multi-stage sequences, manufacturers can reduce labor hours and energy consumption, resulting in substantial operational savings. This economic efficiency makes the production of complex heterocyclic scaffolds more financially viable, allowing for better margin management in volatile markets.
- Enhanced Supply Chain Reliability: The use of readily available starting materials and common solvents ensures a stable supply chain that is less susceptible to geopolitical disruptions often associated with rare earth metals. The robustness of the reaction conditions means that production can be scaled up with greater confidence, reducing the risk of batch failures and ensuring consistent delivery schedules. This reliability is critical for maintaining continuous manufacturing operations and meeting the stringent deadlines of pharmaceutical clients.
- Scalability and Environmental Compliance: The straightforward workup procedure, which primarily involves solvent evaporation and chromatography, is highly amenable to scale-up from laboratory to pilot and commercial scales. The reduced generation of hazardous waste aligns with increasingly strict environmental regulations, minimizing the compliance burden on manufacturing facilities. This green chemistry approach not only safeguards the environment but also future-proofs the production process against tightening regulatory standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the technology's capabilities and limitations for potential adopters.
Q: What are the primary advantages of using cobalt catalysis over precious metals for indoline synthesis?
A: Cobalt catalysts offer a significantly lower cost profile compared to rhodium, palladium, or iridium systems, while maintaining high efficiency for constructing complex [6.5.5] skeletons. This substitution drastically reduces raw material costs without compromising yield or purity.
Q: How does this one-step cycloaddition improve upon traditional multi-step synthetic routes?
A: Traditional methods often require lengthy sequences with harsh conditions to build polycyclic frameworks. This patent describes a direct one-step construction from alkynyl indoles, simplifying the workflow, reducing waste generation, and shortening the overall production timeline.
Q: Is this method suitable for large-scale commercial production of API intermediates?
A: Yes, the process utilizes readily available solvents like toluene or ethyl acetate and operates at moderate temperatures (70-120°C). The simple workup involving solvent removal and chromatography indicates strong potential for scalable manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable [6.5.5] Aza-Condensed Ring Indoline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the cobalt-mediated cycloaddition described in CN116217572A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes are translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of [6.5.5] aza-condensed ring intermediates meets the highest standards required for pharmaceutical applications. We are dedicated to helping our clients navigate the complexities of modern drug development with efficient and reliable manufacturing solutions.
We invite you to explore how our expertise in cobalt catalysis and heterocycle synthesis can optimize your project timelines and budgets. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can serve as your trusted partner in bringing next-generation therapeutics to market.
