Scalable Green Synthesis of 3-Aryl-2H-benzo[b][1,4]benzoxazine-2-one Derivatives for Pharmaceutical Applications
Introduction to Advanced Benzoxazine Synthesis
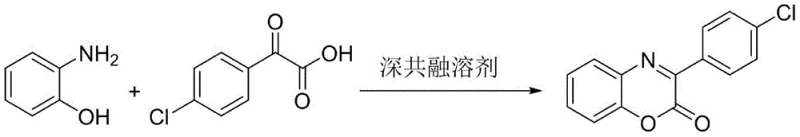
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing heterocyclic scaffolds that possess significant biological activity, such as the 3-aryl-2H-benzo[b][1,4]benzoxazine-2-one core. This specific heterocyclic framework is renowned for its diverse pharmacological profile, exhibiting potent antibacterial, antitumor, and photophysical properties that make it a critical building block for next-generation therapeutic agents. Addressing the urgent need for sustainable manufacturing, patent CN111100085A introduces a groundbreaking preparation method that leverages Deep Eutectic Solvents (DES) to drive the condensation reaction between 2-aminophenol derivatives and keto acids. This innovation represents a paradigm shift away from traditional catalytic systems, offering a pathway that is not only chemically efficient but also aligns perfectly with the principles of green chemistry by eliminating the need for hazardous organic solvents and expensive transition metal catalysts.
![General reaction scheme for the synthesis of 3-aryl-2H-benzo[b][1,4]benzoxazine-2-one using deep eutectic solvent](/insights/img/3-aryl-benzoxazine-synthesis-pharma-supplier-20260303130011-03.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzoxazine derivatives has relied heavily on methodologies that present significant operational and environmental challenges for large-scale manufacturing. Traditional routes often involve the use of strong acidic or basic catalysts, transition metal complexes, or harsh organic solvents that require rigorous removal steps to meet pharmaceutical purity standards. Furthermore, alternative strategies, such as the ring-opening of benzoxazoles followed by reaction with keto acids, frequently suffer from limited substrate scope and poor atom economy. These conventional processes often generate substantial amounts of chemical waste, necessitating complex downstream purification protocols that increase both the production timeline and the overall cost of goods sold, thereby creating bottlenecks for supply chain reliability and cost-effective procurement.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a Deep Eutectic Solvent composed of choline chloride and urea in a precise 1:2 molar ratio, which serves a dual function as both the reaction medium and the promoter. This system operates under remarkably mild thermal conditions, typically requiring heating to only 80°C in an oil bath with magnetic stirring, which drastically reduces energy consumption compared to high-temperature reflux methods. The absence of additional catalysts simplifies the reaction mixture, allowing for a straightforward workup procedure where the target product can be isolated via standard column chromatography and vacuum drying. This streamlined process not only enhances the safety profile of the operation by removing volatile organic compounds but also ensures a cleaner reaction profile with fewer byproducts, directly translating to higher purity outputs and reduced waste treatment burdens.
Mechanistic Insights into DES-Mediated Cyclization
The efficacy of this synthetic route lies in the unique physicochemical properties of the Deep Eutectic Solvent, which facilitates the condensation through a network of hydrogen bonding interactions. The choline chloride and urea mixture creates a highly polar environment that effectively activates the carbonyl group of the keto acid substrate, making it more susceptible to nucleophilic attack by the amino group of the 2-aminophenol derivative. This activation lowers the energy barrier for the initial imine formation, which is subsequently followed by an intramolecular cyclization involving the phenolic hydroxyl group to close the oxazine ring. The stability of the intermediate species within the DES matrix prevents side reactions such as polymerization or hydrolysis, ensuring that the reaction proceeds selectively towards the desired 3-aryl-2H-benzo[b][1,4]benzoxazine-2-one scaffold with high fidelity.
Furthermore, the versatility of this mechanism is evidenced by its tolerance to a wide array of electronic substituents on both the amine and acid components. As illustrated by the general structures of the reactants, the system accommodates electron-withdrawing groups such as halogens and electron-donating groups like methyl moieties without compromising the integrity of the catalytic cycle. Experimental data indicates that substrates bearing electron-withdrawing groups on the keto acid component, such as fluorine or chlorine, tend to exhibit superior reaction kinetics and yield profiles, often reaching conversion rates as high as 98%. This electronic flexibility is crucial for medicinal chemists who need to rapidly generate libraries of analogs for structure-activity relationship (SAR) studies, as it allows for the modular assembly of complex molecules using readily available starting materials without the need for protecting group strategies.
How to Synthesize 3-Aryl-2H-benzo[b][1,4]benzoxazine-2-one Efficiently
Implementing this green synthesis protocol in a laboratory or pilot plant setting requires careful attention to the preparation of the solvent system and the stoichiometry of the reactants to ensure optimal performance. The process begins with the formation of the Deep Eutectic Solvent by heating the choline chloride and urea mixture until a homogenous liquid is achieved, which is then used immediately to dissolve the substrates. Maintaining the reaction temperature at 80°C is critical, as this provides sufficient thermal energy to drive the cyclization without degrading the sensitive heterocyclic product. For detailed operational parameters, including specific molar ratios and purification techniques, please refer to the standardized synthesis guide below which outlines the step-by-step execution of this patented methodology.
- Prepare the deep eutectic solvent by mixing choline chloride and urea in a 1: 2 molar ratio and heating to 80°C until a uniform transparent liquid forms.
- Add the 2-aminophenol derivative and the keto acid derivative to the solvent in a 1: 2 molar ratio and stir magnetically at 80°C for approximately 2 hours.
- Purify the crude reaction mixture using column chromatography with an ethyl acetate and petroleum ether eluent, followed by vacuum drying at 30°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this DES-mediated synthesis offers profound advantages that extend far beyond simple chemical yield improvements. By eliminating the requirement for precious metal catalysts or specialized organometallic reagents, manufacturers can achieve a substantial reduction in raw material costs, insulating the supply chain from the volatility associated with the pricing of rare earth elements and noble metals. Additionally, the use of non-volatile, biodegradable solvent components significantly lowers the regulatory burden and costs associated with hazardous waste disposal and environmental compliance, allowing for a more streamlined audit process and reduced liability for the manufacturing site. This operational simplicity translates directly into a more resilient supply chain, as the reliance on complex, multi-step purification sequences is minimized, thereby reducing the risk of production delays caused by equipment bottlenecks or solvent shortages.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the reaction workflow and the elimination of expensive catalytic systems. Since the Deep Eutectic Solvent acts as both the medium and the promoter, there is no need to purchase, recover, or dispose of costly transition metal catalysts, which traditionally account for a significant portion of the variable costs in fine chemical synthesis. Furthermore, the mild reaction conditions reduce energy consumption requirements for heating and cooling, while the high selectivity of the reaction minimizes the loss of valuable starting materials to side products, ensuring that the overall cost per kilogram of the active pharmaceutical intermediate is significantly optimized for commercial viability.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes to a more stable and predictable supply chain by utilizing commodity chemicals that are globally available in bulk quantities. Choline chloride and urea are produced on a massive industrial scale for other sectors, meaning their availability is not subject to the same supply constraints as specialized fine chemical reagents. This abundance ensures that production schedules can be maintained consistently without the risk of raw material stockouts, while the simplified workup procedure allows for faster batch turnover times, enabling manufacturers to respond more agilely to fluctuating market demands and urgent customer orders for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process from gram-scale laboratory experiments to multi-ton commercial production is inherently safer and more straightforward due to the non-flammable and low-toxicity nature of the solvent system. The absence of volatile organic solvents reduces the risk of fire and explosion in large reactors, lowering insurance premiums and safety infrastructure costs. Moreover, the green credentials of the process align perfectly with increasingly stringent global environmental regulations, facilitating easier approval for new manufacturing sites and ensuring long-term operational continuity without the threat of regulatory shutdowns due to emissions or waste discharge violations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology, providing clarity on its practical application and benefits. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring that stakeholders have access to accurate and verified information. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production lines or for assessing the quality and consistency of the supplied intermediates.
Q: What are the advantages of using Deep Eutectic Solvents (DES) over traditional organic solvents?
A: DES systems, such as the choline chloride/urea mixture described in patent CN111100085A, eliminate the need for volatile organic compounds (VOCs) and toxic metal catalysts. This results in a significantly greener process with easier product isolation and reduced environmental disposal costs.
Q: Does this method tolerate electron-withdrawing and electron-donating groups?
A: Yes, the methodology demonstrates broad substrate scope. Electron-withdrawing groups like halogens on the keto acid generally provide excellent yields (up to 98%), while electron-donating groups are also tolerated, albeit with slightly variable efficiency depending on the specific substitution pattern.
Q: Is the reaction scalable for industrial production?
A: The process utilizes simple equipment (oil bath, magnetic stirring) and mild temperatures (80°C), avoiding high-pressure or cryogenic conditions. This simplicity, combined with the stability of the DES medium, makes the route highly amenable to scale-up from laboratory to commercial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aryl-2H-benzo[b][1,4]benzoxazine-2-one Supplier
As the demand for high-quality heterocyclic intermediates continues to grow within the pharmaceutical sector, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthesis technologies and reliable supply. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to deliver materials that meet the most stringent purity specifications required for drug development. Our rigorous QC labs employ advanced analytical techniques to verify the identity and purity of every batch, guaranteeing that the 3-aryl-2H-benzo[b][1,4]benzoxazine-2-one derivatives we supply are free from critical impurities and fully compliant with international regulatory standards.
We invite potential partners to engage with our technical team to discuss how this green synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits this methodology offers for your specific supply chain. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and commercial reliability.
