Revolutionizing N-Difluoromethyl Compound Production via Mild Photocatalytic Technology for Global Pharma Supply
Introduction to Next-Generation Difluoromethylation Technology
The integration of fluorine atoms into organic molecules has long been a cornerstone strategy in modern medicinal chemistry, particularly for enhancing metabolic stability and membrane permeability in drug candidates. Among various fluorinated motifs, the difluoromethyl group (-CF2H) has emerged as a critical pharmacophore, often serving as a lipophilic hydrogen bond donor that mimics hydroxyl or thiol groups while offering superior metabolic profiles. However, the efficient installation of this group onto nitrogen heterocycles has historically presented significant synthetic challenges, often requiring harsh conditions or prohibitively expensive reagents. The recent disclosure in patent CN110483406B introduces a transformative approach to this problem, detailing a universal, simple, and highly efficient method for synthesizing N-difluoromethyl-containing compounds. This technology leverages visible-light photocatalysis to drive the reaction under exceptionally mild conditions, representing a paradigm shift from thermal activation to photochemical activation. For global pharmaceutical manufacturers and agrochemical developers, this innovation offers a robust pathway to access complex fluorinated intermediates with improved safety profiles and reduced operational costs, addressing a critical bottleneck in the supply chain of high-value fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this technological breakthrough, the synthesis of N-difluoromethyl compounds was plagued by severe operational constraints and economic inefficiencies that hindered large-scale adoption. As illustrated in the prior art, early methods relied heavily on aggressive reagents and extreme reaction parameters that are ill-suited for modern green chemistry standards. For instance, established protocols utilized chlorodifluoromethyl phenylsulfone as the difluoromethylating agent, which is not only costly but also generates stoichiometric amounts of sulfone waste, complicating downstream purification and waste management.
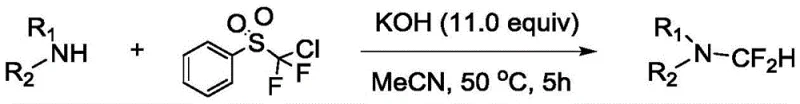
Furthermore, alternative strategies attempted to overcome reactivity issues by employing extreme thermal energy, with some processes requiring temperatures as high as 170°C in triglyme solvents. Such high-temperature operations impose immense stress on reactor equipment, increase energy consumption drastically, and pose significant safety risks regarding thermal runaway, making them economically unviable for the production of heat-sensitive pharmaceutical intermediates. Additionally, methods utilizing phosphonium ylides as reagents, while effective, suffer from the drawback of using extremely expensive starting materials that render the final product cost-prohibitive for commercial applications. These cumulative factors—high cost, harsh conditions, and poor atom economy—have created an urgent demand for a more sustainable and economically feasible synthetic route.
The Novel Approach
The methodology described in patent CN110483406B effectively dismantles these barriers by introducing a photoredox catalytic system that operates at ambient temperature. By utilizing ethyl bromodifluoroacetate as the difluoromethyl source, the process accesses a reagent that is significantly cheaper and more commercially available than traditional sulfones or ylides. The core innovation lies in the use of a specialized copper-based photosensitizer activated by 15W blue LED light, which generates the necessary reactive radical species without the need for external heating. This shift allows the reaction to proceed smoothly at room temperature (rt) over a period of 6 to 24 hours, typically completing within 12 hours.
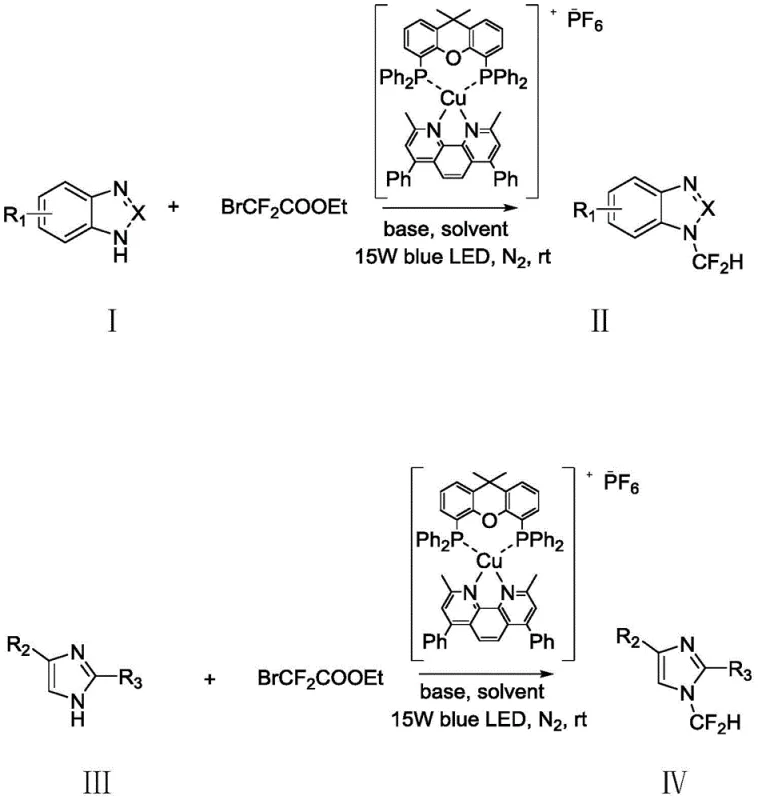
The versatility of this new approach is demonstrated by its broad substrate scope, successfully accommodating various nitrogen nucleophiles including benzimidazoles, imidazoles, and tetrazoles. The reaction tolerates a wide array of functional groups, such as electron-withdrawing nitro and trifluoromethyl substituents, which often deactivate substrates in conventional nucleophilic substitutions. By replacing thermal energy with photon energy and substituting expensive reagents with commodity chemicals, this novel route provides a direct, scalable, and cost-effective solution for the manufacturing of N-difluoromethyl building blocks, aligning perfectly with the industry's push towards greener and more efficient synthesis.
Mechanistic Insights into Copper-Catalyzed Photoredox Difluoromethylation
The success of this synthetic strategy is rooted in the sophisticated interplay between the copper photocatalyst and the ethyl bromodifluoroacetate reagent under visible light irradiation. The mechanism initiates when the copper complex absorbs photons from the blue LED source, transitioning to an excited state capable of single-electron transfer (SET). In this excited state, the catalyst interacts with the ethyl bromodifluoroacetate, facilitating the homolytic cleavage of the carbon-bromine bond. This critical step generates a reactive difluoromethyl radical species along with an ester byproduct, bypassing the high energy barriers associated with thermal decomposition. The generated radical then attacks the nitrogen center of the nucleophilic substrate, forming a new C-N bond and establishing the difluoromethyl motif.
Following the radical addition, the catalytic cycle is closed through a subsequent oxidation or proton transfer step, regenerating the ground-state copper catalyst and releasing the final N-difluoromethylated product. A key advantage of this mechanistic pathway is its high chemoselectivity; the mild nature of the radical generation prevents the degradation of sensitive functional groups on the substrate ring systems. For example, substrates containing nitro groups, which are prone to reduction under harsh thermal or metallic conditions, remain intact throughout the process. Furthermore, the use of mild inorganic bases like potassium phosphate ensures that the reaction medium remains non-corrosive, minimizing equipment wear and tear. This precise control over radical generation and propagation ensures a clean impurity profile, reducing the burden on downstream purification processes and enhancing the overall yield and purity of the target pharmaceutical intermediates.
How to Synthesize N-Difluoromethyl Compounds Efficiently
The practical implementation of this photocatalytic method is designed for straightforward execution in standard laboratory or pilot plant settings, requiring minimal specialized equipment beyond a visible light source. The protocol involves charging a reaction vessel with the nitrogen nucleophile, the copper photosensitizer, and a base, followed by the addition of the solvent and difluoromethylating agent under an inert atmosphere. The simplicity of the work-up procedure, which typically involves silica gel chromatography after solvent removal, further enhances its appeal for process chemists looking to rapidly iterate on lead compounds. For a comprehensive understanding of the specific molar ratios, solvent choices, and purification parameters detailed in the patent, please refer to the standardized synthesis guide below.
- Combine the nitrogen nucleophile (such as benzimidazole or imidazole derivatives), a copper-based photosensitizer, and an alkaline substance like potassium phosphate in a reaction vessel.
- Under a nitrogen atmosphere, add ethyl bromodifluoroacetate as the difluoromethylation reagent and a polar aprotic solvent such as DMF or acetonitrile.
- Irradiate the reaction mixture with 15W blue LED light at room temperature for 6 to 24 hours, followed by standard purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photocatalytic technology translates into tangible strategic benefits that extend far beyond the laboratory bench. The primary driver of value is the drastic reduction in raw material costs achieved by switching from proprietary, high-cost difluoromethylating agents to ethyl bromodifluoroacetate, a bulk commodity chemical. This substitution eliminates the supply chain volatility associated with niche fluorinating reagents, ensuring a stable and predictable sourcing channel for critical intermediates. Moreover, the elimination of high-temperature processing removes the need for energy-intensive heating systems and specialized high-pressure reactors, directly lowering the utility overheads associated with manufacturing.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the replacement of expensive reagents like chlorodifluoromethyl phenylsulfone or phosphonium ylides with inexpensive ethyl bromodifluoroacetate. By removing the dependency on these high-cost inputs, manufacturers can significantly lower the Bill of Materials (BOM) for N-difluoromethyl intermediates. Additionally, the mild reaction conditions reduce energy consumption by eliminating the need for prolonged heating at elevated temperatures, leading to substantial operational expenditure (OpEx) savings. The simplified purification process, facilitated by the clean reaction profile, further reduces solvent usage and waste disposal costs, contributing to a leaner and more profitable production model.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the use of widely available commodity chemicals mitigates the risk of raw material shortages that often plague the production of specialty fluorinated compounds. Ethyl bromodifluoroacetate is produced on a large industrial scale, ensuring consistent availability and price stability compared to custom-synthesized sulfones. The robustness of the reaction across diverse substrates means that a single manufacturing line can be adapted to produce a wide variety of intermediates without extensive retooling, increasing asset utilization rates. This flexibility allows suppliers to respond more agilely to fluctuating market demands, reducing lead times for customers seeking complex fluorinated building blocks for drug development programs.
- Scalability and Environmental Compliance: The inherent safety of operating at room temperature and atmospheric pressure makes this process ideally suited for scale-up from kilogram to multi-ton quantities. The absence of thermal hazards simplifies the engineering controls required for large-scale reactors, accelerating the timeline from process development to commercial production. Furthermore, the method aligns with green chemistry principles by utilizing visible light as a traceless reagent and generating less hazardous waste compared to traditional methods. This environmental compatibility facilitates easier regulatory approval and reduces the carbon footprint of the manufacturing process, a critical factor for multinational corporations committed to sustainability goals.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their existing workflows, we have compiled answers to common inquiries regarding the reaction scope, safety, and scalability. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable foundation for process assessment. Understanding these details is crucial for R&D directors and process engineers planning the transition from legacy synthesis routes to this advanced photocatalytic platform.
Q: What are the primary advantages of this photocatalytic method over traditional difluoromethylation techniques?
A: Unlike traditional methods that require harsh conditions such as high temperatures (up to 170°C) or expensive reagents like chlorodifluoromethyl phenylsulfone, this patented process operates at room temperature using visible light and inexpensive ethyl bromodifluoroacetate, significantly lowering energy consumption and raw material costs.
Q: Which types of nitrogen-containing substrates are compatible with this synthesis route?
A: The method demonstrates excellent substrate adaptability, successfully difluoromethylating a wide range of nitrogen nucleophiles including benzimidazoles, benzotriazoles, imidazoles, tetrazoles, and secondary amines, even those with sensitive functional groups like nitro or trifluoromethyl substituents.
Q: Is this photocatalytic process suitable for large-scale commercial manufacturing?
A: Yes, the reaction conditions are inherently scalable; operating at room temperature without high pressure eliminates significant safety risks, and the use of standard blue LED light sources allows for straightforward adaptation to flow chemistry or large batch reactors for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Difluoromethyl Compound Supplier
As the pharmaceutical industry continues to prioritize fluorinated motifs in drug discovery, the ability to reliably source high-quality N-difluoromethyl intermediates is a critical competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this technological evolution, leveraging advanced photocatalytic methodologies to deliver superior products with unmatched consistency. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of global supply chains. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for API synthesis, minimizing the risk of downstream failures.
We invite procurement leaders and R&D partners to collaborate with us to optimize their supply chains for fluorinated intermediates. By leveraging our expertise in this novel photocatalytic route, we can offer a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating exactly how switching to our process can improve your margins. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our innovative manufacturing capabilities can accelerate your path to market.
