Advanced Synthesis of Monofluoromethylselenide Reagents for Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries are constantly seeking novel building blocks to enhance the metabolic stability and lipophilicity of drug candidates. Patent CN113214124B, published in early 2023, introduces a groundbreaking class of monofluoromethylselenide reagents represented by general structural formula (I). This innovation addresses a critical gap in organic synthesis by providing a direct, efficient, and metal-free method to introduce the unique SeCH2F moiety into complex molecular architectures. The SeCH2F group serves as a potent bioisostere, offering distinct electronic and lipophilic properties that differ significantly from its trifluoromethyl and difluoromethyl counterparts, thereby expanding the chemical space available for medicinal chemists. The patent details a robust synthetic pathway that avoids the use of expensive fluorinating agents and harsh reaction conditions typically associated with selenium chemistry.

For R&D directors evaluating new synthetic routes, the distinction between conventional limitations and this novel approach is paramount. Historically, introducing fluoromethylselenyl groups has been fraught with challenges, often relying on indirect methods that require substrates to be pre-functionalized with selenium atoms. Conventional literature describes processes necessitating strong bases or powerful reducing agents, which can compromise sensitive functional groups on the scaffold. Furthermore, traditional fluoromethylation frequently employs costly reagents like fluoroiodomethane, driving up the raw material costs significantly. In contrast, the novel approach outlined in this patent utilizes common sodium aryl sulfinates and elemental selenium powder as the primary starting materials. This shift not only democratizes access to these specialized reagents but also eliminates the need for transition metal catalysts, thereby removing the burden of heavy metal removal during downstream processing.

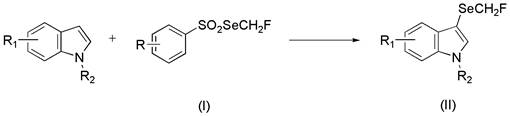
Mechanistically, the synthesis of these arylsulfonate monofluoromethylselenide compounds relies on a carefully orchestrated two-step sequence that leverages specific solvent effects to drive reactivity. The first step involves the selenization of sodium aryl sulfinate with selenium powder in an amine solvent, such as diethylamine or triethylamine, at mild temperatures ranging from 20°C to 40°C. This step generates a reactive selenized intermediate, likely a sodium selenobenzenesulfonate species, which is isolated via simple filtration. The second step is the crucial fluoromethylation, where the isolated intermediate reacts with chlorofluoromethane gas in an amide solvent like DMF or DMA at elevated temperatures of 60°C to 80°C. Unlike nucleophilic fluoromethylation strategies using silicon-based reagents, this process utilizes chlorofluoromethane as an electrophile, representing a fundamental mechanistic divergence that allows for broader substrate tolerance. The resulting reagents are then applied in photocatalytic reactions to functionalize indole derivatives at the 3-position, demonstrating high regioselectivity and compatibility with diverse substituents.
How to Synthesize Arylsulfonate Monofluoromethylselenide Efficiently
To implement this technology in a laboratory or pilot plant setting, operators must adhere to the specific solvent switching protocol defined in the patent to ensure high yields and purity. The process begins with the preparation of the selenized intermediate under inert atmosphere conditions to prevent oxidation, followed by a rigorous workup involving ether washing to remove unreacted amine. The subsequent fluoromethylation step requires careful handling of chlorofluoromethane gas, typically introduced via balloon or pressure equipment, ensuring the reaction mixture reaches the optimal thermal window for substitution. Detailed standardized operating procedures regarding stoichiometry, specifically the 1:1 molar ratio of sulfinate to selenium, are critical for reproducibility. For the complete step-by-step technical guide including exact quantities and safety protocols, please refer to the section below.
- Perform selenization of sodium aryl sulfinate with selenium powder in an amine solvent at room temperature.
- Filter and isolate the selenized intermediate solid via suction filtration and ether washing.
- React the isolated intermediate with chlorofluoromethane gas in an amide solvent at elevated temperatures (60-80°C).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthetic route offers substantial strategic benefits regarding cost structure and supply chain resilience. The primary cost driver in traditional organoselenium synthesis is often the specialized fluorinating agent or the complex multi-step preparation of selenium-containing precursors. By utilizing sodium aryl sulfinates and elemental selenium, both of which are commodity chemicals available from multiple global suppliers, manufacturers can drastically reduce raw material expenditure. Furthermore, the elimination of transition metal catalysts removes the need for expensive scavenging resins and extensive purification steps required to meet stringent pharmaceutical impurity limits. This simplification of the downstream processing workflow translates directly into reduced operational expenditures and shorter batch cycle times, enhancing overall manufacturing efficiency without compromising product quality.
- Cost Reduction in Manufacturing: The replacement of expensive fluoroiodomethane with inexpensive chlorofluoromethane gas represents a significant variable cost saving. Additionally, the metal-free nature of the reaction eliminates the capital and operational costs associated with metal removal technologies, such as specialized filtration systems or chromatography columns dedicated to scavenging heavy metals. The simplicity of the workup, which involves basic extraction and crystallization rather than complex distillation or preparative HPLC, further lowers the utility and labor costs per kilogram of produced reagent. These cumulative efficiencies allow for a more competitive pricing structure for the final active pharmaceutical ingredients derived from these intermediates.
- Enhanced Supply Chain Reliability: Relying on commodity starting materials like selenium powder and sodium sulfinates mitigates the risk of supply disruptions often associated with bespoke or highly specialized reagents. Since the synthesis does not depend on a single source of a complex catalyst, procurement teams can diversify their supplier base for raw materials, ensuring continuity of supply even during market fluctuations. The robustness of the reaction conditions, which tolerate a wide range of temperatures and do not require cryogenic cooling or ultra-high pressure, means that production can be easily transferred between different manufacturing sites or CDMO partners without significant re-validation efforts, thereby securing the supply chain against geopolitical or logistical bottlenecks.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard unit operations such as stirring, filtration, and gas sparging, which are easily replicated from gram to ton scale. The absence of toxic heavy metals in the reaction mixture simplifies waste stream management, reducing the environmental footprint and the costs associated with hazardous waste disposal. The use of common organic solvents like DMF and ethyl acetate facilitates solvent recovery and recycling programs, aligning with modern green chemistry principles and regulatory expectations for sustainable manufacturing practices. This environmental compliance is increasingly becoming a key differentiator for suppliers seeking to partner with top-tier pharmaceutical companies committed to sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this monofluoromethylselenide technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, focusing on practical application scenarios. Understanding these nuances is essential for project managers assessing the feasibility of integrating this new reagent class into existing drug discovery pipelines. The responses highlight the operational flexibility and the specific chemical advantages that distinguish this method from prior art.
Q: What are the advantages of this new reagent over traditional trifluoromethylselenylation methods?
A: Unlike traditional methods requiring expensive fluoroiodomethane or strong reducing agents, this patent utilizes cheap chlorofluoromethane and operates under mild, metal-free conditions, significantly simplifying post-treatment.
Q: Is the synthesis scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process uses commercially available starting materials like sodium aryl sulfinate and selenium powder, with reaction temperatures between 20°C and 80°C, making it highly suitable for commercial scale-up.
Q: What specific substrates can be functionalized using this reagent?
A: The reagent successfully functionalizes indole derivatives at the 3-position under photocatalytic conditions, accommodating various substituents such as halogens, cyano groups, and alkyl chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Monofluoromethylselenide Reagent Supplier
As the demand for fluorinated and selenated building blocks grows in the development of next-generation therapeutics, having a partner with deep technical expertise is crucial. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to clinical supply is seamless. Our rigorous QC labs and adherence to stringent purity specifications guarantee that every batch of monofluoromethylselenide reagent meets the highest standards required for GMP manufacturing. We understand the critical nature of supply continuity for API intermediates and have established robust inventory management systems to support your long-term production needs.
We invite you to engage with our technical procurement team to discuss how this novel synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this metal-free protocol for your specific molecule. We encourage you to contact us today to obtain specific COA data for our available reagents and to schedule a consultation for detailed route feasibility assessments, ensuring your project stays on track and within budget.
