Revolutionizing Organic Thioether Production: A Metal-Free Strategy for High-Purity Pharmaceutical Intermediates
Revolutionizing Organic Thioether Production: A Metal-Free Strategy for High-Purity Pharmaceutical Intermediates
The landscape of fine chemical synthesis is undergoing a paradigm shift towards greener, more sustainable methodologies, particularly in the production of high-value scaffolds like organic thioethers. Patent CN116354781A introduces a groundbreaking approach that eliminates the reliance on transition metal catalysis, a longstanding bottleneck in the manufacturing of sulfur-containing heterocycles and acyclic thioethers. This innovation leverages alkenyl sulfonium salts as versatile precursors, enabling the construction of complex thioether architectures under remarkably mild conditions. For R&D directors and procurement specialists alike, this represents a critical advancement in reducing the environmental footprint while simultaneously enhancing the economic viability of producing key pharmaceutical and agrochemical intermediates. The methodology not only addresses the growing regulatory pressure to minimize heavy metal residues in active pharmaceutical ingredients (APIs) but also streamlines the supply chain by utilizing readily available starting materials.
Organic thioether compounds are ubiquitous in modern medicinal chemistry, serving as essential structural motifs in a vast array of bioactive molecules ranging from antibacterial agents to antidepressants and antitumor drugs. The traditional reliance on transition metals such as palladium or copper for C-S bond formation has often necessitated rigorous purification protocols to meet stringent ppm-level limits for residual metals in final drug products. By circumventing these metal-dependent pathways, the technology disclosed in CN116354781A offers a direct route to high-purity organic thioethers that significantly lowers the barrier for compliance with global pharmacopeial standards. This development is particularly relevant for the synthesis of tetrahydrothiophene derivatives, which are increasingly recognized for their unique pharmacological profiles in enzyme inhibition and receptor modulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of organic sulfide compounds has been dominated by transition metal-catalyzed cross-coupling reactions or nucleophilic substitutions involving toxic sulfur reagents. These conventional pathways are fraught with significant drawbacks that impact both the cost structure and the environmental sustainability of the manufacturing process. The requirement for expensive noble metal catalysts not only inflates the raw material costs but also introduces complex downstream processing challenges, specifically the need for specialized metal scavengers to ensure product purity. Furthermore, many traditional sulfur sources are volatile, malodorous, and hazardous, posing serious safety risks to plant operators and requiring specialized containment infrastructure. The sensitivity of these metal-catalyzed systems to air and moisture often mandates the use of inert atmosphere techniques, which further complicates scale-up operations and increases energy consumption.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a metal-free, base-promoted transformation of alkenyl sulfonium salts. This strategy fundamentally alters the reaction landscape by replacing costly catalytic systems with inexpensive inorganic or alkoxide bases, such as potassium tert-butoxide. The reaction proceeds efficiently under ambient air conditions, eliminating the need for rigorous degassing or nitrogen blanketing, which drastically simplifies reactor setup and operation. The use of stable sulfonium salt intermediates allows for a broader substrate tolerance, accommodating electron-rich and electron-deficient aromatic systems without the risk of catalyst deactivation. This robustness translates directly into higher yields and more consistent batch-to-batch reproducibility, key metrics for any reliable organic thioether supplier aiming to serve the demanding pharmaceutical market.
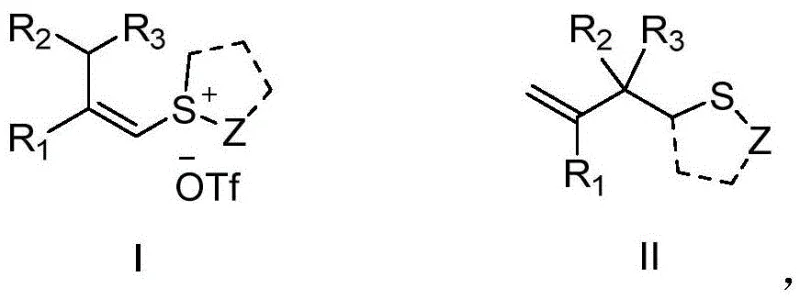
Mechanistic Insights into Base-Promoted Sulfonium Salt Transformation
The core of this innovative synthesis lies in the unique reactivity of the alkenyl sulfonium salt intermediate, which acts as a potent electrophile activated by the presence of a strong base. Upon treatment with a base like t-BuOK, the sulfonium salt undergoes a deprotonation event that triggers a cascade of electronic rearrangements, ultimately leading to the formation of the carbon-sulfur bond characteristic of the target thioether. Unlike radical-based metal catalysis which can suffer from selectivity issues, this ionic pathway offers precise control over the regiochemistry of the substitution. The mechanism avoids the generation of free radical species that often lead to polymerization or side reactions, thereby ensuring a cleaner reaction profile. This mechanistic clarity is crucial for process chemists who need to predict impurity profiles and establish robust control strategies during technology transfer.
Furthermore, the stability of the sulfonium salt precursor allows for the modular assembly of diverse molecular architectures. The leaving group ability of the sulfonium moiety is finely tuned by the adjacent alkene functionality, facilitating the displacement by various nucleophilic centers inherent in the substrate or added reagents. This intrinsic reactivity means that the reaction can tolerate a wide range of functional groups, including halides and ethers, which are commonly found in complex drug candidates. The absence of metal coordination spheres removes the steric constraints often associated with ligand-accelerated catalysis, allowing for the successful transformation of sterically hindered substrates that would otherwise be unreactive. Such mechanistic versatility ensures that this platform technology can be applied to the synthesis of a broad spectrum of high-purity pharmaceutical intermediates with minimal process modification.

How to Synthesize Organic Thioether Compounds Efficiently
Implementing this synthesis protocol in a laboratory or pilot plant setting is straightforward and requires standard equipment available in most organic synthesis facilities. The process begins with the preparation of the alkenyl sulfonium salt, which can be synthesized from readily available sulfoxides and olefins using triflic anhydride, followed by the key base-mediated transformation. The operational simplicity is a major selling point, as it does not require specialized glovebox techniques or high-pressure reactors. Detailed standardized synthesis steps for the efficient production of these compounds are outlined in the guide below, ensuring that technical teams can replicate the high yields reported in the patent literature with confidence.
- Prepare the reaction vessel by adding the alkenyl sulfonium salt substrate and a strong base such as potassium tert-butoxide under ambient air conditions.
- Introduce an appropriate organic solvent like tetrahydrofuran (THF) to the mixture and stir at room temperature for approximately 4 hours to ensure complete conversion.
- Upon completion, remove the solvent under reduced pressure and purify the crude residue via silica gel column chromatography using petroleum ether as the eluent to isolate the high-purity thioether product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of transition metals from the process workflow directly correlates to a substantial reduction in raw material expenditure, as there is no longer a need to procure expensive palladium or copper catalysts along with their proprietary ligands. Moreover, the removal of metal scavenging steps from the downstream processing significantly shortens the production cycle time, allowing for faster turnaround on custom synthesis orders. This efficiency gain enhances the overall agility of the supply chain, enabling manufacturers to respond more rapidly to fluctuating market demands for critical intermediates.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is profound, primarily driven by the drastic simplification of the bill of materials. By removing the dependency on precious metal catalysts, manufacturers can achieve significant cost savings on every kilogram of product produced. Additionally, the simplified purification process reduces the consumption of silica gel and solvents required for metal removal, further lowering the variable costs associated with production. These cumulative savings can be passed down the value chain, making the final API or agrochemical product more competitive in the global marketplace without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals rather than specialized catalytic systems that may be subject to geopolitical supply constraints. The reagents required for this synthesis, such as potassium tert-butoxide and common organic solvents, are widely available from multiple global vendors, reducing the risk of single-source bottlenecks. Furthermore, the robustness of the reaction conditions means that production is less susceptible to delays caused by equipment failures related to inert gas systems or moisture control, ensuring a more consistent and reliable delivery schedule for customers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with the principles of green chemistry, facilitating easier regulatory approval for new manufacturing sites. The absence of heavy metals simplifies waste stream management, as effluent treatment does not require complex precipitation or ion-exchange processes to meet discharge limits. This environmental compatibility reduces the overhead costs associated with waste disposal and regulatory compliance, making the process highly scalable from gram-scale R&D to multi-ton commercial production without the need for extensive retrofitting of existing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential partners.
Q: What are the primary advantages of this metal-free thioether synthesis over traditional methods?
A: The primary advantages include the complete elimination of expensive and toxic transition metal catalysts, operation under mild ambient conditions without the need for inert gas protection, and a simplified purification process that avoids complex metal scavenging steps.
Q: What is the substrate scope for the alkenyl sulfonium salts in this reaction?
A: The method demonstrates a broad substrate scope, accommodating various substituted aryl groups including halogens (F, Cl, Br), alkyl chains, and fused ring systems like naphthalene, as well as different alkyl substituents on the sulfur-containing ring.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method is highly scalable due to its use of inexpensive reagents, tolerance to air and moisture, and straightforward work-up procedures involving standard distillation and chromatography, making it suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Organic Thioether Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthesis technologies in shaping the future of fine chemical manufacturing. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN116354781A can be seamlessly translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the absence of metal contaminants and ensure batch consistency.
We invite forward-thinking pharmaceutical and agrochemical companies to collaborate with us on leveraging this cost-effective synthesis route for their next-generation projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific molecule, along with specific COA data and route feasibility assessments. Let us help you optimize your supply chain and reduce your time-to-market with our advanced organic thioether manufacturing capabilities.
