Advanced Metal-Free Synthesis of Difluoroquinolone Skeletons for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry is constantly seeking robust methodologies to construct complex heterocyclic scaffolds that offer enhanced biological activity and metabolic stability. Patent CN108929267B introduces a groundbreaking synthetic strategy for generating a novel quinolone skeleton featuring a gem-difluoro moiety at the C3 position. This structural motif is highly coveted in medicinal chemistry because the CF2 group serves as an exceptional bioisostere for oxygen or carbonyl functionalities, inducing profound changes in dipole moment, acidity, and molecular conformation. The disclosed method achieves this transformation through a tandem double fluorination and intramolecular cyclization sequence, utilizing commercially available Selectfluor as the fluorine source. By operating under mild conditions without the need for transition metal catalysts, this technology addresses critical pain points regarding metal contamination and process complexity often encountered in the production of high-purity pharmaceutical intermediates.
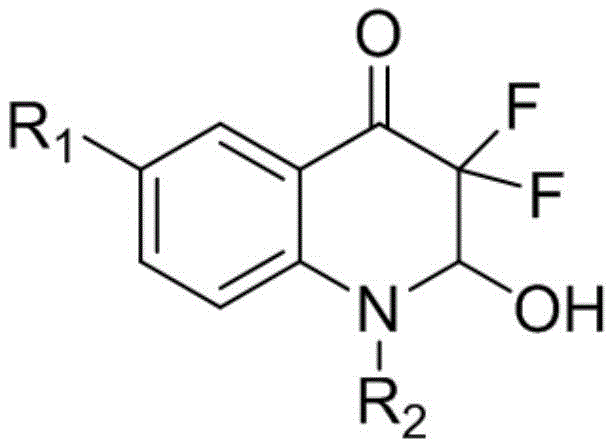
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of quinoline-based frameworks, particularly those resembling azanaphthoquinones or tetrahydroquinolines, has relied heavily on intermolecular 1,4-conjugated organometallic additions to 4-quinolones. These traditional pathways invariably necessitate the use of expensive transition metal catalysts to facilitate the bond formation, introducing significant cost burdens and supply chain vulnerabilities associated with precious metal procurement. Furthermore, the reliance on organometallic reagents often mandates stringent anhydrous conditions and low temperatures, escalating energy consumption and operational hazards in a manufacturing setting. A major drawback for pharmaceutical manufacturers is the subsequent requirement for rigorous purification steps to remove trace transition metal residues to meet strict regulatory limits for active pharmaceutical ingredients. Typically, these legacy processes involve at least three distinct synthetic steps, compounding the loss of material throughput and extending the overall production lead time. The cumulative effect of these inefficiencies results in a higher cost of goods sold and a larger environmental footprint due to increased solvent and reagent usage.
The Novel Approach
In stark contrast, the methodology described in patent CN108929267B offers a streamlined, one-pot solution that bypasses the need for transition metals entirely. The process initiates with a readily accessible enaminone precursor, which undergoes a dual fluorination event followed by spontaneous cyclization to forge the difluoroquinolone core. This approach utilizes a benign solvent system comprising a 1:1 mixture of acetone and water, allowing the reaction to proceed efficiently at room temperature. The elimination of transition metals not only simplifies the downstream purification workflow but also drastically reduces the risk of heavy metal contamination in the final product. By consolidating multiple transformation steps into a single operational unit, the new route significantly improves atom economy and overall process mass intensity. This technological leap enables the rapid assembly of diverse difluoroquinolone analogs with varying substituents at the R1 and R2 positions, facilitating faster structure-activity relationship studies for drug discovery teams.
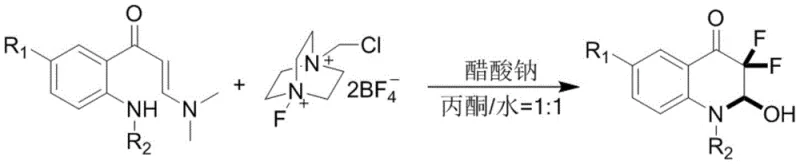
Mechanistic Insights into Selectfluor-Mediated Double Fluorination and Cyclization
The mechanistic pathway elucidated in the patent reveals a sophisticated yet elegant sequence of electrophilic fluorination events driven by the unique reactivity of Selectfluor. Initially, the electron-rich carbon-carbon double bond of the enaminone substrate attacks the fluorinating agent, resulting in the installation of the first fluorine atom and the generation of an iminium ion intermediate. Due to the strong electron-withdrawing nature of the newly introduced fluorine atom, the adjacent enamine functionality becomes activated for a second nucleophilic attack on another equivalent of the fluorinating reagent. This second fluorination step yields a crucial difluoro-iminium intermediate, which sets the stage for the subsequent ring-closing event. The presence of the neighboring amide nitrogen allows for an intramolecular nucleophilic attack on the activated carbonyl or iminium center, effectively closing the six-membered quinolone ring. Finally, hydrolysis of the reactive iminium species by water present in the solvent system releases the dimethylamine byproduct and furnishes the stable 3,3-difluoro-3,4-dihydroquinolin-2(1H)-one scaffold.
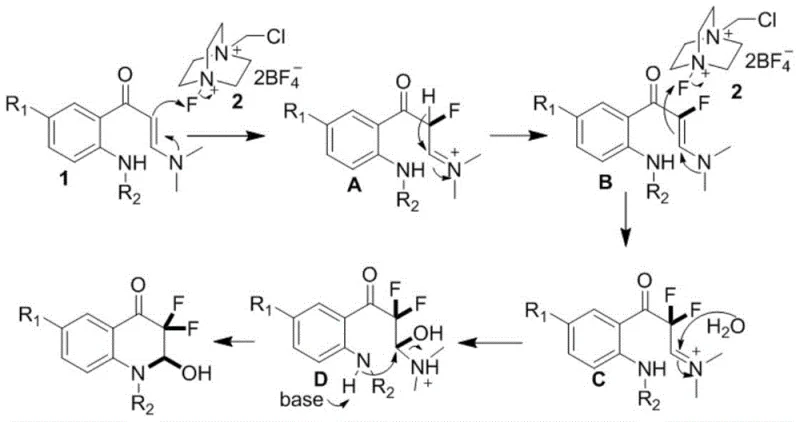
Understanding the impurity profile is critical for scaling this reaction, and the mechanism provides clear insights into potential side reactions. The use of sodium acetate as a base plays a pivotal role in neutralizing the acid generated during the fluorination steps, thereby preventing the decomposition of acid-sensitive functional groups on the aromatic ring. The aqueous component of the solvent system is not merely a diluent but an essential reactant that facilitates the hydrolysis of the intermediate iminium ions to the final ketone product. If the water content is too low, the reaction may stall at the iminium stage, while excessive water could potentially hydrolyze the starting enaminone prematurely. The precise control of stoichiometry, specifically maintaining a molar ratio of raw material to fluorinating agent between 1:2 and 1:3, ensures complete conversion while minimizing the formation of over-fluorinated byproducts. This mechanistic clarity allows process chemists to fine-tune reaction parameters to achieve the reported yields of up to 94%, ensuring a clean impurity profile suitable for GMP manufacturing.
How to Synthesize Difluoroquinolone Skeletons Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and mixing efficiency to replicate the high yields reported in the patent literature. The procedure begins with the dissolution of the specific enaminone raw material, Selectfluor, and sodium acetate in the designated acetone-water solvent mixture. It is imperative to maintain the reaction at ambient temperature to avoid thermal degradation of the sensitive fluorinated intermediates, with reaction times typically ranging from one to three hours depending on the electronic nature of the substituents. Upon completion, as monitored by TLC, the reaction mixture is subjected to standard workup procedures involving column chromatography to isolate the pure white solid product. For detailed operational parameters, safety data, and specific stoichiometric adjustments for different substrates, please refer to the standardized synthesis guide below.
- Mix the enaminone raw material, Selectfluor fluorinating agent, and sodium acetate in a 1: (2-3):(1-2) molar ratio.
- Add a mixed solvent of acetone and water (1: 1 volume ratio) and stir at room temperature for 1 to 3 hours.
- Purify the reaction mixture via column chromatography using ethyl acetate and petroleum ether to isolate the white solid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this metal-free fluorination technology presents a compelling value proposition for reducing the total cost of ownership for complex heterocyclic intermediates. By eliminating the dependency on scarce and volatile transition metal catalysts, manufacturers can insulate their supply chains from geopolitical risks and price fluctuations associated with precious metals like palladium or rhodium. The simplified one-pot nature of the reaction reduces the number of unit operations required, directly translating to lower labor costs and reduced equipment occupancy time in multipurpose manufacturing facilities. Furthermore, the use of acetone and water as solvents aligns with green chemistry principles, significantly lowering the costs associated with solvent recovery and hazardous waste disposal compared to traditional chlorinated or aromatic solvent systems. These cumulative efficiencies enable a more competitive pricing structure for high-purity pharmaceutical intermediates without compromising on quality or regulatory compliance.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts from the process flow eliminates the need for specialized scavenging resins and extensive purification protocols designed to meet strict residual metal limits. This simplification drastically reduces the consumption of auxiliary materials and shortens the batch cycle time, leading to substantial operational expenditure savings. Additionally, the high atom economy of the direct fluorination-cyclization sequence minimizes raw material waste, ensuring that a greater proportion of input costs are converted into valuable saleable product. The ability to run the reaction at room temperature further contributes to cost efficiency by removing the energy load associated with heating or cryogenic cooling systems.
- Enhanced Supply Chain Reliability: Utilizing commodity chemicals such as Selectfluor, sodium acetate, and acetone ensures a robust and diversified supply base that is less susceptible to disruption than specialized organometallic reagents. The stability of the reagents allows for longer shelf-life and easier storage logistics, reducing the risk of production delays caused by expired or degraded materials. Moreover, the scalability of this aqueous-compatible process facilitates seamless technology transfer from laboratory scale to multi-ton commercial production, ensuring consistent supply continuity for downstream API manufacturers. This reliability is crucial for maintaining uninterrupted production schedules in the fast-paced pharmaceutical market.
- Scalability and Environmental Compliance: The replacement of toxic organic solvents with a semi-aqueous system significantly lowers the environmental impact of the manufacturing process, easing the burden on wastewater treatment facilities. The absence of heavy metals simplifies the classification of waste streams, potentially reducing disposal fees and regulatory reporting requirements. From a scale-up standpoint, the exothermic nature of the fluorination can be easily managed in large reactors due to the high heat capacity of the water component, ensuring safe operation even at industrial volumes. This inherent safety and environmental compatibility make the process highly attractive for contract manufacturing organizations aiming to expand their fluorination capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this difluoroquinolone synthesis technology. These answers are derived directly from the experimental data and beneficial effects outlined in patent CN108929267B, providing a reliable foundation for process evaluation. Understanding these details helps stakeholders assess the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of the CF2 group in quinolone skeletons?
A: The CF2 group acts as a bioisostere for oxygen or carbonyl groups, significantly altering dipole moments, acidity, and conformation, which enhances metabolic stability and lipophilicity for drug development.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, unlike conventional methods that rely on transition metal catalysis, this novel approach utilizes Selectfluor and sodium acetate, eliminating the need for expensive metals and complex removal steps.
Q: What is the typical yield range for this fluorination cyclization?
A: The patent data indicates high efficiency, with yields reaching up to 94% under optimized conditions using a simple acetone/water solvent system at room temperature.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluoroquinolone Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting innovative synthetic technologies to deliver superior chemical solutions to the global pharmaceutical market. Our team of expert process chemists has extensively evaluated the metal-free fluorination route described in CN108929267B and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of difluoroquinolone intermediate meets the highest international standards for potency and impurity control.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your supply chain and reduce overall project costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits applicable to your project volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique molecular requirements, ensuring a successful and profitable collaboration.
