Scalable Synthesis of Benzoxazepine Derivatives: A Breakthrough in Pharmaceutical Intermediate Manufacturing
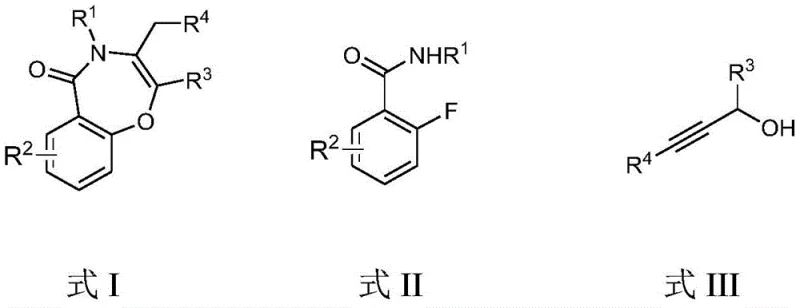
The pharmaceutical industry continuously seeks efficient pathways to access complex heterocyclic scaffolds, particularly the benzoxazepine core, which serves as a privileged structure in numerous bioactive molecules. Patent CN109824620B, published in October 2020, introduces a transformative preparation method for 1,4-benzoxazepin-5(4H)-one derivatives that addresses longstanding challenges in organic synthesis. This innovation leverages a base-mediated cascade reaction between readily available o-fluorobenzamides and propargyl alcohols, bypassing the need for precious metal catalysts. For R&D directors and procurement specialists, this represents a significant shift towards more sustainable and cost-effective manufacturing paradigms. The technology not only streamlines the synthetic route but also enhances the purity profile of the final intermediates, making it an attractive option for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the seven-membered benzoxazepine ring has relied on methodologies that are often fraught with inefficiencies and economic burdens. Traditional approaches frequently employ transition metal catalysts, such as palladium, to facilitate intramolecular carbonylation or coupling reactions. While effective on a small laboratory scale, these methods introduce substantial complications during commercial manufacturing, including the high cost of the catalyst itself and the rigorous downstream processing required to reduce residual metal levels to parts-per-million specifications. Furthermore, alternative routes utilizing reagents like Lawesson's reagent for Schmidt-type reactions generate malodorous sulfur-containing by-products and suffer from poor atom economy. These conventional pathways often involve multi-step sequences with specialized substrates that limit structural diversity, thereby constraining the ability of medicinal chemists to rapidly explore structure-activity relationships (SAR) during drug discovery phases.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a direct nucleophilic aromatic substitution strategy followed by cyclization, driven solely by inexpensive inorganic bases like potassium hydroxide. This approach fundamentally simplifies the supply chain by relying on bulk commodity chemicals rather than specialized organometallic reagents. The reaction proceeds under mild thermal conditions in dimethyl sulfoxide (DMSO), a solvent widely available in the fine chemical industry. By eliminating the reliance on exotic catalysts and complex ligand systems, this novel route drastically reduces the complexity of the reaction setup and the associated safety hazards. The result is a streamlined process that offers superior operational flexibility, allowing for the rapid generation of diverse benzoxazepine libraries essential for modern drug development programs without the logistical overhead of traditional catalytic methods.
Mechanistic Insights into Base-Mediated Cyclization
The core of this technological advancement lies in the elegant mechanistic pathway that converts linear precursors into the cyclic benzoxazepine scaffold. The reaction initiates with the deprotonation of the propargyl alcohol by the strong base, generating a nucleophilic alkoxide species. This alkoxide then attacks the electron-deficient aromatic ring of the o-fluorobenzamide at the position ortho to the fluorine substituent, displacing the fluoride ion in a classic nucleophilic aromatic substitution (SNAr) event. This step forms a crucial ether linkage, setting the stage for the subsequent ring closure. The presence of the amide carbonyl group facilitates the subsequent intramolecular attack by the nitrogen atom or assists in the tautomerization required to finalize the seven-membered lactam ring structure. This cascade occurs with high chemoselectivity, minimizing the formation of polymeric by-products or alternative cyclization modes that often plague alkyne chemistry.
From an impurity control perspective, this mechanism offers distinct advantages for ensuring high-purity pharmaceutical intermediates. The by-product of the defluorination step is potassium fluoride (KF), an inorganic salt that is easily removed during the aqueous workup phase, unlike organic impurities that often co-elute with the product in chromatography. The high specificity of the SNAr reaction ensures that substituents on the benzene ring, such as methoxy or chloro groups, remain intact, preserving the intended molecular architecture. This robustness allows for the tolerance of various functional groups (R1, R2, R3, R4), enabling the synthesis of a wide array of derivatives without compromising the integrity of sensitive moieties. Consequently, the crude reaction mixture is cleaner, reducing the burden on purification units and increasing the overall yield of the isolated target compound.
How to Synthesize 1,4-Benzoxazepin-5(4H)-one Efficiently
To implement this synthesis effectively, precise control over reaction parameters is essential to maximize yield and minimize side reactions. The process begins with the careful selection of anhydrous solvents, as water content exceeding 0.1% can significantly inhibit the reaction efficiency by competing with the nucleophile or hydrolyzing sensitive intermediates. The stoichiometry of the base is critical, with a molar excess of 2.0 to 4.0 equivalents relative to the benzamide substrate recommended to drive the equilibrium towards product formation. The detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining o-fluorobenzamide compound (Formula II) and propargyl alcohol compound (Formula III) in anhydrous dimethyl sulfoxide (DMSO) with a molar ratio of base to substrate between 2.0 and 4.0.
- Conduct the reaction in two temperature stages: first maintain at 30°C for 12 hours to initiate nucleophilic substitution, then raise to 50°C for another 12 hours to complete the intramolecular cyclization.
- Upon completion, quench the reaction with saturated brine, extract with ethyl acetate, dry over anhydrous magnesium sulfate, and purify via silica gel column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route translates into tangible strategic benefits regarding cost stability and operational reliability. The shift away from precious metal catalysis removes a major source of price volatility associated with commodities like palladium, whose market value can fluctuate wildly due to geopolitical factors. By utilizing earth-abundant elements and bulk solvents, the variable cost of goods sold (COGS) becomes more predictable and manageable. Furthermore, the simplified workflow reduces the demand for specialized equipment and highly trained personnel capable of handling air-sensitive catalysts, thereby lowering the barrier to entry for contract manufacturing organizations (CMOs) and enhancing the overall resilience of the supply network.
- Cost Reduction in Manufacturing: The most significant economic driver is the total elimination of expensive transition metal catalysts and phosphine ligands, which traditionally account for a substantial portion of raw material expenses in heterocycle synthesis. Additionally, the generation of potassium fluoride as the primary by-product simplifies waste disposal protocols compared to processes generating heavy metal sludge or toxic sulfur waste. This reduction in hazardous waste treatment fees, combined with the use of low-cost starting materials like o-fluorobenzoyl chloride derivatives and propargyl alcohol, results in substantial cost savings throughout the production lifecycle.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals ensures a robust and continuous supply of raw materials, mitigating the risk of production stoppages due to shortages of specialized reagents. Since the reactants are widely produced by multiple global suppliers, procurement teams can leverage competitive bidding to secure favorable pricing and delivery terms. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in raw material quality, further stabilizing the supply chain against upstream fluctuations and ensuring consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, operating under atmospheric pressure and moderate temperatures that are easily managed in standard stainless steel reactors. The absence of pyrophoric reagents or high-pressure hydrogenation steps enhances plant safety and reduces insurance and compliance costs. Moreover, the high atom economy and the generation of benign inorganic salts align perfectly with green chemistry principles, facilitating easier regulatory approval and reducing the environmental footprint of the manufacturing facility, which is increasingly important for maintaining corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzoxazepine synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering a realistic view of the process capabilities. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this benzoxazepine synthesis method over traditional palladium-catalyzed routes?
A: The primary advantage is the complete elimination of expensive and toxic transition metal catalysts like palladium. This method utilizes inexpensive inorganic bases (KOH/NaOH) and common solvents, significantly reducing raw material costs and simplifying the removal of heavy metal residues, which is critical for pharmaceutical compliance.
Q: How does this process ensure high atom economy and environmental compliance?
A: The reaction produces potassium fluoride (KF) as the sole inorganic by-product, which possesses high atom utilization efficiency. Unlike methods generating complex organic waste or sulfur-containing by-products, this route offers a greener profile with simpler waste treatment protocols, aligning with modern environmental regulations.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method is highly scalable due to its operational simplicity. It requires only standard heating and stirring equipment without the need for specialized high-pressure reactors or inert gas protection typically associated with organometallic chemistry. The robust substrate scope allows for the production of diverse derivatives efficiently.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Benzoxazepin-5(4H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in accelerating drug development timelines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific requirements of this base-mediated cyclization, maintaining stringent purity specifications through our rigorous QC labs. We are committed to delivering high-quality benzoxazepine intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain and reduce your overall production costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your target benzoxazepine derivatives, and let us demonstrate how our expertise can add value to your next project.
