Revolutionizing m-Aminoacetanilide Production: A Green Melt-Process for Global Supply Chains
Introduction to Next-Generation m-Aminoacetanilide Synthesis
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic routes. A pivotal development in this sector is detailed in Chinese patent CN114181103A, which discloses a novel method for synthesizing m-aminoacetanilide by utilizing m-phenylenediamine as both the raw material and the reaction solvent. This approach represents a paradigm shift from conventional liquid-phase reactions that rely heavily on hazardous mineral acids and volatile organic solvents. By operating in a melt state under nitrogen protection, this technology addresses critical pain points regarding environmental compliance and operational safety. For R&D directors and procurement specialists alike, understanding this methodology is essential for securing a competitive edge in the supply of high-value intermediates used in disperse dyes and pharmaceutical formulations.
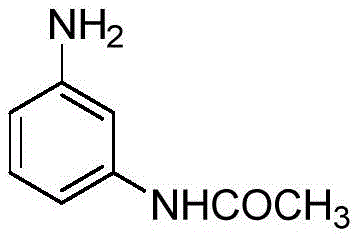
The strategic importance of m-aminoacetanilide cannot be overstated, as it serves as a crucial coupling component for rapid dyes and a key building block for various medical intermediates. The ability to produce this compound with exceptional purity and yield directly impacts the quality of downstream products such as N,N-diethyl m-acetamino aniline. The patent highlights a process that not only streamlines production but also enhances the economic viability of the entire value chain. As we delve deeper into the technical specifics, it becomes clear that this innovation offers a robust solution for reliable pharma intermediate supplier networks seeking to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of m-aminoacetanilide has been plagued by inefficiencies inherent in traditional acylation protocols. As illustrated in the reaction scheme below, conventional methods typically involve the use of acetic acid or acetic anhydride in conjunction with hydrochloric acid at temperatures around 90°C. This reliance on strong mineral acids introduces severe corrosion risks to equipment and necessitates complex neutralization steps during workup. Furthermore, the generation of large volumes of acidic wastewater poses significant environmental challenges, increasing the burden on waste treatment facilities. The separation of the product often requires energy-intensive concentration and crystallization processes, which drive up utility costs and extend the overall production cycle time.
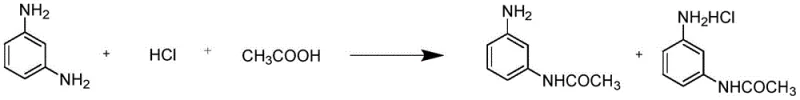
Beyond the immediate operational hazards, the traditional route suffers from difficulties in recycling the mother liquor effectively. The presence of salt by-products complicates the recovery of unreacted starting materials, leading to lower atom economy and increased raw material consumption. Previous attempts to optimize this process, such as those disclosed in CN101328133A, have struggled with issues related to large equipment volume requirements and prolonged reaction periods. These factors collectively contribute to a higher cost base and a less agile supply chain, making it difficult for manufacturers to respond quickly to market fluctuations. The need for amino protective agents in some variations further adds to the complexity and cost, creating a bottleneck for scalable production.
The Novel Approach
In stark contrast, the novel approach outlined in CN114181103A leverages the physical properties of m-phenylenediamine to create a self-sustaining reaction medium. By heating the diamine to its melting point and using it in significant excess, the process eliminates the need for external solvents entirely. This solvent-free strategy drastically reduces the volume of waste generated and simplifies the reactor setup. The reaction is conducted under a nitrogen atmosphere, which serves a dual purpose: it prevents the oxidation of the sensitive amine groups and inhibits thermal coking at elevated temperatures. This results in a cleaner reaction profile with fewer side products, directly translating to higher yields and easier purification.
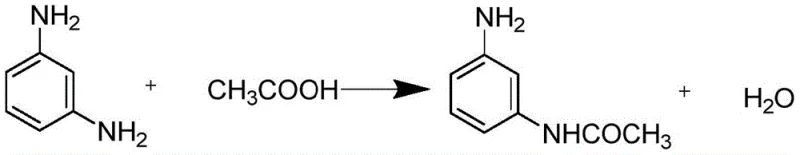
The workup procedure in this new method is elegantly simple yet highly effective. After the acylation is complete, water is added to the reaction mass to dissolve the excess m-phenylenediamine, while the desired product precipitates out due to its lower solubility in the aqueous medium at low temperatures. This crystallization step acts as a primary purification stage, yielding a filter cake of high-purity m-aminoacetanilide. Crucially, the filtrate containing the dissolved diamine is not discarded; instead, it is subjected to reduced pressure distillation to recover the raw material for reuse. This closed-loop system exemplifies the principles of green chemistry, offering substantial cost reduction in dye intermediate manufacturing by maximizing resource utilization.
Mechanistic Insights into Solvent-Free Acylation
The core of this technological breakthrough lies in the unique mechanistic pathway enabled by the melt-phase reaction conditions. At temperatures ranging from 120°C to 180°C, the kinetic energy of the molecules is sufficient to overcome the activation barrier for nucleophilic attack without the need for aggressive acid catalysts. The excess m-phenylenediamine acts as a proton shuttle, facilitating the transfer of protons during the formation of the amide bond. This autocatalytic behavior ensures a steady reaction rate while maintaining a homogeneous phase that promotes efficient heat and mass transfer. The absence of diluting solvents means that the concentration of reactive species is maximized, driving the equilibrium towards product formation according to Le Chatelier's principle.
Impurity control is another critical aspect where this mechanism excels. In traditional acidic environments, the aromatic ring is susceptible to electrophilic substitution side reactions, and the amine groups can undergo over-acylation or salt formation that is difficult to reverse. By avoiding hydrochloric acid, the new process minimizes the formation of chlorinated by-products and ammonium salts. The nitrogen blanket is essential here, as m-phenylenediamine is prone to oxidative degradation which can lead to colored impurities that are notoriously difficult to remove. The patent data indicates that maintaining strict inert conditions allows for the consistent production of material with HPLC purity exceeding 99%, meeting the stringent specifications required for high-purity OLED material and pharmaceutical applications.
How to Synthesize m-Aminoacetanilide Efficiently
Implementing this synthesis route requires precise control over thermal parameters and stoichiometry to achieve optimal results. The process begins with charging a four-neck flask with a substantial excess of m-phenylenediamine, typically in a mass ratio of 10:1 to 14:1 relative to acetic acid. Once the system is purged with nitrogen and the solid is melted, mechanical stirring is initiated to ensure uniformity. Acetic acid is then added dropwise while the temperature is maintained between 120°C and 180°C for a duration of 5 to 9 hours. Following the reaction, the mixture is cooled, and water is introduced to induce precipitation. For a comprehensive guide on the exact operational parameters and safety precautions, please refer to the standardized protocol below.
- Load excess m-phenylenediamine into a reactor, purge with nitrogen, and heat to melt completely.
- Heat the melt to 120-180°C and slowly dropwise add acetic acid while maintaining a nitrogen atmosphere.
- Cool the reaction mixture, add water to dissolve unreacted diamine, cool further to precipitate the product, and filter.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this melt-synthesis technology offers transformative benefits that extend far beyond the laboratory bench. The elimination of hydrochloric acid and organic solvents removes a major category of hazardous raw materials from the supply chain, thereby reducing regulatory compliance burdens and insurance costs associated with handling corrosive substances. The simplified workflow, which combines reaction and crystallization into a streamlined sequence, significantly shortens the batch cycle time. This agility allows manufacturers to respond more rapidly to demand spikes, ensuring a more reliable agrochemical intermediate supplier status for clients who depend on just-in-time delivery models.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the drastic reduction in raw material waste. Since the excess m-phenylenediamine is recovered and recycled with high efficiency, the net consumption of this expensive starting material is minimized. Furthermore, the removal of solvent purchase, storage, and disposal costs creates a leaner cost structure. The energy demand is also optimized; although the reaction runs at higher temperatures, the absence of solvent evaporation steps during workup and the simplified distillation of the filtrate result in lower overall utility consumption compared to traditional multi-step processes.
- Enhanced Supply Chain Reliability: Dependence on a complex web of solvent suppliers and acid vendors introduces vulnerability to market volatility. By simplifying the input requirements to essentially two main chemicals—m-phenylenediamine and acetic acid—the supply chain becomes more robust and easier to manage. The ability to recycle the primary raw material internally acts as a buffer against external supply shocks. This self-sufficiency ensures consistent production schedules and reduces the risk of delays caused by raw material shortages, providing a stable foundation for long-term contracts with downstream users.
- Scalability and Environmental Compliance: Scaling up chemical processes often amplifies safety and environmental risks, but this technology mitigates those concerns inherently. The solvent-free nature means there is no risk of solvent fires or explosions, and the reduced wastewater load simplifies effluent treatment. This aligns perfectly with increasingly strict global environmental regulations, future-proofing the manufacturing facility against tighter emission standards. The straightforward equipment requirements—essentially a heated reactor and a filtration unit—mean that capacity can be expanded with minimal capital expenditure, facilitating the commercial scale-up of complex polymer additives and intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this route for large-scale production.
Q: What are the primary advantages of the melt synthesis method over traditional acid-catalyzed routes?
A: The melt process eliminates the need for hazardous hydrochloric acid and complex organic solvents, significantly reducing waste generation and simplifying the post-reaction workup compared to traditional methods.
Q: How does this process ensure high purity levels suitable for pharmaceutical applications?
A: By utilizing nitrogen protection to prevent oxidation and coking, combined with a precise temperature-controlled precipitation step, the method consistently achieves HPLC purity exceeding 99%.
Q: Is the excess raw material recoverable in this synthesis route?
A: Yes, the unreacted m-phenylenediamine remaining in the filtrate after product precipitation can be efficiently recovered via reduced pressure distillation and recycled for subsequent batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable m-Aminoacetanilide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthesis technologies play in maintaining a competitive edge in the global chemical market. Our team of experts has thoroughly analyzed the potential of the melt-process described in CN114181103A and is prepared to leverage this knowledge for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards.
We invite you to collaborate with us to explore how this efficient synthesis route can benefit your specific application needs. Whether you require high-purity intermediates for pharmaceutical development or cost-effective solutions for dye manufacturing, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to see firsthand how we can drive value and efficiency in your supply chain.
