Advanced NHC-Catalyzed Synthesis of Boronic Acid Derivatives for Commercial Scale-up
The pharmaceutical landscape is continuously evolving with the demand for more efficient synthetic routes to complex bioactive molecules. Patent CN103408573A introduces a groundbreaking methodology for the preparation of diverse boronic acid derivatives, specifically targeting the structural motif represented by general formula (I). These compounds are not merely academic curiosities; they serve as pivotal intermediates in the synthesis of next-generation therapeutics for treating tumors and diabetes. The patent highlights the unique pseudo-peptide nature of aminoboronic acids, which allows them to act as potent transition-state analogues for serine hydrolases. By leveraging a novel catalytic system involving azacarbene ligands and alkali bases, this technology enables the direct transformation of alkyne compounds into valuable vinyl boronates. This represents a significant leap forward for any reliable pharmaceutical intermediate supplier seeking to optimize their portfolio with high-value building blocks.
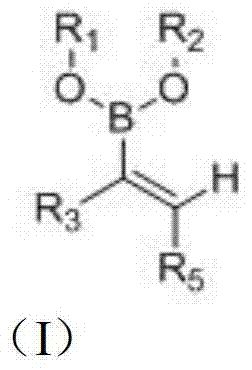
The strategic importance of this technology lies in its ability to access chemical space that was previously difficult or expensive to navigate. Traditional methods often struggled with regioselectivity and functional group tolerance, but the approach disclosed in CN103408573A demonstrates remarkable versatility. The resulting boronic acid derivatives can be further elaborated into complex drug candidates, including protease inhibitors and kinase modulators. For R&D teams focused on oncology and metabolic disorders, having access to a robust library of these intermediates is crucial for accelerating lead optimization campaigns. The patent explicitly mentions applications in preparing medicines for tumors and diabetes, aligning perfectly with current global health priorities and market demands for effective treatments in these therapeutic areas.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aminoboronic acids and related derivatives has been fraught with challenges that hindered their widespread adoption in process chemistry. The seminal work by Matteson, while foundational, relies heavily on the use of pinanediol diborane esters and chiral auxiliaries to induce stereochemistry. This approach typically necessitates multi-step sequences involving the preparation of chloroboronic esters followed by subsequent conversion, which inherently lowers the overall yield and increases the cost of goods. Furthermore, the side chains in Matteson's method are largely derived from alkylboronic acids, severely limiting the structural diversity accessible to medicinal chemists. Another notable method developed by Ellman utilizes chiral tert-butylsulfinamides and copper-NHC complexes; however, this protocol often requires stringent anhydrous conditions and glovebox techniques for catalyst preparation, posing significant operational hurdles for large-scale manufacturing. Additionally, Ellman's method has shown inconsistent results with aryl-substituted substrates, often yielding poor results or failing to produce the target product entirely, which restricts its utility in synthesizing aromatic drug scaffolds.
The Novel Approach
In stark contrast to these legacy technologies, the method disclosed in CN103408573A offers a streamlined, one-pot solution that directly constructs the target carbon-boron framework from readily available alkyne precursors. By employing a catalytic system composed of an azacarbene precursor and a base in a protic solvent, this novel approach eliminates the need for pre-functionalized boron reagents or sensitive chiral auxiliaries in the initial bond-forming step. The reaction proceeds efficiently at temperatures ranging from room temperature to reflux, accommodating a broad spectrum of substrates including aryl, alkyl, and heteroaryl alkynes. This shift from stoichiometric chiral reagents to a catalytic manifold not only simplifies the synthetic route but also drastically reduces the generation of chemical waste. The ability to tolerate various functional groups, such as protected amines and silyl groups, further underscores the robustness of this method, making it an ideal candidate for cost reduction in API manufacturing where step economy and raw material efficiency are paramount.
Mechanistic Insights into NHC-Copper Catalyzed Borylation
The core innovation of this technology rests on the unique electronic properties of N-heterocyclic carbene (NHC) ligands coordinated to copper centers. Unlike traditional phosphine ligands, NHCs act as strong sigma-donors with negligible pi-acceptor character, which stabilizes the copper catalyst and enhances its nucleophilicity towards the diboron reagent. In the catalytic cycle, the active copper-boryl species is generated in situ from the diboron reagent and the base. This species then undergoes regioselective insertion into the carbon-carbon triple bond of the alkyne substrate. The use of protic solvents, such as methanol or ethanol, plays a dual role: it serves as the reaction medium and potentially participates in the protonolysis step to release the final vinyl boronate product while regenerating the active catalyst. This mechanistic pathway avoids the formation of unstable intermediates that plague other methods, ensuring high fidelity in the product structure. The patent details a wide array of azacarbene precursors, ranging from simple imidazolium salts to more complex fused ring systems, allowing for fine-tuning of the steric and electronic environment around the metal center to optimize reactivity for specific substrate classes.
From an impurity control perspective, this catalytic system offers distinct advantages. The high selectivity of the NHC-copper complex minimizes side reactions such as homocoupling of the alkyne or over-borylation, which are common pitfalls in transition metal catalysis. The reaction conditions are mild enough to preserve sensitive functional groups like Boc-protected amines and ester moieties, which are frequently encountered in pharmaceutical intermediates. This chemoselectivity simplifies the downstream purification process, often allowing for isolation of the product via standard silica gel chromatography without the need for extensive recrystallization or preparative HPLC. For quality control teams, this translates to a cleaner crude profile and easier validation of the manufacturing process. The ability to achieve high yields, as demonstrated in the examples with values reaching up to 91%, indicates a highly efficient turnover number for the catalyst, reducing the residual metal load in the final product and easing the burden on metal scavenging steps.
How to Synthesize Vinyl Boronates Efficiently
The synthesis of these high-value boronic acid derivatives is designed to be operationally simple yet chemically robust, facilitating easy transfer from the laboratory to the pilot plant. The general procedure involves mixing the alkyne starting material with a slight excess of the diboron reagent in the presence of a catalytic amount of the azacarbene ligand precursor and a base. The choice of solvent is flexible, with methanol being a preferred option due to its ability to dissolve both organic substrates and inorganic bases while participating in the reaction mechanism. Reaction monitoring is straightforward using thin-layer chromatography (TLC), and the workup procedure involves standard aqueous extraction and drying techniques. Detailed standardized synthesis steps see the guide below.
- Combine alkyne compound, diboron reagent, azacarbene precursor (ligand), and base in a protic solvent such as methanol or ethanol.
- Stir the reaction mixture at room temperature to reflux for 24 to 48 hours under monitored conditions.
- Dilute with ethyl acetate, wash organic layers, dry over sodium sulfate, and purify via silica gel chromatography to isolate the target boronic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology presents a compelling value proposition centered on reliability and efficiency. The primary raw materials, specifically the alkyne compounds and diboron reagents like bis(pinacolato)diboron, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. This abundance ensures a stable supply chain and competitive pricing for the input materials. Furthermore, the elimination of exotic or highly sensitive reagents reduces the complexity of inventory management and storage requirements. The process does not require specialized equipment such as gloveboxes or cryogenic cooling systems, allowing it to be executed in standard stainless steel reactors found in most multipurpose chemical manufacturing facilities. This compatibility with existing infrastructure significantly lowers the barrier to entry for scaling up production.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by step reduction and improved yield. By consolidating what was traditionally a multi-step sequence into a single catalytic transformation, manufacturers can save substantially on labor, energy, and solvent consumption. The high atom economy of the borylation reaction ensures that a greater proportion of the starting mass ends up in the final product, reducing waste disposal costs. Additionally, the use of copper-based catalysts is generally more cost-effective compared to precious metal alternatives like palladium or platinum, further driving down the cost of goods sold. The simplified purification process also contributes to lower operational expenses by reducing the time and materials needed for isolation.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply chain resilience. Since the reaction tolerates protic solvents and does not demand strictly anhydrous environments, the risk of batch failure due to moisture ingress is minimized. This tolerance allows for more flexible scheduling and reduces the downtime associated with rigorous reactor drying protocols. The broad substrate scope means that a single manufacturing line can be adapted to produce a variety of different boronic acid derivatives simply by changing the alkyne feedstock, providing agility in responding to fluctuating market demands for different drug intermediates. This flexibility is crucial for maintaining continuity of supply in a dynamic pharmaceutical market.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is facilitated by the exothermic nature of the reaction being manageable and the absence of hazardous gas evolution. The use of alcohol solvents aligns with green chemistry principles, offering a safer and more environmentally friendly alternative to chlorinated or aromatic solvents often used in older methodologies. Waste streams are easier to treat, and the reduced use of stoichiometric chiral auxiliaries decreases the solid waste burden. For companies aiming to meet strict environmental, social, and governance (ESG) targets, adopting this catalytic borylation method demonstrates a commitment to sustainable manufacturing practices while maintaining high production throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this boronic acid synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this chemistry into their existing pipelines. The responses cover aspects ranging from catalyst stability to product purity specifications.
Q: What are the primary advantages of this NHC-catalyzed method over traditional Matteson synthesis?
A: Unlike the multi-step Matteson synthesis which relies on chiral auxiliaries and often suffers from low yields, this NHC-catalyzed method allows for direct construction of the carbon-boron bond from alkynes in a single pot, significantly simplifying the workflow and improving overall efficiency.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process utilizes commercially available diboron reagents and operates in common protic solvents like methanol without requiring extreme conditions or specialized glovebox environments for catalyst handling, making it highly amenable to commercial scale-up.
Q: What types of therapeutic applications do these boronic acid derivatives support?
A: These derivatives serve as critical intermediates for developing inhibitors against serine hydrolases, including DPP-4 inhibitors for type II diabetes and proteasome inhibitors like Bortezomib for multiple myeloma and other oncology indications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Boronic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN103408573A for the future of drug development. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless. We are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications for complex intermediates, guaranteeing that every batch meets the exacting standards required by global regulatory agencies. Our commitment to quality and consistency makes us a trusted partner for pharmaceutical companies looking to secure their supply of critical building blocks.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Whether you require custom synthesis of novel boronic acid analogues or scale-up of existing routes, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your path to market while optimizing your manufacturing economics.
