Advanced 5,5'-Linked Biphenyl Axial Chiral Ligands for High-Performance Asymmetric Synthesis
Introduction to Novel Axial Chiral Ligand Technology
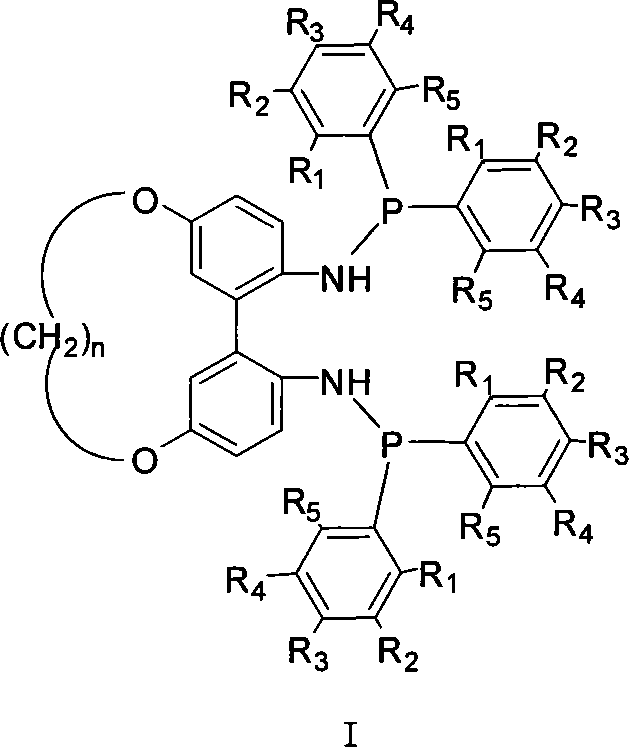
The landscape of asymmetric catalysis is continually evolving, driven by the demand for higher stereoselectivity and operational efficiency in the synthesis of complex organic molecules. Patent CN101168549A introduces a groundbreaking class of 1,1'-biphenyl axial chiral bisphosphonamidite ligands connected at the 5,5' positions, representing a significant departure from traditional binaphthyl-based systems. This innovation addresses the critical limitation of rigid dihedral angles found in conventional ligands like BINAP, offering a tunable structural framework that can be optimized for specific metal-catalyzed transformations. By incorporating a flexible alkylene bridge with varying chain lengths (where n ranges from 5 to 12), chemists can precisely modulate the spatial arrangement of the phosphorus centers relative to the biphenyl axis. This structural adaptability translates directly into enhanced catalytic performance, with reported reactivity reaching up to 100% and stereoselectivity exceeding 90% in model reactions. For R&D directors seeking reliable chiral ligand supplier partnerships, this technology offers a versatile platform for developing next-generation asymmetric processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of asymmetric hydrogenation and allylic alkylation has been dominated by C2-symmetric bisphosphine ligands derived from binaphthyl scaffolds, such as the ubiquitous BINAP. While these ligands have achieved industrial success, their structural rigidity imposes inherent constraints on the dihedral angle between the aromatic rings. Literature studies, including those on SYNPHOS, indicate that while ortho-substitution can stabilize axial chirality, it simultaneously restricts the rotational freedom necessary to achieve the optimal bite angle for certain substrates. This geometric inflexibility often results in suboptimal catalyst-substrate matching, leading to lower turnover frequencies or compromised enantiomeric excess in challenging transformations. Furthermore, the synthesis of highly substituted binaphthyl derivatives often requires harsh conditions and multiple protection-deprotection steps, complicating the supply chain for high-purity asymmetric catalyst precursors and increasing the overall cost of goods.
The Novel Approach
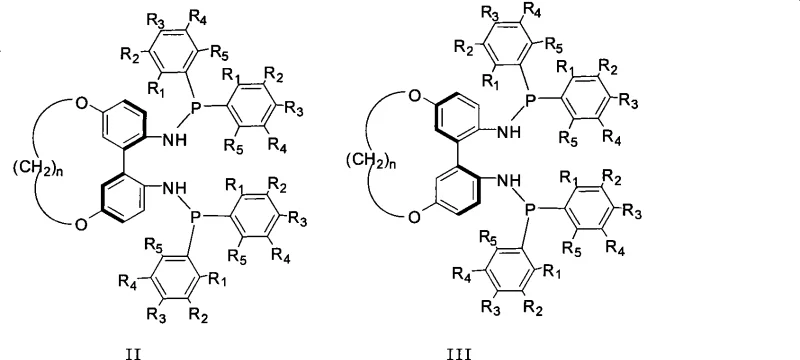
The novel approach detailed in the patent data circumvents these geometric bottlenecks by utilizing a 1,1'-biphenyl backbone linked at the 5,5' positions via a flexible aliphatic chain. This design strategy decouples the stabilization of axial chirality from the steric bulk at the ortho-positions, allowing the dihedral angle to be dynamically adjusted based on the length of the connecting chain (n=5 to 12). This flexibility enables the ligand to adapt its conformation during the catalytic cycle, accommodating diverse substrate geometries more effectively than rigid analogues. Additionally, the presence of phosphoramidite moieties introduces hemilabile characteristics and distinct electronic properties compared to pure phosphines, further expanding the scope of applicable reactions. For procurement managers focused on cost reduction in pharmaceutical intermediate manufacturing, this modular design allows for the fine-tuning of ligand properties without necessitating a complete overhaul of the synthetic route, thereby streamlining process development.
Mechanistic Insights into Tunable Dihedral Angle Catalysis
The mechanistic superiority of these 5,5'-linked ligands stems from their ability to create a chiral environment that is both robust and adaptable. In metal-catalyzed asymmetric reactions, the dihedral angle of the ligand backbone critically influences the geometry of the active metal complex, dictating the approach trajectory of the substrate. By varying the methylene chain length (n), the tension on the biphenyl axis is modulated, which in turn alters the energy barrier between different conformers. This tunability allows chemists to identify a 'sweet spot' where the steric repulsion between the ligand and the substrate maximizes facial selectivity. The phosphoramidite functionality, formed by the reaction of the diamine intermediate with aryl phosphine chlorides, provides a nitrogen donor that can participate in secondary coordination sphere interactions, potentially stabilizing transition states through hydrogen bonding or electrostatic effects. This dual control of steric and electronic factors is essential for achieving the high stereoselectivity (>90% ee) reported in the patent data.
Furthermore, the impurity profile of the final catalyst system is heavily influenced by the stability of the axial chirality during the reaction conditions. Traditional ligands may suffer from racemization at elevated temperatures if the rotational barrier is insufficient. However, the 5,5'-linkage in this novel series creates a macrocyclic constraint that significantly raises the barrier to rotation, ensuring configurational stability even under rigorous reaction conditions such as refluxing dichloromethane or elevated hydrogenation pressures. The synthetic route ensures that the chiral information is locked in place prior to metal coordination, minimizing the formation of meso-compounds or diastereomeric impurities that could degrade product quality. For supply chain heads concerned with reducing lead time for high-purity chiral ligands, this inherent stability simplifies storage and handling requirements, reducing the risk of degradation during transit and long-term inventory holding.
How to Synthesize 5,5'-Linked Biphenyl Ligands Efficiently
The synthesis of these advanced ligands follows a logical and scalable sequence that begins with readily available commodity chemicals. The process initiates with an Ullmann coupling reaction to construct the biphenyl core, followed by functional group manipulations to install the bridging chain and the phosphorus centers. Each step has been optimized to maximize yield and minimize waste, reflecting a design philosophy oriented towards industrial viability. The detailed standardized synthesis steps are outlined in the guide below, providing a clear roadmap for laboratory scale-up and process validation.
- Perform Ullmann coupling of 3-bromo-4-nitroanisole with activated copper powder at 170°C to form the biphenyl backbone, followed by demethylation using lithium chloride in DMF.
- React the resulting diphenol with dihaloalkanes (X(CH2)nX') to create the flexible bridge, then reduce the nitro groups to amines using Pd/C catalyzed hydrogenation.
- Condense the diamine intermediate with aryl phosphine chlorides in the presence of triethylamine, followed by chiral HPLC resolution to isolate optically pure (R) and (S) enantiomers.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this ligand technology offers substantial strategic advantages for organizations managing complex chemical supply chains. The reliance on commodity starting materials such as 3-bromo-4-nitroanisole eliminates the dependency on scarce or geopolitically sensitive precursors often associated with specialized chiral pool chemicals. This foundational accessibility ensures a stable supply base, mitigating risks related to raw material shortages that can disrupt production schedules. Moreover, the synthetic pathway avoids the use of extremely low-temperature cryogenic conditions or ultra-high vacuum systems, allowing the process to be executed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. This compatibility with existing infrastructure significantly lowers the capital expenditure required for technology transfer and commercialization.
- Cost Reduction in Manufacturing: The synthetic route leverages high-yielding transformations, such as the quantitative reduction of nitro groups to amines using palladium on carbon, which minimizes material loss and solvent consumption. By eliminating the need for expensive transition metal catalysts in the ligand synthesis itself (using copper powder instead), the direct material costs are significantly reduced. Furthermore, the ability to tune the ligand performance through simple chain length variation reduces the R&D iteration cycles required to find the optimal catalyst for a specific API, accelerating time-to-market and lowering overall development expenditures.
- Enhanced Supply Chain Reliability: The robustness of the Ullmann coupling and subsequent alkylation steps ensures consistent batch-to-batch reproducibility, a critical factor for regulatory compliance in pharmaceutical manufacturing. The use of standard reagents like triethylamine and dichloromethane facilitates straightforward sourcing from multiple global vendors, preventing single-source bottlenecks. Additionally, the final purification via chiral HPLC, while sophisticated, is a well-established unit operation in the fine chemical industry, ensuring that high-purity specifications can be met reliably without requiring novel or unproven separation technologies.
- Scalability and Environmental Compliance: The process generates manageable waste streams, primarily consisting of inorganic salts and common organic solvents that can be treated using standard effluent processing facilities. The avoidance of heavy metal contaminants in the ligand structure itself simplifies the downstream purification of the final drug substance, reducing the burden on metal scavenging resins. The high atom economy of the key coupling steps contributes to a greener manufacturing profile, aligning with increasingly stringent environmental regulations and corporate sustainability goals regarding waste minimization and energy efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ligand technology in industrial settings. These answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is crucial for evaluating the feasibility of integrating this chemistry into existing production workflows.
Q: What is the primary advantage of the 5,5'-linked biphenyl structure over traditional BINAP ligands?
A: Unlike the rigid binaphthyl structure of BINAP which limits dihedral angle adjustments, the 5,5'-linked biphenyl scaffold with a flexible alkyl chain (n=5-12) allows for tunable dihedral angles. This flexibility enables the optimization of steric hindrance and electronic properties, leading to significantly improved reactivity and stereoselectivity in asymmetric catalytic reactions.
Q: How does the synthesis route ensure high purity for pharmaceutical applications?
A: The synthesis employs robust purification techniques at critical stages, including recrystallization of the Ullmann coupling product and column chromatography for the alkylation step. The final optical purity is guaranteed through chiral high-performance liquid chromatography (HPLC) resolution, ensuring the isolation of distinct (R) and (S) enantiomers with high ee values suitable for sensitive drug synthesis.
Q: Can these ligands be scaled for industrial production?
A: Yes, the synthetic pathway utilizes commercially available starting materials like 3-bromo-4-nitroanisole and standard reagents such as copper powder and palladium on carbon. The reaction conditions, including temperatures up to 170°C and atmospheric pressure hydrogenation, are amenable to large-scale reactor operations, facilitating the commercial scale-up of complex fine chemicals without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bisphosphonamidite Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that advanced chiral ligands play in the efficient synthesis of high-value pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the optical purity and chemical integrity of every batch. Our commitment to quality assurance means that clients can rely on us for consistent supply of complex fine chemicals that meet the exacting standards of the global pharmaceutical industry.
We invite potential partners to engage with our technical procurement team to discuss how this novel ligand technology can be tailored to your specific synthetic challenges. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this flexible ligand platform for your asymmetric catalysis needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive innovation and profitability in your manufacturing operations.
