Advanced Synthesis of Ilaprazole Key Intermediate via Thiourea Dioxide Reduction
Advanced Synthesis of Ilaprazole Key Intermediate via Thiourea Dioxide Reduction
The pharmaceutical industry continuously seeks robust synthetic routes for proton pump inhibitors (PPIs), with ilaprazole representing a significant advancement in gastric acid suppression therapy. A critical bottleneck in the supply chain for this active pharmaceutical ingredient (API) has historically been the efficient production of its key precursor, 5-(1H-pyrrole-1-yl)-2-mercaptobenzimidazole. Recent intellectual property developments, specifically patent CN113354623A, disclose a transformative preparation method that addresses long-standing issues regarding yield, purity, and environmental impact. This technical insight report analyzes the novel three-step synthesis pathway detailed in the patent, highlighting its potential to redefine the manufacturing landscape for this high-value pharmaceutical intermediate. By shifting away from hazardous metal catalysts towards a thiourea dioxide-based reduction system, the technology offers a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to optimize their production capabilities.
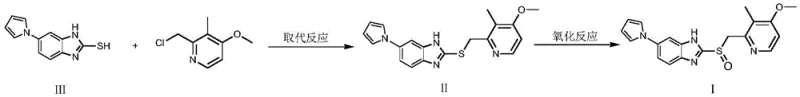
The strategic importance of this intermediate cannot be overstated, as it serves as the foundational scaffold for the final drug substance. As illustrated in the reaction scheme, the intermediate undergoes subsequent substitution and oxidation reactions to yield the final ilaprazole structure. Ensuring the quality and availability of this precursor is paramount for maintaining the continuity of ilaprazole manufacturing. The disclosed method not only streamlines the synthesis of the mercaptobenzimidazole core but also ensures that the resulting material possesses the requisite physicochemical properties to support downstream processing without extensive purification burdens. This alignment with Good Manufacturing Practice (GMP) principles makes the technology highly attractive for integration into existing commercial supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing 5-(1H-pyrrole-1-yl)-2-mercaptobenzimidazole have been plagued by significant technical and economic inefficiencies that hinder large-scale adoption. For instance, earlier Korean patents described a route utilizing titanium tetrachloride and succinaldehyde, which suffered from abysmally low yields of approximately 21 percent and generated substantial hazardous waste due to the corrosive nature of the titanium reagent. Furthermore, alternative Chinese patents introduced methods relying on stannous chloride or palladium-carbon for the critical reduction step. While these methods achieved moderate conversion, they introduced severe complications regarding post-reaction processing, such as the difficult removal of metal sludge and the risk of heavy metal contamination in the final product. The presence of reddish-brown solids and the need for complex recrystallization procedures further exacerbated the cost of goods sold, rendering these traditional approaches suboptimal for modern, high-volume API manufacturing.
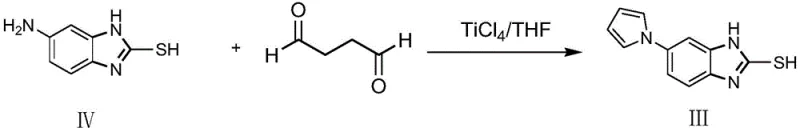
The Novel Approach
In stark contrast to these legacy processes, the methodology outlined in patent CN113354623A introduces a streamlined, three-step sequence that prioritizes operational simplicity and environmental sustainability. The innovation lies primarily in the selection of thiourea dioxide as the reducing agent for the nitro group, coupled with a carefully optimized solvent system comprising ethanol and water. This approach effectively circumvents the need for expensive transition metal catalysts, thereby eliminating the associated filtration bottlenecks and heavy metal residue risks. Additionally, the initial pyrrole ring formation utilizes a homogeneous solvent system of tetrahydrofuran and water with acetic acid buffering, which significantly accelerates reaction kinetics compared to the biphasic systems of the past. The result is a robust process capable of delivering high-purity material with a stable overall yield, directly addressing the pain points of cost reduction in API manufacturing.
Mechanistic Insights into Thiourea Dioxide-Mediated Reduction and Cyclization
The core chemical innovation of this process revolves around the efficient transformation of the nitro-aniline derivative into the corresponding diamine without compromising the sensitive pyrrole moiety. The mechanism involves the nucleophilic attack of the thiourea dioxide species on the nitro group under alkaline conditions provided by sodium hydroxide. This redox reaction proceeds smoothly at moderate temperatures between 50°C and 60°C, ensuring that the thermal stress on the molecule is minimized. Unlike catalytic hydrogenation which requires high-pressure equipment and pyrophoric catalysts, this chemical reduction operates under atmospheric pressure with simple stirring. The stoichiometry is carefully balanced, typically employing a molar ratio of thiourea dioxide to sodium hydroxide ranging from 1:1 to 1:8, with optimal results observed at a 1:2 ratio. This precise control prevents over-reduction or side reactions that could lead to polymeric impurities, ensuring a clean reaction profile that simplifies downstream isolation.
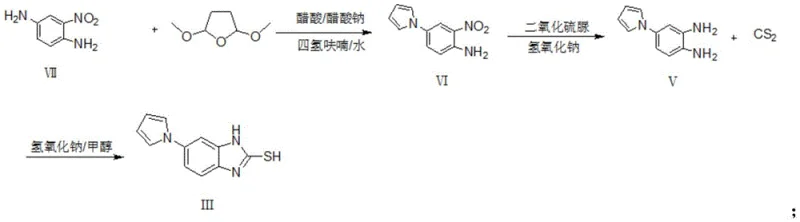
Following the reduction, the final cyclization step leverages the high nucleophilicity of the newly formed ortho-diamine to attack carbon disulfide (CS2) in the presence of a strong base. This step constructs the benzimidazole-2-thione ring system with high fidelity. The use of methanol as the solvent in this stage facilitates the dissolution of both the organic diamine and the inorganic base, creating a pseudo-homogeneous environment that drives the reaction to completion within 2 hours at 50°C. The subsequent cooling to 0-10°C induces crystallization of the product directly from the reaction mixture, a phenomenon known as reactive crystallization. This eliminates the need for energy-intensive solvent swaps or chromatographic purification, allowing the product to be isolated via simple filtration. The mechanistic elegance of this route ensures that impurity levels remain exceptionally low, with single impurities controlled below 0.10 percent.
How to Synthesize 5-(1H-pyrrole-1-yl)-2-mercaptobenzimidazole Efficiently
Implementing this synthesis protocol requires strict adherence to the specified reaction parameters to maximize yield and purity. The process begins with the condensation of 2-nitro-1,4-phenylenediamine with 2,5-dimethoxytetrahydrofuran, followed by the critical thiourea dioxide reduction and final cyclization with carbon disulfide. Each step has been optimized to balance reaction rate with product stability, ensuring that the process is amenable to scale-up from laboratory to pilot plant. Operators must pay particular attention to temperature control during the exothermic reduction phase and the precise addition of bases to maintain the correct pH profile. Detailed standard operating procedures regarding reagent grades, solvent drying, and workup techniques are essential for reproducibility. For a comprehensive breakdown of the standardized synthesis steps, please refer to the technical guide below.
- Condense 2-nitro-1,4-phenylenediamine with 2,5-dimethoxytetrahydrofuran in THF/water with acetic acid/sodium acetate to form 2-nitro-4-(1H-pyrrole-1-yl)aniline.
- Reduce the nitro group of the intermediate using thiourea dioxide and sodium hydroxide in ethanol/water at 50-60°C to obtain 4-(1H-pyrrole-1-yl)benzene-1,2-diamine.
- Cyclize the diamine with carbon disulfide (CS2) and a base like sodium hydroxide in methanol at 50°C to yield the final mercaptobenzimidazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthetic route offers profound strategic advantages that extend beyond mere technical feasibility. The elimination of precious metal catalysts such as palladium and hazardous reagents like titanium tetrachloride fundamentally alters the cost structure of the intermediate. By removing the dependency on volatile commodity markets for these specialized reagents, manufacturers can achieve significant stabilization in their raw material costs. Furthermore, the simplified workup procedure, which avoids complex filtration of metal sludges and extensive recrystallization cycles, drastically reduces the consumption of auxiliary solvents and processing time. This operational efficiency translates directly into improved throughput and reduced utility consumption, making the process highly competitive in a price-sensitive generic pharmaceutical market.
- Cost Reduction in Manufacturing: The substitution of expensive catalytic systems with inexpensive chemical reductants like thiourea dioxide creates a substantial downward pressure on the variable cost of production. Since thiourea dioxide is a commodity chemical with a stable supply chain, the risk of price spikes is minimized compared to noble metals. Additionally, the high yield of the final step (reported up to 96 percent in optimized examples) ensures that raw material utilization is maximized, reducing the effective cost per kilogram of the active intermediate. The avoidance of chromatographic purification further lowers the cost of goods by minimizing silica gel or resin usage and reducing solvent recovery loads.
- Enhanced Supply Chain Reliability: The robustness of this chemical process enhances supply chain resilience by reducing the likelihood of batch failures due to catalyst poisoning or sensitivity. Traditional methods often suffered from inconsistent yields due to the variability in metal catalyst activity; this new chemical reduction is far more forgiving and reproducible. The use of common solvents like ethanol, methanol, and tetrahydrofuran ensures that sourcing logistics are straightforward, as these materials are readily available globally. This reliability allows supply chain managers to forecast production timelines with greater accuracy, reducing the need for excessive safety stock and improving working capital efficiency.
- Scalability and Environmental Compliance: Scalability is inherently built into this design through the use of homogeneous reaction conditions and the avoidance of heterogeneous catalysis which often presents mass transfer limitations at large scales. From an environmental standpoint, the process generates significantly less hazardous waste, particularly heavy metal contaminated sludge, which is costly to dispose of. The aqueous workup and the ability to crystallize the product directly reduce the volume of organic waste streams. This aligns with increasingly stringent environmental regulations and corporate sustainability goals, facilitating smoother regulatory approvals and reducing the carbon footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is critical for R&D teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of the supply source. The clarity provided here aims to mitigate risks associated with adopting new chemical processes in a regulated environment.
Q: Why is thiourea dioxide preferred over traditional reducing agents for this synthesis?
A: Thiourea dioxide eliminates the need for expensive and hazardous transition metal catalysts like palladium-carbon or stannous chloride, thereby preventing heavy metal residues in the final API and simplifying wastewater treatment.
Q: What is the expected purity of the intermediate produced by this method?
A: The patented process consistently achieves a product purity of 99.8% with single impurity control below 0.10%, which is critical for meeting pharmacopeia standards for ilaprazole.
Q: How does this route improve scalability compared to previous methods?
A: By avoiding difficult filtration steps associated with metal sludge and utilizing a homogeneous reaction system in the first step, the process significantly reduces operational complexity and enhances batch-to-batch consistency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-(1H-pyrrole-1-yl)-2-mercaptobenzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We understand the critical nature of stringent purity specifications for ilaprazole precursors and have invested heavily in rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch. Our capability to implement the thiourea dioxide reduction route positions us as a strategic partner capable of delivering high-purity intermediates that meet global pharmacopeia standards.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements. By leveraging our manufacturing capabilities, you can secure a stable supply of this critical building block while optimizing your overall cost structure. We encourage you to request a Customized Cost-Saving Analysis to quantify the potential economic benefits for your specific application. Furthermore, our team is ready to provide specific COA data and route feasibility assessments to support your regulatory filings and ensure a smooth integration into your supply chain.
