Advanced Catalyst-Free Synthesis of Quinoline-2-Thiocarboxamides for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking more efficient and environmentally benign pathways for constructing complex heterocyclic scaffolds. Patent CN111704575A introduces a groundbreaking methodology for the synthesis of quinoline-2-thiocarboxamide compounds, a class of molecules with significant potential in medicinal chemistry and crop protection. This technology distinguishes itself by operating under strictly catalyst-free and additive-free conditions, addressing a critical pain point in traditional organic synthesis regarding metal contamination. By utilizing readily available starting materials such as 2-methylquinoline and various amine derivatives, this process offers a streamlined route that aligns perfectly with the principles of green chemistry. For R&D directors and procurement managers alike, this represents a shift towards more sustainable and cost-effective manufacturing protocols that do not compromise on yield or purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of thioamide derivatives has heavily relied on the use of transition metal catalysts or specific additives to drive the reaction forward efficiently. These conventional methods often introduce significant complications downstream, particularly concerning the removal of toxic metal residues from the final active pharmaceutical ingredients. The presence of such impurities can necessitate expensive and time-consuming purification steps, such as specialized scavenging resins or repeated recrystallization processes, which drastically increase the overall production cost. Furthermore, the disposal of waste streams containing heavy metals poses severe environmental compliance challenges for chemical manufacturers aiming to meet increasingly stringent global regulations. The reliance on sensitive catalysts also limits the substrate scope, often failing to tolerate diverse functional groups present in complex drug intermediates. Consequently, the industry has long suffered from processes that are not only economically inefficient but also environmentally burdensome, creating a pressing need for innovation in this chemical space.
The Novel Approach
In stark contrast to legacy methods, the technology disclosed in patent CN111704575A eliminates the need for any external catalyst or additive, relying instead on the intrinsic reactivity of the substrates under thermal conditions. This novel approach utilizes elemental sulfur as a sustainable sulfur source, reacting directly with 2-methylquinoline and amines in a polar aprotic solvent like DMSO. The simplicity of this system means that the reaction mixture is far less complex, significantly reducing the burden on downstream purification teams. By avoiding metal catalysts entirely, the risk of heavy metal contamination in the final product is effectively nullified, ensuring higher purity profiles right from the crude stage. This method demonstrates excellent substrate adaptability, accommodating both aromatic and aliphatic amines with varying electronic properties. For a reliable pharmaceutical intermediates supplier, adopting this catalyst-free protocol translates directly into cost reduction in fine chemical manufacturing by simplifying the operational workflow and waste management strategies.
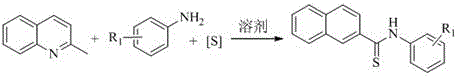
Mechanistic Insights into Catalyst-Free Thioamidation
The mechanistic pathway of this transformation is fascinating as it proceeds without the activation typically provided by metal centers. It is hypothesized that the reaction initiates through the thermal activation of the methyl group on the quinoline ring, facilitating a nucleophilic attack by the amine species in the presence of elemental sulfur. The high temperature range of 130-150°C provides sufficient energy to overcome the activation barrier, allowing for the formation of the carbon-sulfur and carbon-nitrogen bonds in a concerted or stepwise manner. The use of DMSO as a solvent plays a crucial role not just as a medium but potentially as a mild oxidant or stabilizer for the intermediate species formed during the reaction. This metal-free mechanism ensures that the electronic nature of the substituents on the amine ring does not interfere with catalyst coordination, thereby broadening the scope of compatible substrates. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters for commercial scale-up of complex heterocycles without the unpredictability associated with catalyst deactivation.
Impurity control is inherently superior in this system due to the absence of metal-based side reactions that often generate difficult-to-remove byproducts. In traditional catalytic cycles, ligand degradation or metal aggregation can lead to colored impurities or trace metals that persist through standard workups. Here, the primary byproducts are likely limited to unreacted starting materials or simple sulfur-containing species that are easily separated during the aqueous workup and column chromatography steps. The patent data indicates high yields across a range of substrates, suggesting that the reaction is robust and selective for the desired thioamide bond formation. This high level of selectivity minimizes the formation of isomeric impurities, which is a critical quality attribute for high-purity quinoline-2-thiocarboxamide used in sensitive biological applications. For supply chain heads, this predictability in impurity profiles means reducing lead time for high-purity intermediates as fewer analytical iterations are required to release batches.
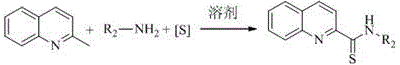
How to Synthesize Quinoline-2-Thiocarboxamide Efficiently
Implementing this synthesis route in a production environment requires careful attention to the stoichiometry and thermal parameters outlined in the patent documentation. The process begins with the precise charging of 2-methylquinoline, the selected amine substrate, and elemental sulfur into a dry reaction vessel under an inert atmosphere. The molar ratio of the amine to the quinoline derivative is typically maintained between 1.2:1 to 3:1, with a preference for a 2:1 ratio to drive the reaction to completion. Elemental sulfur is added in excess, usually at a ratio of 2-4:1 relative to the amine, to ensure sufficient sulfur availability for the thioamidation process. The reaction mixture is then heated to a temperature range of 130-150°C and stirred for a duration of 9 to 15 hours, depending on the specific reactivity of the amine substrate. Following the reaction, the mixture is cooled to room temperature and subjected to a standard extraction protocol using ethyl acetate, followed by drying and purification. The detailed standardized synthesis steps see the guide below.
- Charge 2-methylquinoline, aromatic or aliphatic amine, elemental sulfur, and DMSO solvent into a reaction vessel.
- Stir the mixture at 130-150°C for 9-15 hours under nitrogen atmosphere.
- Cool to room temperature, extract with ethyl acetate, dry, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this catalyst-free technology offers substantial strategic advantages for procurement and supply chain management teams looking to optimize their vendor partnerships. The elimination of expensive transition metal catalysts removes a significant variable cost from the bill of materials, directly contributing to overall cost reduction in manufacturing operations. Furthermore, the simplified workup procedure reduces the consumption of solvents and purification media, enhancing the environmental sustainability profile of the production process. This efficiency gain allows suppliers to offer more competitive pricing structures while maintaining healthy margins, a critical factor for long-term supply agreements. The robustness of the reaction conditions also ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by failed runs or out-of-specification results. For supply chain heads, this reliability translates into enhanced supply chain reliability, ensuring that critical intermediates are available when needed for downstream API synthesis without unexpected interruptions.
- Cost Reduction in Manufacturing: The removal of metal catalysts eliminates the need for costly scavenging steps and specialized waste disposal protocols associated with heavy metals. This simplification of the downstream processing significantly lowers the operational expenditure per kilogram of product produced. Additionally, the use of elemental sulfur as a reagent is economically favorable compared to specialized thioating agents, further driving down raw material costs. The high yields reported in the patent examples suggest that material throughput is maximized, reducing the amount of starting material wasted in side reactions. These factors combine to create a highly cost-effective manufacturing process that can withstand market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The simplicity of the reaction setup means that it can be easily scaled from laboratory to pilot and commercial plants without requiring specialized equipment for catalyst handling. The readily available nature of the starting materials, such as 2-methylquinoline and common amines, ensures that supply chains are not vulnerable to shortages of exotic reagents. This accessibility of raw materials guarantees a steady flow of production, reducing the risk of bottlenecks that often plague complex synthetic routes. Moreover, the robust nature of the reaction conditions allows for flexibility in manufacturing scheduling, accommodating urgent orders without compromising on quality or safety standards.
- Scalability and Environmental Compliance: Operating without metal catalysts aligns perfectly with modern environmental regulations that strictly limit heavy metal discharge in industrial effluents. This compliance reduces the regulatory burden on manufacturing sites and minimizes the risk of fines or shutdowns due to environmental violations. The process generates less hazardous waste, simplifying the waste management logistics and reducing the associated disposal costs. Scalability is further enhanced by the fact that the reaction does not rely on sensitive catalysts that might degrade or lose activity upon scale-up, ensuring that laboratory results translate accurately to commercial production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalyst-free synthesis technology. These answers are derived directly from the technical specifications and beneficial effects described in the patent literature to provide clarity for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios. The information provided here serves as a foundational guide for technical discussions between R&D teams and supply chain stakeholders.
Q: Does this synthesis method require metal catalysts?
A: No, the method described in patent CN111704575A operates under catalyst-free and additive-free conditions, eliminating metal residue risks.
Q: What is the preferred sulfur source for this reaction?
A: Elemental sulfur is used as the sulfur source, typically in a molar ratio of 2-4:1 relative to the amine substrate.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the simple reaction conditions and easy workup procedure make it highly suitable for commercial scale-up of complex heterocycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline-2-Thiocarboxamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this catalyst-free synthesis route for the production of high-value heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which guarantee that every batch of quinoline-2-thiocarboxamide meets the highest international standards. We are equipped to handle the specific nuances of this sulfur-mediated chemistry, providing a secure and compliant manufacturing environment for your critical projects. Partnering with us means gaining access to a team that values both technical excellence and commercial reliability.
We invite you to engage with our technical procurement team to discuss how this technology can optimize your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this catalyst-free method for your projects. We encourage you to reach out for specific COA data and route feasibility assessments to validate the performance of this synthesis in your context. Let us collaborate to drive efficiency and innovation in your chemical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
