Advancing Isotope Labeling: A Mercury-Free Route for High-Purity Heavy Oxygen Labeled Amides
The landscape of isotopic labeling in organic synthesis is undergoing a significant transformation, driven by the urgent need for greener, safer, and more efficient methodologies. Patent CN108383747B, published in early 2021, introduces a groundbreaking preparation method for heavy oxygen-labeled amide compounds that fundamentally shifts the paradigm away from toxic heavy metal catalysis. This technology addresses a critical bottleneck in the production of stable isotope-labeled compounds, which are indispensable tools for elucidating organic reaction mechanisms and advancing proteomics research via mass spectrometry. By utilizing thioamides as substrates and activating them with N-halosuccinimide type oxidants in the presence of heavy oxygen water, this invention achieves high-yield conversion to 18O-labeled amides without the environmental and safety liabilities associated with prior art. For R&D directors and procurement strategists in the fine chemical sector, this represents a pivotal opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials through a sustainable, scalable process that aligns with modern regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the activation of thioamides for nucleophilic substitution has relied heavily on the use of potent Lewis acids and heavy metal catalysts, creating substantial operational hazards and waste management challenges. As illustrated in the traditional synthetic pathway, the use of inorganic mercury (II) salts, such as Hg(OTf)2, was the standard for facilitating the exchange of sulfur for oxygen in the presence of heavy water. 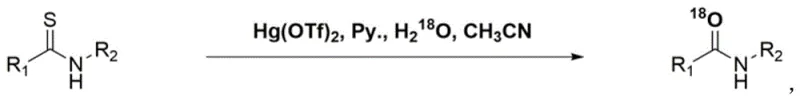 This conventional approach suffers from severe drawbacks, primarily the extreme toxicity of mercury reagents which necessitates rigorous containment protocols and expensive disposal procedures for heavy metal waste. Furthermore, the reaction generates trifluoromethanesulfonic acid as a byproduct, a superacid known for its corrosive nature that can degrade reactor linings and compromise equipment integrity over time. The formation of large amounts of precipitates and heavy metal sludge complicates the downstream processing, requiring extensive filtration and purification steps that lower overall throughput and increase the cost of goods sold. These factors collectively render the traditional mercury-based route unsuitable for modern, large-scale commercial manufacturing where environmental compliance and operator safety are paramount concerns.
This conventional approach suffers from severe drawbacks, primarily the extreme toxicity of mercury reagents which necessitates rigorous containment protocols and expensive disposal procedures for heavy metal waste. Furthermore, the reaction generates trifluoromethanesulfonic acid as a byproduct, a superacid known for its corrosive nature that can degrade reactor linings and compromise equipment integrity over time. The formation of large amounts of precipitates and heavy metal sludge complicates the downstream processing, requiring extensive filtration and purification steps that lower overall throughput and increase the cost of goods sold. These factors collectively render the traditional mercury-based route unsuitable for modern, large-scale commercial manufacturing where environmental compliance and operator safety are paramount concerns.
The Novel Approach
In stark contrast to the hazardous legacy methods, the novel methodology disclosed in the patent employs a mild, metal-free oxidation system that dramatically simplifies the synthetic workflow while maintaining exceptional efficiency. The core innovation lies in the use of N-halosuccinimide oxidants, such as N-bromosuccinimide (NBS) or N-chlorosuccinimide (NCS), to activate the thioamide functionality in situ.  This activation allows heavy oxygen water (H2-18O) to act as a potent nucleophile, directly attacking the activated carbon center to form the desired 18O-labeled amide bond with high fidelity. The reaction proceeds smoothly at room temperature in common anhydrous solvents like acetonitrile or ethyl acetate, eliminating the need for cryogenic conditions or high-energy inputs. By replacing toxic mercury salts with benign organic oxidants and mild bases like triethylamine or pyridine, the process generates a much cleaner reaction profile with minimal inorganic waste. This shift not only enhances the safety profile of the manufacturing facility but also streamlines the purification process, as the absence of heavy metals removes the need for complex scavenging resins or specialized extraction protocols, thereby accelerating the time-to-market for critical research intermediates.
This activation allows heavy oxygen water (H2-18O) to act as a potent nucleophile, directly attacking the activated carbon center to form the desired 18O-labeled amide bond with high fidelity. The reaction proceeds smoothly at room temperature in common anhydrous solvents like acetonitrile or ethyl acetate, eliminating the need for cryogenic conditions or high-energy inputs. By replacing toxic mercury salts with benign organic oxidants and mild bases like triethylamine or pyridine, the process generates a much cleaner reaction profile with minimal inorganic waste. This shift not only enhances the safety profile of the manufacturing facility but also streamlines the purification process, as the absence of heavy metals removes the need for complex scavenging resins or specialized extraction protocols, thereby accelerating the time-to-market for critical research intermediates.
Mechanistic Insights into N-Halosuccinimide Mediated Thioamide Oxidation
The mechanistic elegance of this transformation relies on the electrophilic activation of the sulfur atom within the thioamide group by the N-halosuccinimide oxidant. Upon addition of the oxidant to the reaction mixture containing the thioamide and base, the sulfur atom is likely halogenated or otherwise activated to form a highly electrophilic intermediate species. This activation significantly increases the susceptibility of the carbonyl-like carbon to nucleophilic attack, lowering the energy barrier for the subsequent substitution step. The heavy oxygen water, acting as the nucleophile, then attacks this activated center, leading to the displacement of the sulfur moiety and the formation of the C-18O bond. The presence of a base, such as triethylamine or diisopropylethylamine, plays a crucial role in neutralizing the acidic byproducts generated during the oxidation, maintaining the reaction pH within an optimal range that prevents hydrolysis of sensitive functional groups elsewhere in the molecule. This delicate balance of reactivity ensures that the isotopic label is incorporated specifically at the amide position without scrambling or loss of stereochemical integrity, which is particularly vital when dealing with chiral amino acid derivatives or complex peptide backbones.
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed pathways. The mild, near-neutral conditions prevent the racemization of chiral centers adjacent to the amide bond, a common pitfall in harsh acidic environments. Furthermore, the byproducts of the reaction, primarily succinimide and the corresponding amine salt, are highly soluble in polar organic solvents and can be easily removed during the aqueous workup or silica gel chromatography. The patent data highlights that monitoring the reaction via thin-layer chromatography (TLC) is straightforward, as the polarity difference between the starting thioamide and the product amide is significant, allowing for precise endpoint determination. This controllability minimizes the formation of over-oxidized side products or degradation impurities, ensuring that the final isolated material meets the stringent purity specifications required for biological applications. The high labeling efficiency reported, exceeding 95% in model systems, confirms that the mechanistic pathway is highly selective for the desired oxygen exchange, maximizing the value of the expensive heavy oxygen water reagent.
How to Synthesize Heavy Oxygen Labeled Amide Efficiently
The practical implementation of this synthesis route is designed for robustness and ease of execution, making it accessible for both laboratory-scale discovery and pilot-scale production. The protocol involves a sequential addition of reagents into a nitrogen-protected vessel to exclude moisture and oxygen, ensuring the integrity of the anhydrous solvent and the heavy oxygen water. Typically, the thioamide substrate is dissolved in a solvent such as anhydrous acetonitrile along with the base and the isotopically enriched water, followed by the controlled addition of the solid oxidant.
- Prepare the reaction mixture by adding thioamide, heavy oxygen water (H2-18O), a base such as triethylamine or pyridine, and an anhydrous solvent like acetonitrile into a nitrogen-protected vessel.
- Initiate the oxidation by adding an N-halosuccinimide oxidant (e.g., N-bromosuccinimide or N-chlorosuccinimide) to the stirred mixture at room temperature.
- Monitor the reaction progress via TLC until the thioamide starting material is consumed, then filter, concentrate, and purify the resulting 18O-labeled amide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this mercury-free technology translates into tangible strategic benefits that extend far beyond simple chemical yield. The elimination of highly regulated toxic substances like mercury salts fundamentally alters the cost structure of the manufacturing process by removing the need for specialized hazardous waste disposal contracts and expensive effluent treatment systems. This reduction in environmental compliance overhead allows for a more streamlined operation where resources can be redirected towards capacity expansion and quality assurance rather than waste management. Additionally, the use of commodity chemicals such as N-bromosuccinimide and triethylamine ensures a stable and resilient supply chain, as these reagents are produced globally in massive quantities and are not subject to the geopolitical restrictions often associated with rare earth metals or specialized catalysts. This availability mitigates the risk of supply disruptions, ensuring consistent delivery schedules for downstream customers who rely on these labeled intermediates for time-sensitive clinical trials or research programs.
- Cost Reduction in Manufacturing: The economic impact of switching to this novel oxidative protocol is profound, primarily driven by the simplification of the downstream processing train. In traditional heavy metal routes, a significant portion of the production budget is consumed by the necessity to scavenge trace metals to meet ppm-level specifications, often requiring multiple chromatography passes or specialized resin treatments. By completely avoiding heavy metals, this new method obviates those costly purification steps, resulting in a drastic reduction in solvent consumption, resin costs, and labor hours per kilogram of product. Furthermore, the high reaction yields reported in the patent, approaching quantitative conversion in many cases, mean that less starting material is wasted, directly improving the atom economy and reducing the raw material cost per unit of active pharmaceutical ingredient. These cumulative efficiencies create a leaner manufacturing model that offers substantial cost savings without compromising on the quality or isotopic enrichment of the final product.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness of the reaction conditions and the accessibility of the reagent portfolio. Unlike processes that require custom-synthesized catalysts or air-sensitive organometallic complexes, the reagents for this thioamide oxidation are shelf-stable and readily available from multiple global vendors. This multi-sourcing capability prevents single-point failures in the supply network and provides procurement teams with greater leverage in negotiations. Moreover, the reaction's tolerance to ambient temperatures and standard pressure conditions means it can be performed in a wider range of manufacturing facilities without the need for specialized cryogenic or high-pressure reactors. This flexibility allows for faster technology transfer between sites and easier scale-up, ensuring that production volumes can be ramped up quickly to meet surging demand from the biopharmaceutical sector without lengthy equipment qualification delays.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden inefficiencies, but this method is inherently designed for growth due to its benign thermal profile and lack of exothermic runaway risks associated with strong acid generation. The absence of corrosive trifluoromethanesulfonic acid extends the lifespan of reactor vessels and piping, reducing capital expenditure on equipment maintenance and replacement. From an environmental standpoint, the process aligns perfectly with green chemistry principles by minimizing the E-factor (mass of waste per mass of product), as the primary byproducts are organic and biodegradable rather than persistent heavy metal contaminants. This eco-friendly profile not only satisfies increasingly strict regulatory frameworks in Europe and North America but also enhances the corporate sustainability metrics of the manufacturing partner, a factor that is becoming increasingly weighted in vendor selection criteria by major multinational pharmaceutical companies committed to net-zero goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this heavy oxygen labeling technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear picture of what partners can expect when integrating this route into their supply chain.
Q: What are the primary advantages of this new method over traditional mercury-catalyzed routes?
A: The new method eliminates the use of highly toxic inorganic mercury salts and corrosive trifluoromethanesulfonic acid, significantly reducing hazardous waste generation and simplifying post-reaction purification processes.
Q: What is the labeling efficiency achievable with this thioamide oxidation process?
A: Experimental data from the patent indicates extremely high labeling efficiency, with specific examples demonstrating isotopic incorporation rates exceeding 95% for simple amides and over 85% for complex polypeptide structures.
Q: Is this process suitable for large-scale manufacturing of peptide intermediates?
A: Yes, the reaction operates under mild conditions at room temperature using commercially available reagents like N-bromosuccinimide, making it highly scalable and safer for commercial production compared to heavy metal protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heavy Oxygen Labeled Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced, greener synthesis routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in the laboratory can be faithfully reproduced on an industrial scale. We understand that isotopic purity is non-negotiable in proteomics and drug metabolism studies; therefore, our facilities are equipped with rigorous QC labs and state-of-the-art analytical instrumentation to verify stringent purity specifications and isotopic enrichment levels for every batch we produce. Our commitment to quality assurance means that every shipment of heavy oxygen labeled amides is accompanied by comprehensive documentation, giving R&D teams the confidence to proceed with their critical experiments without delay or uncertainty regarding material performance.
We invite procurement leaders and technical directors to engage with us to explore how this innovative mercury-free technology can optimize your specific project requirements. By leveraging our process development capabilities, we can provide a Customized Cost-Saving Analysis tailored to your target molecules, demonstrating exactly how the elimination of heavy metal steps impacts your bottom line. We encourage you to contact our technical procurement team today to request specific COA data for our standard labeled intermediates or to discuss route feasibility assessments for your proprietary compounds. Let us collaborate to build a more sustainable, efficient, and reliable supply chain for the next generation of isotopically labeled pharmaceutical intermediates.
