Advanced Organocatalytic Synthesis of Chiral Nitrogen Heterocycles for Commercial Pharmaceutical Manufacturing
Introduction to High-Efficiency Organocatalytic Synthesis
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral nitrogen-containing heterocycles, which serve as fundamental scaffolds in countless bioactive molecules. Patent CN102153501A introduces a groundbreaking approach utilizing chiral phosphoric acid or chiral thiourea catalysts to facilitate the intramolecular aza-Michael reaction of cyclohexadienone derivatives. This technology represents a significant leap forward in asymmetric synthesis, offering a metal-free pathway to generate chiral pyrrolidine, piperidine, and morpholine compounds with exceptional efficiency. Unlike traditional methods that often rely on expensive transition metals or stoichiometric chiral auxiliaries, this organocatalytic strategy operates under mild conditions while delivering superior stereocontrol. The ability to construct crowded chiral quaternary carbon centers with high fidelity makes this process particularly valuable for the production of complex alkaloids and pharmaceutical intermediates.
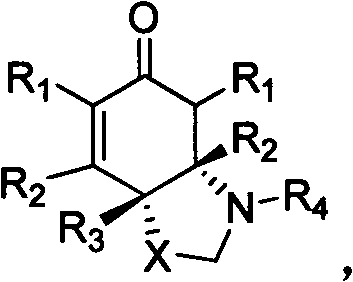
Furthermore, the versatility of this synthetic platform allows for broad substrate scope tolerance, accommodating various substituents on the cyclohexadienone ring without compromising yield or optical purity. For R&D directors and process chemists, this implies a reliable route for generating diverse libraries of chiral building blocks essential for drug discovery programs. The absence of metal residues simplifies downstream processing, aligning perfectly with stringent regulatory requirements for active pharmaceutical ingredients (APIs). As we delve deeper into the technical specifics, it becomes evident that this patent provides not just a laboratory curiosity, but a scalable solution for the commercial manufacturing of high-value chiral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral nitrogen heterocycles has relied heavily on transition metal catalysis or the use of chiral auxiliaries, both of which present significant drawbacks in a commercial setting. Transition metal catalysts, while effective, often introduce the risk of heavy metal contamination, necessitating costly and time-consuming purification steps to meet ppm-level specifications required by health authorities. Additionally, many metal-catalyzed processes require harsh reaction conditions, such as high temperatures or strong bases, which can limit functional group tolerance and lead to decomposition of sensitive substrates. The use of chiral auxiliaries, on the other hand, involves stoichiometric amounts of chiral material that must be attached and subsequently removed, resulting in poor atom economy and increased waste generation. These inefficiencies translate directly into higher production costs and longer lead times, creating bottlenecks for supply chain managers aiming for lean manufacturing operations.
The Novel Approach
In stark contrast, the methodology described in CN102153501A leverages the power of small-molecule organocatalysis to overcome these traditional hurdles. By employing chiral phosphoric acids or thioureas, the reaction proceeds through a highly organized transition state driven by hydrogen bonding interactions, eliminating the need for any metal salts. This metal-free nature is a decisive advantage for pharmaceutical manufacturing, as it drastically reduces the burden on quality control laboratories regarding residual metal testing. The reaction conditions are remarkably mild, often proceeding efficiently at room temperature or with gentle heating, which enhances energy efficiency and operational safety. Moreover, the catalytic loading is low, typically ranging from 1 mol% to 20 mol%, yet it delivers impressive yields between 73% and 97% with enantiomeric excess values soaring up to 99%. This combination of high efficiency, environmental compatibility, and operational simplicity positions this novel approach as a superior alternative for the cost reduction in pharmaceutical intermediates manufacturing.
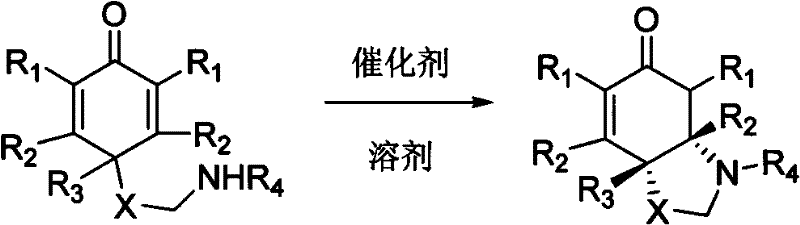
Mechanistic Insights into Organocatalytic Asymmetric Aza-Michael Reaction
The success of this synthetic route lies in the precise molecular recognition and activation provided by the chiral organocatalysts. Chiral phosphoric acids, derived from BINOL scaffolds, act as Brønsted acids that simultaneously activate the electrophilic enone moiety and orient the nucleophilic amine through a dual hydrogen-bonding network. This bifunctional activation lowers the energy barrier for the cyclization while enforcing a rigid chiral environment that dictates the facial selectivity of the attack. Similarly, chiral thioureas function by stabilizing the developing negative charge on the carbonyl oxygen via hydrogen bonding, effectively increasing the electrophilicity of the Michael acceptor. The structural rigidity of catalysts like those shown in the patent ensures that the substrate is locked in a specific conformation, leading to the formation of a single enantiomer with high fidelity.
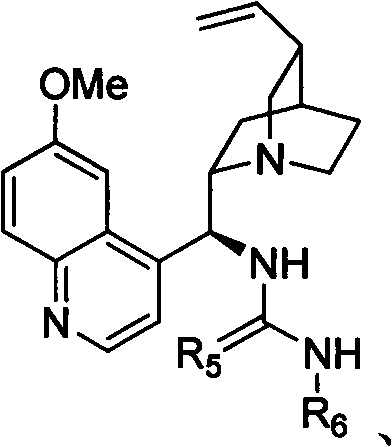
Understanding the impurity profile is crucial for process validation, and this mechanism inherently suppresses side reactions. Because the catalysis is highly specific, competing pathways such as polymerization or non-selective background reactions are minimized. The patent data demonstrates that even with diverse substituents ranging from electron-donating methoxy groups to electron-withdrawing halogens, the stereochemical outcome remains consistent. For instance, substrates bearing bulky aryl groups still achieve excellent ee values, indicating that the catalyst pocket is sufficiently spacious yet discriminating. This robustness against structural variations means that process engineers can expect consistent quality across different batches, reducing the risk of off-spec material. The ability to fine-tune the catalyst structure, as evidenced by the variety of phosphoric acid and thiourea derivatives exemplified in the patent, allows for further optimization tailored to specific difficult substrates, ensuring that impurity levels remain well within acceptable limits for GMP production.
How to Synthesize Chiral Nitrogen Heterocycles Efficiently
Implementing this technology in a production environment requires careful attention to reaction parameters to maximize the benefits of the organocatalytic system. The general procedure involves dissolving the cyclohexadienone precursor in a suitable organic solvent, such as dichloromethane, toluene, or acetonitrile, under an inert atmosphere to prevent moisture interference. The choice of solvent can influence both the reaction rate and the stereoselectivity, with non-polar solvents often favoring tighter ion-pairing interactions essential for high ee. Once the substrate is in solution, the chiral catalyst is introduced, and the mixture is stirred at the prescribed temperature, which can range from cryogenic conditions for highly sensitive substrates to reflux for more robust systems. Monitoring the reaction via TLC or HPLC ensures that the conversion is complete before workup, which typically involves simple extraction and chromatography. The detailed standardized synthesis steps for specific derivatives are outlined below to guide technical teams in replicating these high-performance results.
- Prepare the reaction system by dissolving cyclohexadienone derivatives in an organic solvent such as dichloromethane or toluene under inert atmosphere.
- Add the chiral organocatalyst, either a chiral phosphoric acid or chiral thiourea derivative, at a molar ratio ranging from 1: 0.01 to 1:0.5 relative to the substrate.
- Maintain the reaction temperature between -78°C and 100°C depending on the specific substrate reactivity, monitoring progress until completion before purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic technology translates into tangible strategic benefits that extend beyond mere chemical yield. The elimination of transition metals from the synthesis route removes a major cost center associated with metal scavenging resins and extensive analytical testing for heavy metals. This simplification of the purification train not only reduces the consumption of expensive consumables but also shortens the overall cycle time from raw material to finished intermediate. Furthermore, the high atom economy and catalytic nature of the reaction mean that less raw material is wasted, contributing to substantial cost savings in manufacturing overheads. The mild reaction conditions also imply lower energy consumption for heating or cooling, aligning with corporate sustainability goals and reducing the facility's carbon footprint. These factors combined create a more resilient and cost-effective supply chain capable of responding quickly to market demands.
- Cost Reduction in Manufacturing: The primary driver for cost efficiency in this process is the removal of precious metal catalysts and chiral auxiliaries. Traditional methods often require palladium, rhodium, or stoichiometric amounts of chiral starting materials, all of which carry high price tags and volatile market values. By switching to organocatalysts, which are generally easier to synthesize and handle, the direct material cost is significantly lowered. Additionally, the high yields reported in the patent, often exceeding 90%, minimize the loss of valuable intermediates, ensuring that every kilogram of input generates maximum output. The simplified workup procedure further reduces labor costs and solvent usage, making the overall process economically superior to legacy methods.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the scarcity of specialized reagents or the complexity of multi-step syntheses. This method utilizes readily available cyclohexadienone derivatives and robust organocatalysts that can be sourced or manufactured in bulk with high consistency. The operational simplicity reduces the dependency on highly specialized equipment or extreme conditions that might cause unplanned downtime. Moreover, the high selectivity reduces the formation of difficult-to-separate diastereomers, streamlining the purification process and ensuring faster turnaround times. This reliability allows procurement teams to negotiate better terms with confidence, knowing that the production schedule is less likely to be disrupted by technical failures or quality issues.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often reveals hidden challenges, but this organocatalytic route is designed with scalability in mind. The absence of exothermic metal-catalyzed steps reduces safety risks associated with thermal runaways, facilitating safer scale-up to multi-ton quantities. From an environmental perspective, the metal-free nature of the process significantly reduces the toxicity of the waste stream, simplifying wastewater treatment and disposal compliance. The use of common organic solvents and the potential for solvent recovery further enhance the green chemistry profile of the operation. This alignment with environmental regulations mitigates the risk of future compliance costs and supports the company's commitment to sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the implementation and capabilities of this technology, we have compiled answers based on the technical data provided in the patent documentation. These questions reflect the practical concerns of technical directors and quality assurance teams who are evaluating new synthetic routes for integration into their existing portfolios. Understanding the nuances of catalyst loading, substrate scope, and downstream processing is essential for making informed decisions about technology transfer. The following responses provide clarity on how this method performs under various conditions and its suitability for producing high-purity pharmaceutical ingredients.
Q: What are the primary advantages of using organocatalysts over metal catalysts for chiral nitrogen heterocycles?
A: Organocatalysts eliminate the risk of heavy metal contamination, which is critical for pharmaceutical compliance. They also offer easier removal processes and often operate under milder conditions compared to transition metal complexes.
Q: What level of enantioselectivity can be achieved with this synthetic method?
A: The patent data indicates that this method consistently achieves high enantiomeric excess (ee) values, typically ranging from 89% to 99%, ensuring the production of high-purity chiral intermediates suitable for drug synthesis.
Q: Can this methodology be applied to the synthesis of complex alkaloids like (-)-Mesembrine?
A: Yes, the technology is specifically validated for the synthesis of (-)-Mesembrine. The chiral pyrrolidine intermediates generated serve as key precursors that can be further transformed into the target alkaloid through standard reduction and functionalization steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Nitrogen Heterocycles Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic technologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the potential of the organocatalytic methods described in CN102153501A and is fully prepared to leverage this knowledge for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high enantioselectivity and yields observed at the lab scale are faithfully reproduced in our manufacturing facilities. Our rigorous QC labs and stringent purity specifications guarantee that every batch of chiral nitrogen heterocycles meets the highest international standards, providing you with a reliable source of high-purity pharmaceutical intermediates.
We invite you to collaborate with us to explore how this advanced synthesis method can optimize your supply chain and reduce your overall production costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments for your target molecules. Let us partner with you to transform innovative patent chemistry into commercial reality, driving efficiency and quality in your drug development pipeline.
